


Fossil freshwater sponges: Taxonomy, geographic distribution, and critical review
ROBERTO PRONZATO, ANDRZEJ PISERA, and RENATA MANCONI
Pronzato, R., Pisera, A., and Manconi, R. 2017. Fossil freshwater sponges: Taxonomy, geographic distribution, and critical review. Acta Palaeontologica Polonica 62 (3): 467–495.
Sponges are one of the most ancient animal phyla with about 8850 living species and about 5000 described fossil taxa. Most sponges are marine and live at all depths of all oceans. Freshwater bodies (lakes, rivers) are inhabited only by a small minority of species, ca. 240 (< 3%) comprising the order Spongillida (Demospongiae) most of which are able to produce specialized resting bodies to survive harsh terrestrial environmental conditions. This highly disproportionate ratio of marine and freshwater sponges is even more accentuated in the field of palaeontology with rare records reported up to the Miocene (< 0.4% of all known fossil sponges). Only a few fossil taxa were correctly supported by strong and convincing taxonomic morphotraits at genus and species level, thus we provide here an overview of fossil freshwater sponges focusing on their morphotraits and distribution in time and space. Each recorded taxon is described in detail following the modern taxonomy and nomenclature. All fossil data suggest a clear trend of long term conservative morphology in the evolutionary history of Spongillida, although some traits of Recent gemmules evolved in a wide array of adaptive morpho-functional novelties. The majority of accepted fossil species belongs to the cosmopolitan family Spongillidae. The genera Oncosclera and Potamophloios of the family Potamolepidae seem to have had, in the past, a much larger geographic range than today. A synthesis of fossil taxa morphotraits is also provided in an Appendix 1.
Key words: Porifera, Spongillida, fossil record, morphotraits, palaeobiogeography.
Roberto Pronzato[pronzato@dipteris.unige.it], Dipartimento di Scienze della Terra, dell’Ambiente e della Vita (DISTAV) Università di Genova, Corso Europa 26, 16132 Genova, Italia.
Andrzej Pisera [apis@twarda.pan.pl] (corresponding author), Institute of Paleobiology, Polish Academy of Sciences, ul. Twarda 51/55, 00-818 Warszawa, Poland.
Renata Manconi [r.manconi@uniss.it], Dipartimento di Scienze della Natura e del Territorio (Dip.Ne.T.), Università di Sassari, Via Muroni 25, 07100, Sassari, Italy.
Received 13 February 2017, accepted 3 July 2017, available online 4 September 2017.
Copyright © 2017 R. Pronzato et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The fossil record of marine siliceous sponges, albeit punctuated and biased toward taxa with fused/articulated skeletons, is relatively good and well studied (Rezvoi et al. 1971; Pisera 1999; Rigby 2004). The oldest bodily preserved fossil described as a hexactinellid sponge Paleophragmodictya, described by Gehling and Rigby (1996) from the Ediacaran of Southern Australia (late Proterozoic), might be questioned but since the lower Cambrian the presence of undoubted siliceous sponges, certainly belonging to Hexactinellida, is proven by the presence of loose spicules (Chang et al. 2017). On the other hand, the fossil record of freshwater sponges is very poor, much more punctuated (except for the Neogene) and the first undoubted forms are late Palaeozoic in age (Pisera and Saez 2003). Recently, a report of the doubtful presence of gemmules (indicating possible occurrence in freshwater) from the Ediacaran period was also published (Du et al. 2015). This rarity of fossil occurrences, is without doubt a taphonomical effect and not a real phenomenon, as indicated by recent Eocene finds (Pisera 2010; Pisera et al. 2014). The skeleton of freshwater sponges consists of only loose spicules bound together by spongin, that decays after the sponge’s death, and apart from some special situations, only loose, disassociated spicules are preserved. The very limited set of diagnostic morphotraits makes determination and comparative analysis of any fossils sponge a difficult matter because, apart from spicules shape and ornamentation, their arrangement and association in the sponge body architecture are important traits in freshwater sponge taxonomy.
Another problem is that there are no good descriptions of the freshwater skeleton architecture in palaeontological literature and commonly marine sponges (that differ considerably) are used for comparisons (see for example Du et al. 2015) leading to misunderstandings. To overcome this problem, we have included here illustrations of morphotraits typical of Recent freshwater sponges, i.e., spicules and gemmule structure, to facilitate further fossil studies.
It is also important to note that the presence of gemmules is not an exclusive trait of freshwater sponges because the ability to form gemmule-like resting bodies, however rare, is known also among marine sponges (Fell 1974, 1993; Weert 2002; Pisera et al. 2010 and literature therein; Marra et al. 2013; Melis et al. 2016) and fossil marine gemmules are also known (Petit and Charbonnier 2012).
The literature reports few records of fossil freshwater sponge taxa, i.e., 13 identified at the species level, 3 to order level (Spongillida), and one possibly a spongillid but in need of confirmation. This latter “tentative sponge” would be the most ancient (Ediacaran) record of fossil freshwater sponges. The oldest confirmed fossil record of Spongillida is from the upper Carboniferous. Fossil occurrences are scattered up to the Miocene in the northern and southern hemisphere, mainly in America, Africa, Europe, and Asia.
From a taxonomic point of view, one species (Palaeospongilla chubutensis) represents the exclusively fossil family Palaeospongillidae, two (Oncosclera kaniensis and Potamophloios canadensis) belong to the family Potamolepidae, and eight to the family Spongillidae. As for two of these latter species, new monospecific genera were erected, i.e., Eospongilla and Lutetiospongilla. Four species are ascribed to the extant genus Ephydatia (E. chileana, E. gutenbergiana, E. cf. facunda, and E. kaiseri) and two to the extant genus Spongilla (S. patagonica and S. purbekensis).
Generic attribution of some of these species, especially those from the Mesozoic, is questionable, owing to the few available traits as well as from an evolutionary point of view. However, Palaeogene and Neogene (or even older) taxa that are attributed to Recent genera or even species are common, not only among sponges, e.g., the hexactinellid Laocoetis is known since the Late Jurassic (see Vodrážka and Crame 2011; Pisera and Tabachnick 2014) or the sphinctozoan demosponge Vaceletia known since the Eocene (Frisone et al. 2016). Among brachiopods, e.g., Megathiris detruncata, is known since the Eocene (Bitner 1990), and Argyrotheca since the Cretaceous (Bitner and Pisera 1979). It has been confirmed recently (Pisera and Tabachnick 2014) that the hexactinellid sponges of genus Laocoetis, reported since the Jurassic to the Recent (Pisera 1997; Rigby 2004; Vodrážka and Crame 2011) indeed belong to the same morphogenus. Thus one cannot reject a priori that some Palaeogene or Neogene sponge taxa that are identical morphologically are conspecific with extant species, although some traits of Recent gemmules evolved in a wide array of morphological novelties.
To clarify some doubtful aspects of the fossil record of freshwater sponges we provide here a revision based on the literature of all known fossil freshwater sponges (Porifera: Demospongiae: Spongillida) reported up to the Miocene.
Order Spongillida Manconi and Pronzato, 2002
Species richness of sponges of the order Spongillida Manconi and Pronzato, 2002 inhabiting continental waters (previous suborder Spongillina Manconi and Pronzato, 2002, and subsequent emendations 2011, 2015) reach 240 valid species (about 3%) of the 8850 species of all extant Porifera (Hooper et al. 2011; Van Soest et al. 2017). The order Spongillida comprises 48 genera belonging to 6 families i.e., Metaniidae, Spongillidae, Lubomirskiidae, Malawispongiidae, Potamolepidae, and the exclusively fossil Palaeospongillidae. The monophyletic Spongillida (Manconi and Pronzato 2002, 2015, 2016a, b; Erpenbeck and Wörheide 2007; Cárdenas et al. 2012; Morrow and Cardenas 2015) according to earlier studies, colonized continental water not later than in the Triassic from a hypothetical marine Haplosclerida ancestor (Pronzato and Manconi 1994; Manconi and Pronzato 2002, 2015).
Spongillida are widespread on all continents, excluding Antarctica, and several species, possible species complexes, show a wide geographic range (Manconi and Pronzato 2007, 2008, 2015, 2016a, b). Several studies focus on their occurrence under extremely stressing conditions e.g., temporary water (permafrost) in circumpolar northern regions, temporary water in hot deserts, small temporary ponds in sub arid-arid conditions, and tropical hydrographic basins characterised by flooding and extreme water level variations (Manconi and Pronzato 2008, 2015, 2016b).
The abundance of freshwater sponges varies widely from region to region, and in some cases sponge populations are so dense and flourishing that they strongly impact human health and/or productive activities. In the Amazon basin, for example siliceous spicules of sponges are so abundant in the water column, during the raining season, that they are causing permanent eye damage (Volkmer-Ribeiro et al. 2006) to riverine populations. In the lower Mekong basin (Thailand) high biomass of large sponges heavily colonizes fishermen nets, damaging the fishery and aquaculture infrastructures (Manconi et al. 2013). On the contrary, sponges are very rare or absent in some Mediterranean islands, in the Canary Archipelago, and several Pacific Ocean archipelagos (Manconi and Pronzato 1994a, 2009, 2015; Manconi 2008).
Freshwater sponges display highly variable body shape and dimensions, consistency and colour. Specimens range from thin whitish crusts a few mm thick strictly adhering to the substratum, to dark brown massive cushions, and to green branching or erected growth forms. In most species the body consistency is soft and fragile while in other species it is hard with a notably compact texture (Fig. 1). Under unfavourable conditions, such as harsh climate periods, freshwater sponges are reduced, by cryptobiosis, to small spherules known as gemmules (resting bodies) in carpets on the substratum, scattered in the skeletal network, or grouped at the sponge basal portion; but they can be also dispersed in sediments and water column.
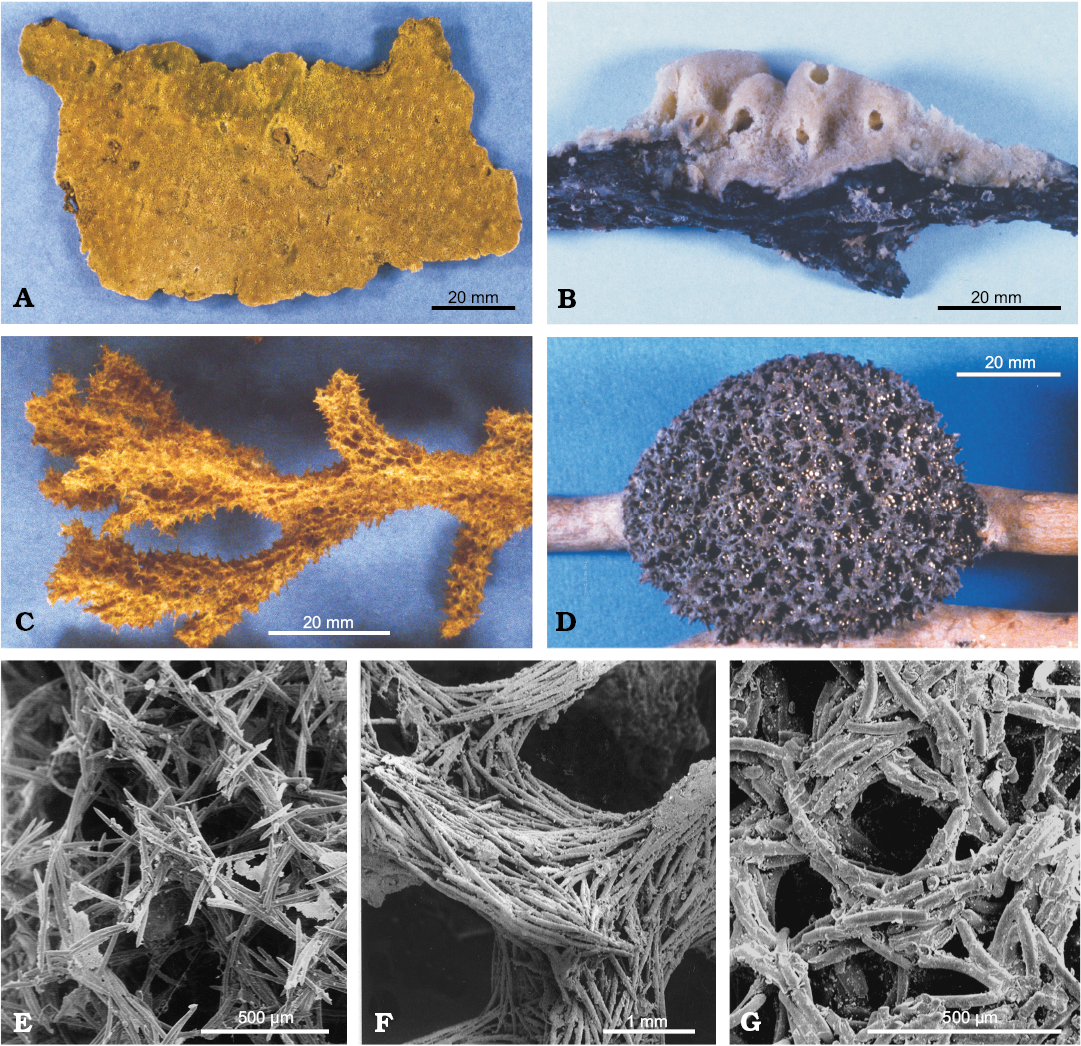
Fig. 1. Growth forms (A–D) and skeletal architecture with variable amount of spongin (E–G) of Recent freshwater sponges of the order Spongillida. A. Oncosclera jewelli (Volkmer-Ribeiro, 1963), Neotropical Region. B. Stratospongilla sp. C. Metania rhodesiana (Burton, 1938), Afrotropical Region. D. Drulia sp. E. Heterorotula capewelli (Bowerbank, 1863), Australasian Region. F. Drulia browni (Bowerbank, 1863), Neotropical Region. G. Uruguayella repens (Hinde, 1888), Neotropical Region. E–G SEM images.
The main diagnostic traits that enable us to identify Spongillida are few i.e., skeletal architecture, range of spicule arrangement, geometry, size and shape of spicules, and gemmular traits. Skeletal network is a reticulum of siliceous spicules associated with a notably variable amount of spongin. Spicules, both megascleres (Fig. 2) and microscleres (Fig. 3) are monaxial from smooth i.e., oxeas and strongyles, to spiny i.e., acanthoxeas and acanthostrongyles. Gemmules of freshwater sponges are subspherical to hemispherical (100–1200 µm in diameter, Fig. 4), and bear a structured coat of spongin (gemmular theca) variably armed by the spicules (gemmuloscleres) to protect a mass of staminal, totipotent cells contained inside. Morphotraits of this resting stage (i.e., gemmular cage, gemmular theca, gemmular foramen, arrangement of spicules, architecture of spongin structures, together with shape and ornamentations of spicules) are notably diversified and diagnostic at the genus and species level (Manconi and Pronzato 2002) (Fig. 4). Particularly important, from a taxonomic and phylogenetic point of view, are gemmuloscleres (Fig. 5).
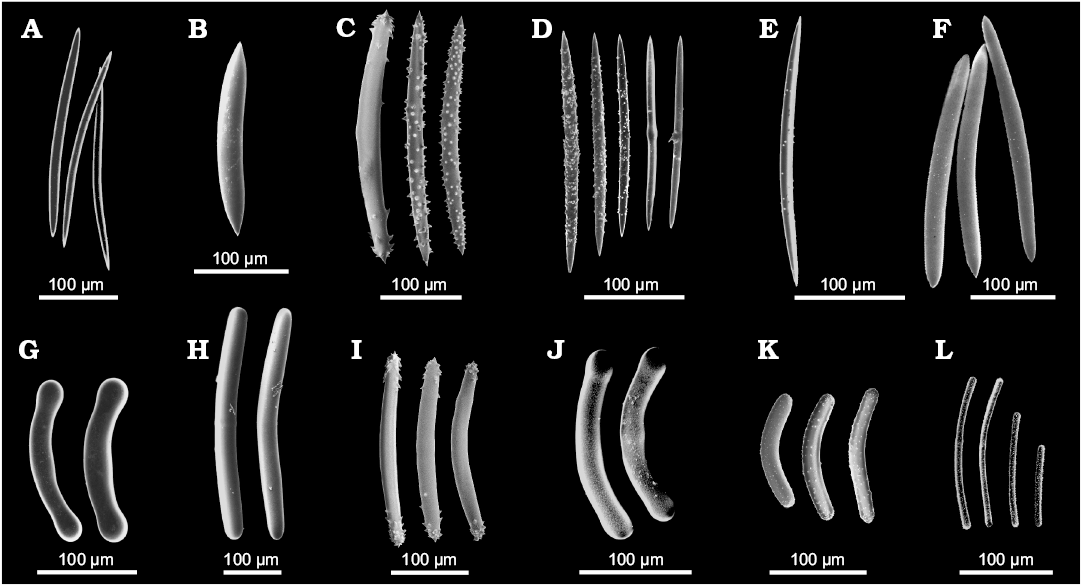
Fig. 2. Morphology of megascleres of Recent freshwater sponges of the order Spongillida. A–F. Oxeas. A. Spongilla lacustris (Linnaeus, 1759), Palaearctic-Nearctic Region, slim smooth oxeas. B. Drulia browni (Bowerbank, 1863), Neotropical Region, stout smooth oxeas. C. Lubomirskia baikalensis (Pallas, 1771), Palaearctic Region, spiny oxeas. D. Makedia tanaensis Manconi, Cubeddu, and Pronzato, 1999, Afrotropical Region, variously spiny slim oxeas. E. Anheteromeyenia argyrosperma (Potts,1880), Nearctic Region, irregularly micro-spiny oxea. F. Cortispongilla barroisi (Topsent, 1892), Palaearctic Region, irregularly micro-granulated oxea. G–L. Strongyles. G. Potamolepis marshalli Burton, 1938, Afrotropical Region, smooth, stout strongyles with inflated tips. H. Sterrastrolepis brasiliensis Volkmer-Ribeiro and De Rosa Barbosa, 1978, Neotropical Region, smooth strongyles. I. Baikalospongia bacillifera Dybowsky, 1880, Palaearctic Region, strongyloxeas with hooked tips. J. Potamolepis micropora Burton, 1938, Afrotropical Region, strongyles with microspines and inflated tips. K. Swartschewskia papyracea (Dybowsky, 1880), Palaearctic Region, spiny strongyles. L. Nudospongilla cunningtoni (Kirkpatrick, 1906), Afrotropical Region, spiny, slim strongyles. SEM images. Modified from Manconi and Pronzato (2002, 2009).
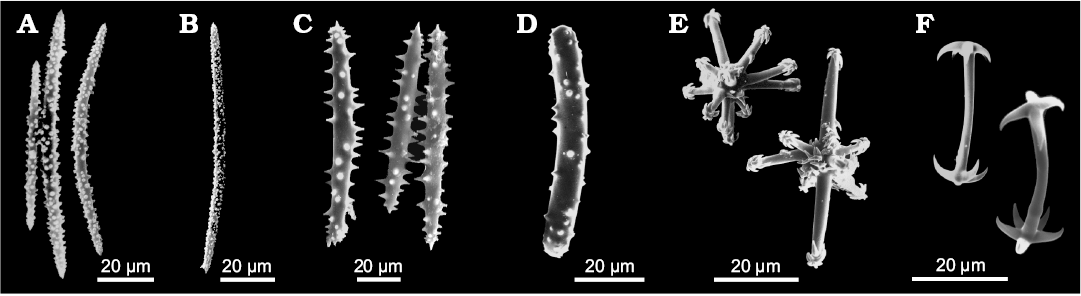
Fig. 3. Morphology of microscleres of Recent freshwater sponges of the order Spongillida. A–C. Microxeas variously ornate by spines or tubercles. A. Spongilla lacustris (Linnaeus, 1759), Palaearctic-Nearctic Region. B. Sterrastrolepis brasiliensis Volkmer-Ribeiro and De Rosa Barbosa, 1978, Neotropical Region. C. Drulia browni (Bowerbank, 1863), Neotropical Region. D. Microstrongyle with spines of Metania reticulata (Bowerbank, 1863), Neotropical Region. E. Aster-like microscleres with hooks of Dosilia plumosa (Carter, 1849), Oriental Region. F. Pseudomicrobirotules of Corvospongilla burmanica (Kirkpatrick, 1908), Oriental Region. SEM images. Modified from Manconi and Pronzato (2002).
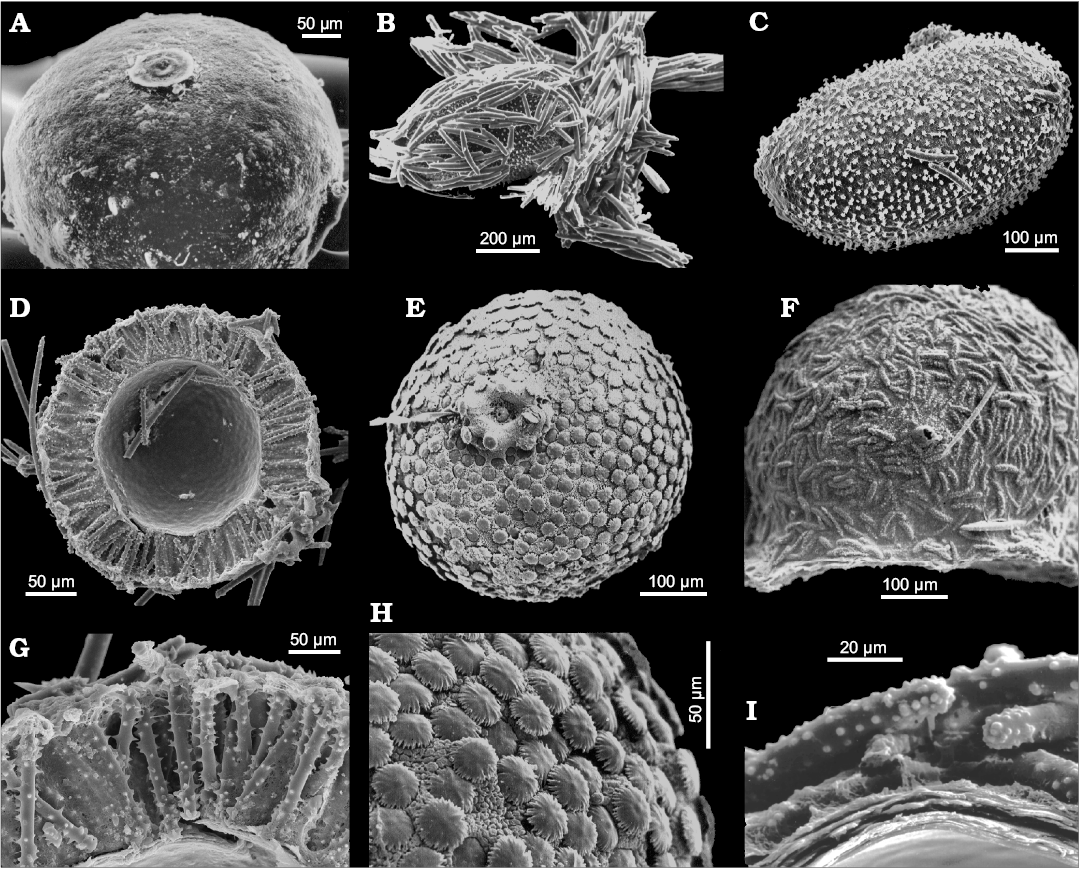
Fig. 4. Gemmule morphology (resting bodies) of Recent freshwater sponges of the order Spongillida. A. Spongilla lacustris (Linnaeus, 1759), Palaearctic-Nearctic Region, subspherical unarmed gemmular theca. B. Metania reticulata (Bowerbank, 1863), Neotropical Region, cage of tangential megascleres around the gemmule. C. M. reticulata, ovoid gemmule (removed from the cage, see B) with radial birotules in the gemmular theca. D. Radiospongilla crateriformis (Potts, 1882), Cosmopolitan, gemmuloscleres radially arranged in the theca (cross section). E. Umborotula bogorensis (Weber, 1890), Palaearctic-Oriental-Australasian Region, subspherical gemmule with distal rotules of gemmuloscleres at the theca surface. F. Stratospongilla bombayensis (Carter, 1882), Oriental-Afrotropical Region, hemispherical gemmule with tangential gemmulosclere at the surface. G. R. crateriformis, gemmular theca with radial gemmuloscleres (close up, cross section). H. U. bogorensis, gemmule surface with distal rotules of birotule gemmuloscleres (close up). I. Oncosclera rousseleti (Kirkpatrick, 1906), Afrotropical Region, gemmule surface with tangential strongyle gemmuloscleres (close up, cross section). SEM images. Modified from Manconi and Pronzato (2002, 2005, 2009).
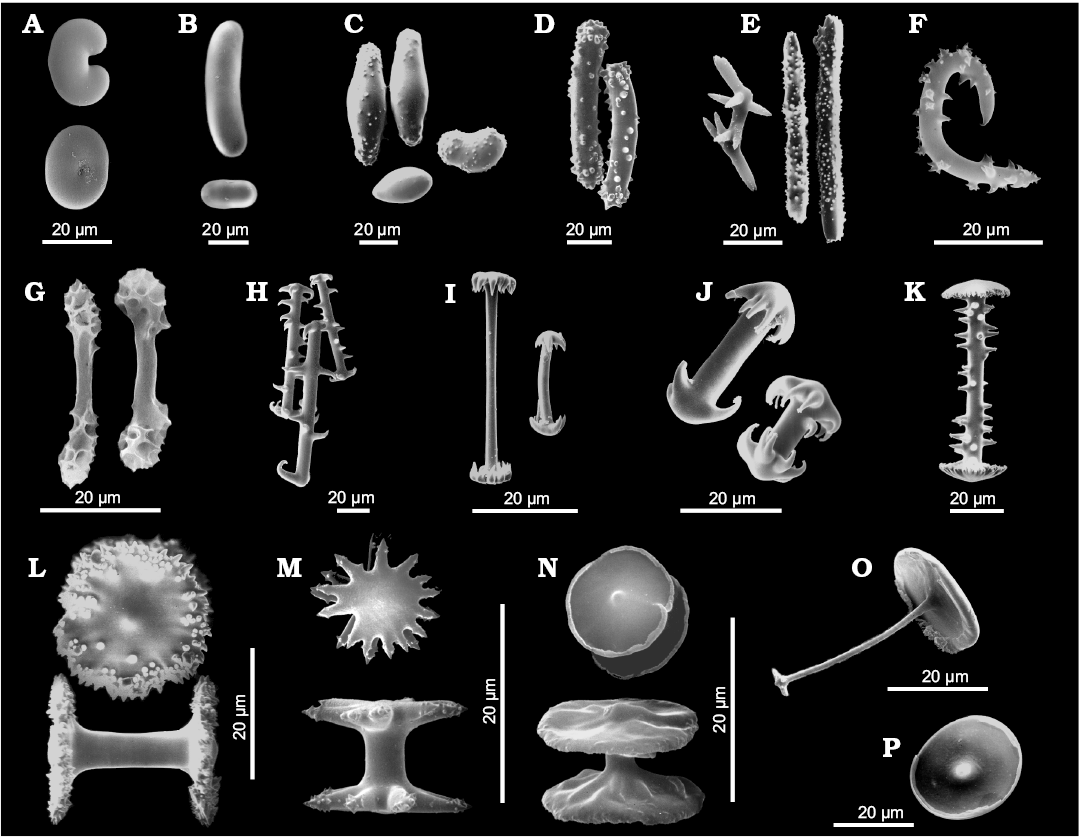
Fig. 5. Morphology of gemmuloscleres of Recent freshwater sponges of the order Spongillida. A. Cherokeesia armata Copeland, Pronzato, and Manconi, 2015, Nearctic Region, bean-like to ovoid strongyles. B. Potamophloios stendelli (Jaffé, 1916), Afrotropical Region, elongate to ovular strongyles. C. Oncosclera jewelli (Volkmer-Ribeiro, 1963), Neotropical Region, irregularly ovoid strongyles smooth or covered with short spines and tubercles. D. Corvospongilla loricata (Weltner, 1895), Afrotropical Region, slightly curved, stout, spiny strongyles. E. Sanidastra yokotonensis Volkmer-Ribeiro and Watanabe, 1983, Palaearctic Region, oxeas with 2–9 rays rounded to pointed or tricuspidate at tips. F. Spongilla lacustris (Linnaeus, 1759), Nearctic-Palaearctic Region, oxeas strongly bent with large spines. G. Pectispongilla aurea Annandale, 1909, Oriental Region, botryoidal gemmuloscleres with smooth shaft with at the apex polygonal concavities of the botryum. H. Anheteromeyenia argyrosperma (Potts, 1880), Nearctic Region, birotules with spiny shafts and circular rotules with crenulated to indented margins. I. Corvomeyenia everetti (Mills, 1884), Nearctic Region, pseudobirotules with straight, smooth shafts and pseudo-rotules at the apex (up to 10 hooks). J. Acalle recurvata (Bowerbank, 1863), Neotropical Region, pseudobirotules with cylindrical shaft and hooks of umbonate pseudorotules stout and notably recurved bearing microspines at their apices. K. Dosilia plumosa (Carter, 1849), Oriental Region, birotules with large spines on the straight shaft and flat to slightly umbonate rotules with margins bearing small blunt, recurved teeth (spines). L. Racekiela ryderi (Potts, 1882), Nearctic-Neotropical-Palaearctic Region, birotules with smooth shafts and large disk-like rotules with serrated margins and scattered spines or tubercles. M. Ephydatia muelleri (Lieberkühn, 1855), Afrotropical-Nearctic-Palaearctic Region, birotules with smooth, short shaft and rotules with indented margins. N. Trochospongilla horrida (Weltner, 1893), Nearctic-Palaearctic Region, birotules with smooth, short shaft and approximately circular rotules with irregularly thickened margins. O. Acalle recurvata (Bowerbank, 1863), Neotropical Region, tubelliform gemmuloscleres with the proximal large irregularly circular flat rotule with entire margin supporting a smooth shaft decreasing in thickness toward the distal end shaped as umbonate knob-like rotule with a few teeth. P. Drulia browni (Bowerbank, 1863), Neotropical Region, parmuliform gemmuloscleres with the single circular proximal rotule, with lower concave surface, supporting a distal short acute conical stem (shaft). SEM images. Modified from Manconi and Pronzato (2001, 2002) and Copeland et al. (2015).
Identification of gemmule-producing sponge species, however, is usually not possible when gemmules are absent from specimens during a phase of their life cycle. Moreover, a number of freshwater sponges, mostly belonging to taxa endemic to ancient lakes, do not produce gemmules. In the palaeontological context, most of the diagnostic traits mentioned above are not useful. Skeletal architecture/network is usually not preserved because spicules joined with spongin become dispersed after its decay. The main character in palaeontological studies is morphology of gemmuloscleres and, to a lesser degree, microscleres, but these are limited only to certain taxa, as indicated above. Megascleres are usually not characteristic enough for generic determination, but some inferences can be made in some cases, e.g., presence of strongyles vs. oxeas.
If the about 240 species of living freshwater sponges are a small minority compared to the marine ones, among the 5000 of all fossil sponge taxa (Hooper et al. 2011) freshwater species prior to the Quaternary, are, putatively, no more than 16. Their geographic distribution is largely scattered in all continents, except Australia and Antarctica (Fig. 6). Considering the palaeogeography, fossil freshwater sponge taxa seem to have preferred temperate and tropical climates (Fig. 7).
All discussed here freshwater sponge taxa are Miocene or older in age. Sponges from younger deposits, generally ascribed to extant genera and species, are not included. The sixteen taxa are arranged in chronological order, beginning from the oldest to the most recent. Measurements are reported as length and/or length × thickness.
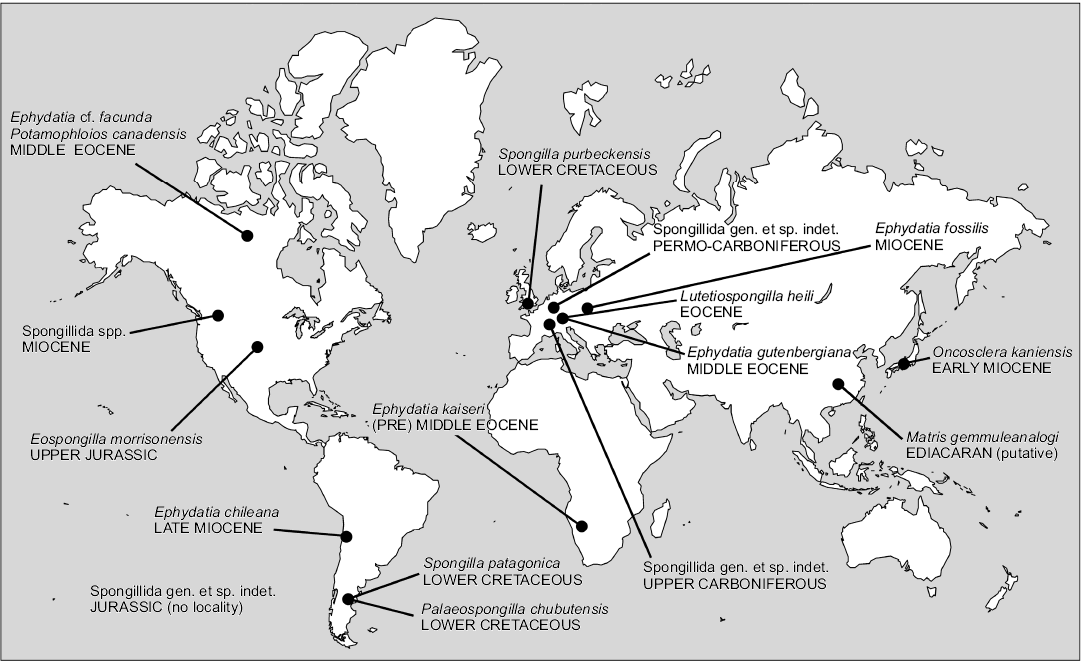
Fig. 6. Geographic distribution of fossil freshwater sponges (Porifera, Demospongiae, Spongillida).
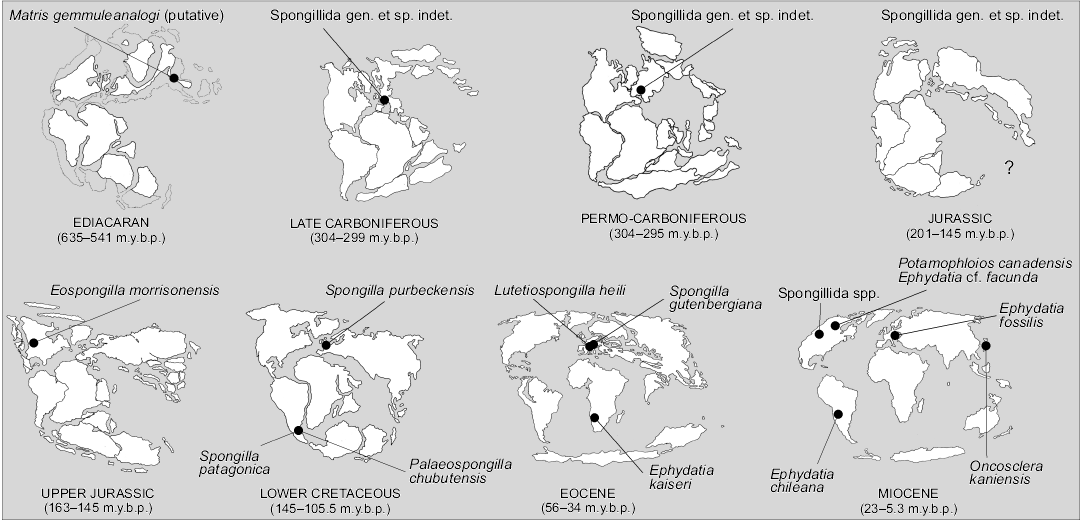
Fig. 7. Palaeogeographic distribution of fossil freshwater sponges (Porifera, Demospongiae, Spongillida).
Institutional abbreviations.—MABA, Museo Argentino de Ciencias Naturales “Bernardino Rivadavia”, Buenos Aires, Argentina; PWL, Naturhistorisches Museum (Palaeontological Collection), Mainz, Germany; SMF, Senckenberg Museum Frankfurt, Germany; TMNH, Toyohashi Museum of Natural History, Japan; USNM, National Museum of Natural History, Smithsonian Institution, Washington D.C., USA (formerly United States National Museum).
Systematic review
Matris gemmuleanalogi Du, Wang, and Komiya, 2015
Fig. 8.
Type horizon: Ediacaran.
Type locality: Yangtze Gorges area, South China.
References: Du et al. 2015.
Description (emended from Du et al. 2015).—Putative unarmed gemmules (80–100 µm in diameter). Putative siliceous spicules remnants (7–10 × 1–2 µm).
Remarks.—The main problem in evaluating this report is the very low quality of SEM pictures and the fact that inmost cases the objects are covered/sunken in a (?) glue. The sponge affinity of the studied material is based on general shape and size of the described objects, their allegedly tri-layered wall structure and presence of siliceous spicules. Detailed examination of both SEM and thin section photos allow us to conclude that:
(i) Alleged spicules do not resemble any of spicules known to science because they are irregular; additionally, they are very small, too small for megascleres or even gemmuloscleres. Most probably they are mineral grains.
(ii) Compared to many other fossil remains of Porifera, even more recent, these “spicules” do not show any kind of diagenetic corrosion. As a consequence the animal origin of this siliceous material is strongly questionable.
(iii) As for presumed gemmules, considering their size and shape it is not possible to exclude a priori that these remains are gemmules of sponges, but this is highly improbable. There is no evidence of a protective layer of gemmuloscleres on the surface (it is suggested that spicules occur inside of the presumed gemmules); of the 3 layers described in the text, only one layer is visibly encrusted from inside with, most probably, pyrite framboids.
(iv) The morphology of the isolated spheres only vaguely resemble gemmules but is nearly identical in shape to some Arcellinida (known already from the Cambrian; especially Recent genus Arcella), that have a foramen located in a depression at one pole.
The oldest known certain fossils of sponges are all marine and the earliest are late Palaeozoic in age, with no trace of older freshwater sponges. If the report of Du et al. (2015) is accepted as true, it would mean that continental sponges appeared first and later colonized the sea, which seems very improbable considering the fossil record. Remnants of gemmules have never been found among abundant marine spiculitic rocks; in this case gemmules are abundant (and described as intact) while the spicules are rare, a rather unusual situation.
In conclusion the supposed Ediacaran (Precambrian) sponge gemmules show no features that allow their attribution to Porifera, especially freshwater sponges, but resemble some arcellid testate amoebae.
The text states that the rocks from which gemmules are described are considered as marine deposits “marine Ediacaran environment” (Du et al. 2015). Thus there is a clear discrepancy between how these object are treated (like freshwater sponge gemmules) and rocks where they were discovered (marine rocks).
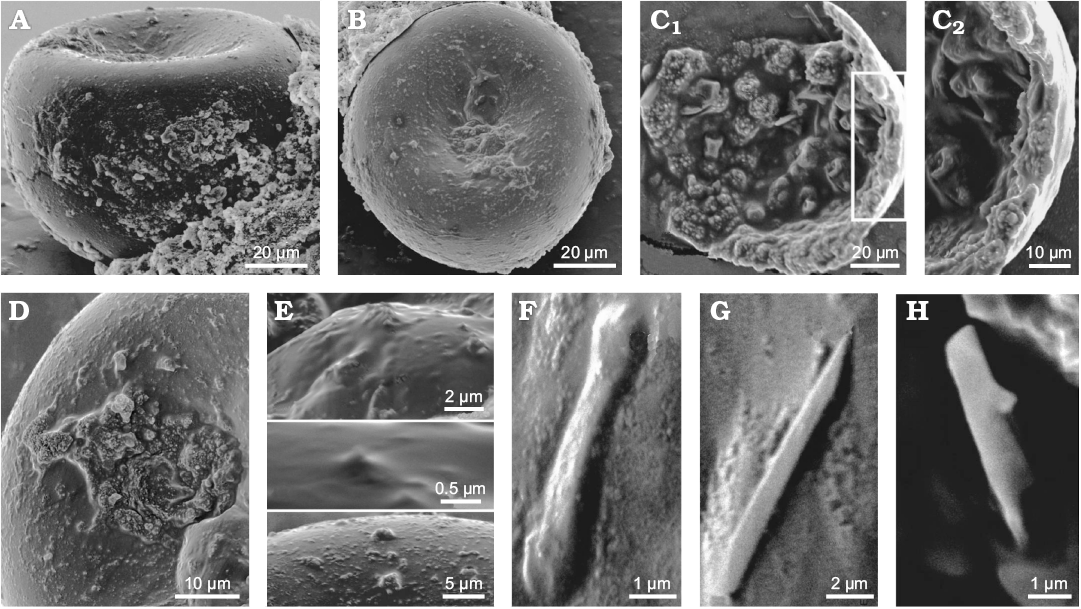
Fig. 8. Matris gemmuleanalogi Du, Wang, and Komiya, 2015, from the Ediacaran of Yangtze Gorges area, South China (originally interpreted as Ediacaran freshwater sponge but most likely representing amoebe tests). A. Gemmule-like structure in lateral view. B. Gemmule-like structure view from the top. C. Internal microstructures of a broken fossil. Gemmuloscleres-like structures are projected into the coat of the broken gemmule-like; C2, detail of C1 showing three possible layers of a coat (cross-section): a thin homogeneous inner layer, a pneumatic layer-like structure embedded with gemmuloscleres-like structure and a naked outer layer. D. Gemmule like surface with a possible gemmuloscleres protruding in the middle. E. Spine and tubercles on the gemmule-like surface. F. Short, dumbbell-like structure. G. Raphide microsclere-like structure. H. Gemmuloscleres-like structure. SEM images. Modified from Du et al. (2015).
Spongillida gen. et sp. indet. sensu Cayeux 1929
Fig. 9.
Horizon: Stephanian, upper Carboniferous.
Locality: Freshwater deposits at Montgros, Gard of the Massif Central, France.
References: Cayeux 1929; Geyer 1962; Zimmerle 1991; Schindler et al. 2008.
Description (emended from Cayeux 1929: 300).—Spiculite with monaxial spicules (200–250 × 10–20 µm) typically oriented in all directions, or grouped in bunches showing a perfect parallelism with thoroughly matted spicules aligned parallel to bedding. The spicules show traces of the central axial canal.
Remarks.—The sample is reported simply as freshwater sponges spicules. We could only judge the French material from the published evidence. Schindler et al. (2008: fig. 2) show a reproduction of the two original thin sections (see also Cayeux 1929: pl. 17: 64–65). Attribution to freshwater sponges is based on geological context i.e., occurrence among coal deposits far from any marine basin/deposits. Associated sediment consists of quartz grains, phyllite clasts, rare pyrite, and an additional clay mineral. There are spots of organic matter cautiously described (Cayeux 1929: 301) as an unknown form of sapropelitic coal.
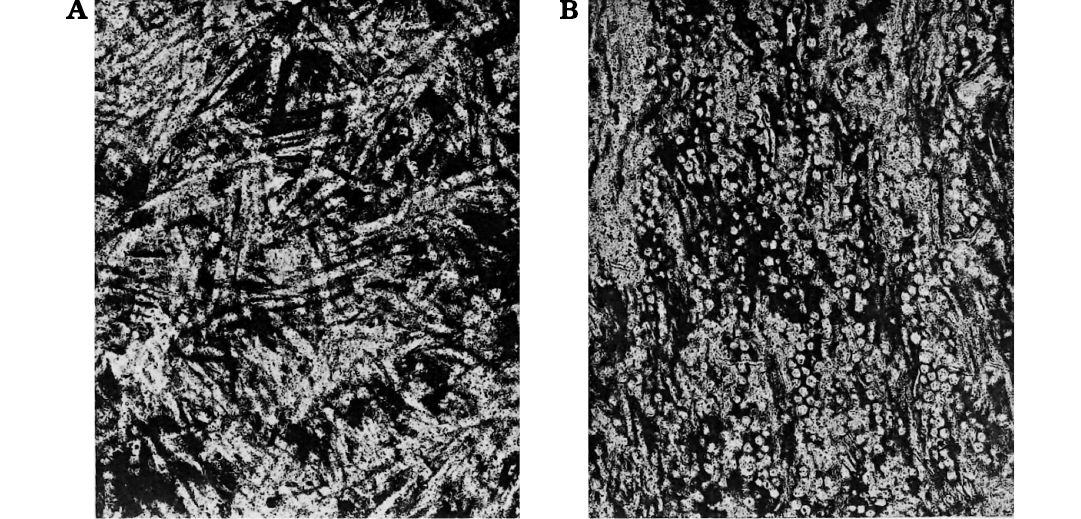
Fig. 9. Spongillida gen. et sp. indet, sensu Cayeux (1929). Stephanian, upper Carboniferous; Montgros, Gard of the Massif Central, France. Freshwater spiculites where spicules (200–250 µm long) are horizontally arranged (A) and cross cut (B). Modified from Cayeux (1929: pl. XVII).
Spongillida gen. et sp. indet. sensu Schindler et al. 2008
Fig. 10.
Horizon: Stefanian–Autunian, Permo-Carboniferous.
Locality: Donnersberg, Friedelhausen, Lemberg, Saar-Nahe Basin, Germany.
References: Schindler et al. 2008.
Description (emended from Schindler et al. 2008).—Megascleres. In Lemberg, oxeas (379–1250 µm in length) smooth, slightly curved, sharp at both tips and with traces of the central axial canal. The oxeas length/diameter ratio is between 8.4 and 12.5. In Donnersberg, oxeas (300–900 µm in length) smooth, some slightly curved. Traces of a threadlike and thin central axial canal only occasionally visible. The length/diameter ratio is between 10.4 and 26.7. In Friedelhausen, the only proven megasclere is a translucent smooth oxea just under 1 mm in length. Microscleres, gemmuloscleres and gemmules not detected.
Remarks.—The material is reported simply as freshwater siliceous sponges from three localities. The spicules from Donnersberg and Lemberg seem to be identical. In comparison to extant Spongillida species the dimensional range of spicules is unusually wide, indicating the possibility that more than a single species might have been involved. There are no doubts about sponge nature of the spicules, and its freshwater occurrence is based on geological context.
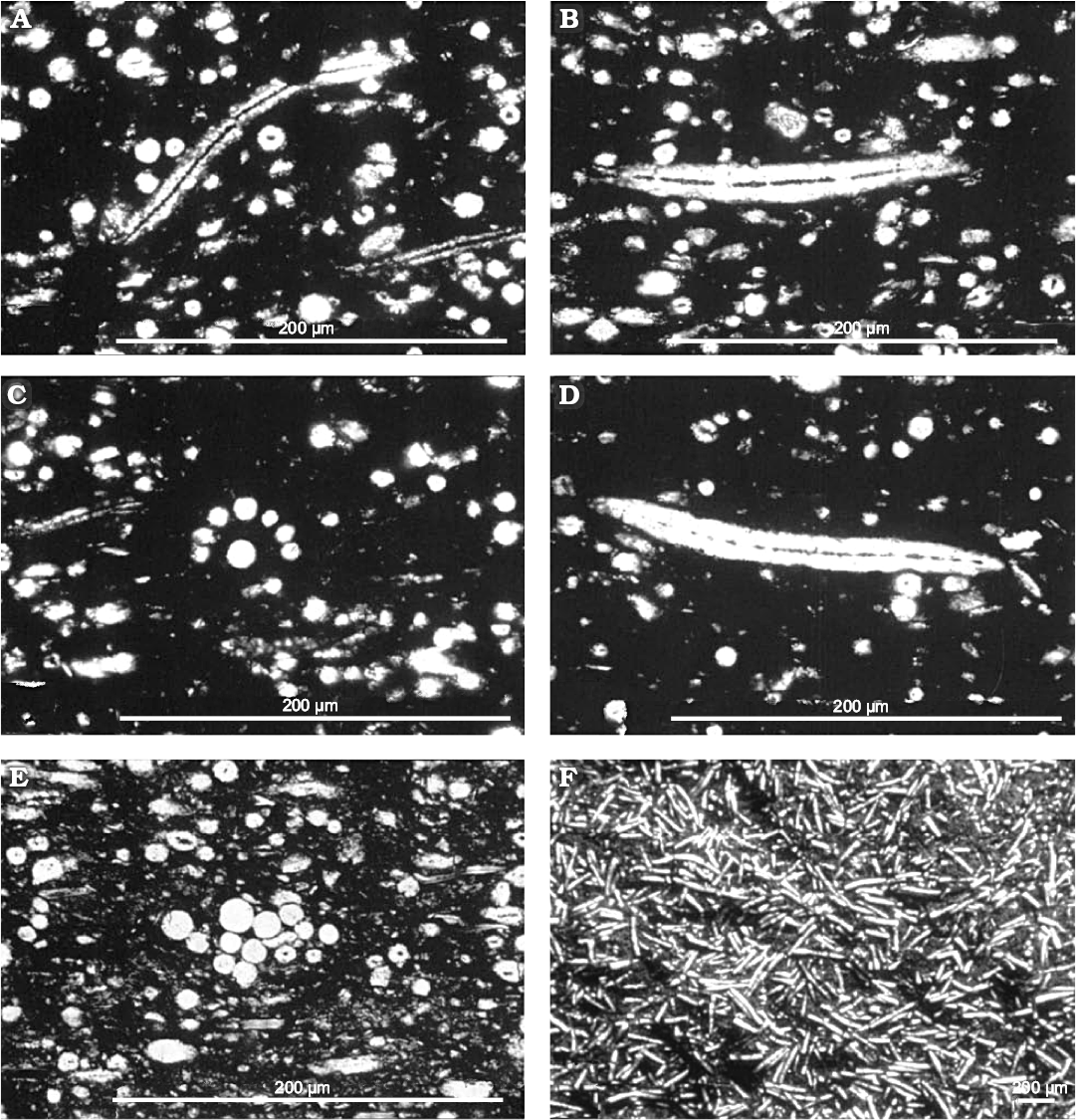
Fig. 10. Spongillida gen. et sp. indet. sensu Schindler et al. 2008. Stefanian-Autunian, Permo-Carboniferous, Donnersberg, Friedelhausen, Saar-Nahe Basin, Germany. Thin section of spiculite from the site Lemberg/Saar-Nahe Basin, layer 4 (PWL2004/5035a-LS). A. Longitudinal and cross sections of slim oxeas with axial canal. B. Thick oxea with axial canal. C. Concentric spicule bundle. D. Slim oxea with axial canal. E. Radially arranged spicule bundle. F. Layer surface (find from stockpile) etched with hydrofluoric acid (PWL2004/5039-LS). Modified from Schindler et al. (2008).
Spongillidae spp. sensu de Laubenfels 1955
Horizon: Jurassic.
Locality: Unknown.
References: De Laubenfels 1955, Schindler et al. 2008.
Description.—No description and figures available.
Remarks.—The material is reported simply as freshwater siliceous sponges. Schindler et al. (2008: 1) in their introductive chapter, refer that “the oldest freshwater siliceous sponges are known from Mesozoic deposits (Jurassic, no details of location and taxon)”, indicating de Laubenfels (1955) as reference. This latter paper (de Laubenfels 1955: E34) refers to freshwater sponges indicating that 3 genera of fossil freshwater sponges are known from the Jurassic to Recent. More details are supplied at page E37: “Spongilla (Jurassic to Recent); Meyenia (Pleistocene to Recent), and Heteromeyenia (Pleistocene to Recent)”. Jurassic occurrence refers most probably to Hinde (1883).
Spongillidae Gray, 1867
Eospongilla morrisonensis Dunagan, 1999
Fig. 11.
Type horizon: Morrison Formation, Upper Jurassic.
Type locality: Fort Collins (Highway 287 roadcut, 40°40.432’N, 105°11.319’W), Colorado, USA.
References: Waldschmidt and LeRoy 1944: 1097; Yen 1952: 31; Dunagan 1999.
Description (emended from Dunagan 1999).—Spiculite with two massive accumulation of spicules, small (10–15 mm), low domical to cylindrical. Megascleres dominant strongyles and oxeas (180–300 × 20–35 µm) with smooth surface, spineless. Microscleres, gemmuloscleres and gemmules not detected.
Remarks.—The most important evidence, stressed by Dunagan (1999), is the very rare discovery of two entire fossil specimens of freshwater sponges. Unfortunately, as it can be judged from published photos, it is clear that we deal not with specimens of sponges with original spicules arrangements, but rather artificial accumulation of spicules (spiculite) most probably within a burrow. Dunagan (1999) highlights in the discussion the morphological differences of Eospongilla morrisonensis vs. Palaeospongilla chubutensis considering in the latter species the presence of microscleres and prevailing oxeas (megascleres) as morphotraits supporting the erection of a new taxon. No other extant or fossil taxa were taken into account. The erection of the new taxon is based on the supposed absence of microscleres (thus, a negative character), in opposition to the case of P. chubutensis, but first of all the preservation of the material is very different in the two cases. In the case of Palaeospongilla the rock is silicified and spicules are preserved in situ, while E. morrisonensis is embedded in carbonate matrix that facilitates dissolution of silica spicules (also the megascleres are substituted with calcium carbonate). Thus the absence of microscleres may be just a taphonomic effect, for example, winnowing of smaller spicules. Their absence cannot be used as taxonomic character justifying erection of a new taxon. It is not really possible to conclude whether microscleres and gemmules were really absent or present in this sponge. Indeed, gemmules may be absent both due to taphonomical effect, as well as the fact that sponges were fossilized (if we accept interpretation of the material as real sponges, not artificial aggregation of spicules) in a period in which gemmulating sponges are devoid of gemmules (vegetative phase). Thus any taxonomical conclusion based on presented material must be limited to the statement that we deal with freshwater sponges, without more precise attribution, and the taxon E. morrisonensis should be considered as nomen nudum.
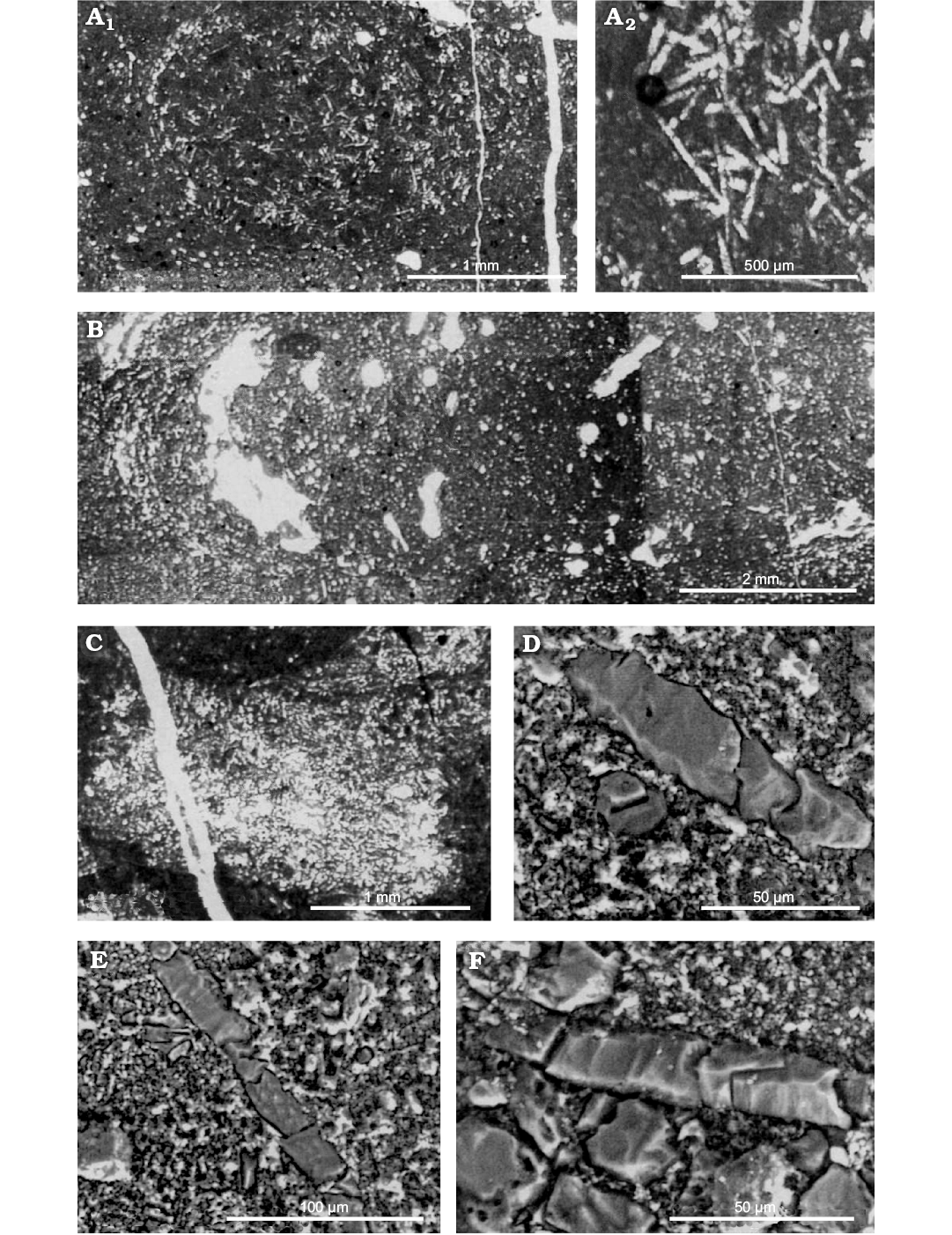
Fig. 11. Eospongilla morrisonensis Dunagan, 1999, Spongillida: Spongillidae; Morrison Formation, Upper Jurassic, Fort Collins, Colorado, USA. A. Holotype USNM 496326; A2, enlargement of the central area of A1 showing straight oxeas and slightly curved strongyles. B–D. Paratype USNM 496327. B. Longitudinal section showing arcuate distribution of spicules around the perimeter. C. Spicule bundle (large clear area). D. Longitudinal and transverse sections of oxeas. E. Longitudinal section of strongyle. F. Longitudinal section of oxeas (note recrystallized texture of spicules). Modified from (Dunagan 1999).
Spongillidae Gray, 1867
Spongilla purbeckensis Young, 1878
Fig. 12.
Type horizon: Purbeck Limestone, Lower Cretaceous.
Type locality: Stare Cove, Dorset, England.
Refrences: Young 1878; Hinde 1883; Waldschmidt and Leroy 1944; Greensmith 1978; Clements 1967.
Description (emended from Young 1878 and Hinde 1883).—Oxeas (megascleres) abundant, fusiform, acerate, slightly curved, thickest in the centre (ca. 450 × 15–25.5 µm, Hinde 1883) and gradually diminishing to the tips. On the contrary, oxeas are minutely tubercled (Young 1878).
Remarks.—Clements (1967) considered that the supposed sponge spicules of this species are not fossils at all but are actually silica pseudomorphs after lenticular crystals of gypsum. So, this species should be considered with caution. Attribution of this material to any Recent genera seems precarious due to its age. On the other hand, if we accept its sponge origin, the very few diagnostic characters (megascleres of similar type occur in several genera and families) allow only the attribution to the order Spongillida.
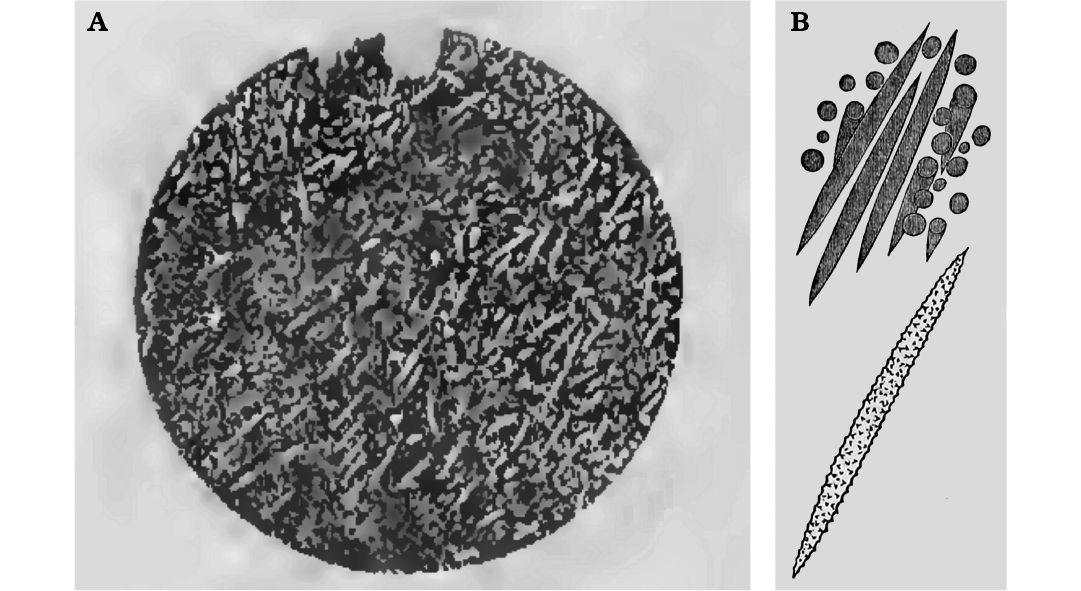
Fig. 12. Spongilla purbeckensis Young, 1878, Spongillida: Spongillidae; Purbeck Limestone, Lower Cretaceous, Stare Cove, Dorset, England. A. Spicules in the spiculite as illustrated by Hinde (1883). B. Spiny oxeas in the drawing of Young (1878). Originally not to scale; it may be inferred from the text that spicules are 450 μm long.
Palaeospongillidae Ott and Volkheimer, 1972
Palaeospongilla chubutensis Ott and Volkheimer, 1972
Fig. 13.
Type horizon: Aptian, Lower Cretaceous.
Type locality: 16 km NNW of Cerro Condor, Patagonia Chubut River Valley, Argentina.
References: Ott and Volkheimer 1972; Racek and Harrison 1975; Volkmer-Ribeiro and Reitner 1991; Manconi and Pronzato 2002.
Description (emended from Ott and Volkheimer 1972; Racek and Harrison 1975; Volkmer-Ribeiro and Reitner 1991; Manconi and Pronzato 2002).—Body shape encrusting-lobose, 2–6 mm thick. Surface smooth even though not well discernible, owing to the calcareous algal cover. Ectosomal skeleton undetected. Choanosomal skeleton a regular, anisotropic meshwork with multispicular (up to 40) ascending primary fibres which, with perpendicular secondary fibres, form a reticulum of square meshes (mesh diameter, 1 mm). Abundant spongin. Megascleres oxeas, straight to feebly curved, of two dimensional classes from stout (230–540 × 10–22 µm) cylindrical abruptly pointed and smooth, rarely bearing 1–2 spines, to abundant slender, fusiform, sharply pointed, finely spiny except at their tips (180–308 × 4–14 µm). Stout oxeas form the main skeleton of central and basal sponge body whereas slender oxeas are more frequent in peripheral skeletal fibres. Microscleres (more abundant above the layer of gemmules), oxeas abruptly pointed, straight to feebly curved, from incipiently to conspicuously spiny (179–190 × 3–4 µm) to smooth (77–147 × 3–10 µm). Gemmuloscleres acanthostrongyles to acanthoxeas dependent on the position of spines at the tips (70–168 × 3.5–8 µm) slender, slightly curved. Spines, scattered along the entire surface, more grouped and recurved at the tips, often with a single spine as prolongation of the axis. Gemmules subspherical (483–644 µm in diameter) scattered in the skeleton, singly or grouped, belonging to two types i.e., armed and unarmed. Unarmed gemmules, that is, without gemmuloscleres, enveloped in a cage of tangential spiny megascleres; gemmular theca bilayered with thin pneumatic layer and conspicuous inner layer, foramen simple without a porous tube. Armed gemmules with gemmuloscleres in one layer, variably arranged and crossing each other at various angles; thick trilayered theca, outer layer ill defined, well developed inner (20 µm in thickness) and pneumatic (100 µm in thickness) layers, nipple-like foramen.
Remarks.—Palaeospongilla chubutensis was described and redescribed in three subsequent papers (Ott and Volkheimer 1972; Racek and Harrison 1975; Volkmer-Ribeiro and Reitner 1991). Finally, the previous descriptions of P. chubutensis were merged by Manconi and Pronzato (2002). P. chubutensis is represented by a complete and intact skeletal arrangement of megascleres and microscleres, both of two types, as well numerous gemmules with (and without) gemmuloscleres in situ. In the original description (Ott and Volkheimer 1972) the new species is described as a new genus Palaeospongilla of the family Spongillidae. Racek and Harrison (1975) re-examined the type material and described a single type of gemmule with more or less radially arranged gemmuloscleres suggesting an affinity with the genus Radiospongilla. Volkmer-Ribeiro and Reitner (1991) changed significantly the palaeontological interpretation and re-description of P. chubutensis (partim) describing the new species Spongilla patagonica (see below) and erecting the new family Palaeospongillidae, attributing unarmed gemmules to P. chubutensis and the armed ones to the new species. The decision by Manconi and Pronzato (2002) to declare S. patagonica a junior synonym of P. chubutensis is based on the fact that the production of two gemmular morphs in the same specimen was also known in two genera of Spongillidae i.e., Spongilla and Corvospongilla, both characterised by unarmed and armed gemmules with or without pneumatic layer. Thus the erection of the family Palaeospongillidae, based on the absence of gemmuloscleres, is enigmatic. Further studies are needed.
Spongillidae Gray, 1867
Spongilla patagonica Volkmer-Ribeiro and Reitner, 1991
Fig. 13.
Type horizon: Aptian, Lower Cretaceous.
Type locality: 16 km NNW of Cerro Condor, Patagonia, Chubut River Valley, Argentina.
References: Volkmer-Ribeiro and Reitner 1991; Manconi and Pronzato 2002.
Description (emended from Volkmer-Ribeiro and Reitner 1991).—Megascleres oxeas (210–260 × 7–10 µm), slim, smooth, gradually sharply pointed, slightly curved. Microscleres oxeas (100–110 × 9–10 µm) spiny, usually straight to slightly curved. Gemmuloscleres strongyles (70–80 × 8 µm) strongly spiny, short, cylindrical, slightly to strongly bent, rarely straight with some larger and curved spines at the star-like tip. Gemmules subspherical (470 µm in diameter) with thick inner layer (20 µm) and thick pneumatic layer (100 µm). Gemmuloscleres randomly oriented and loosely packed in the gemmular theca, with tips protruding in places. Foramen single, nipple-like.
Remarks.—Spongilla patagonica was erected by Volkmer-Ribeiro and Reitner (1991), offering reasonable taphonomical explanation, within the framework of the re-description of P. chubutensis (partim). Subsequently, the species was considered invalid by Manconi and Pronzato (2002). The presumed spicular mixture and overgrowing of two sponge species, perfectly preserved and retaining even the eggs (or so interpreted), was based on previous descriptions on the presence of two gemmular morpha, i.e., unarmed and armed. Both skeleton spicules and the two gemmular types of S. patagonica were ascribed to P. chubutensis in the 1975 paper of Racek and Harrison. Moreover, Volkmer-Ribeiro and Reitner (1991) did not clearly discriminate between specimens and spicular complement descriptions of the two species. Thus this case could be solved with additional material and studies. In any case the complex P. chubutensis and S. patagonica shows that in the Early Cretaceous the freshwater sponge gemmule architecture was virtually identical to that of the more complex gemmules of Recent species, denoting a very early conservative evolutionary trend with no subsequent major functional changes.
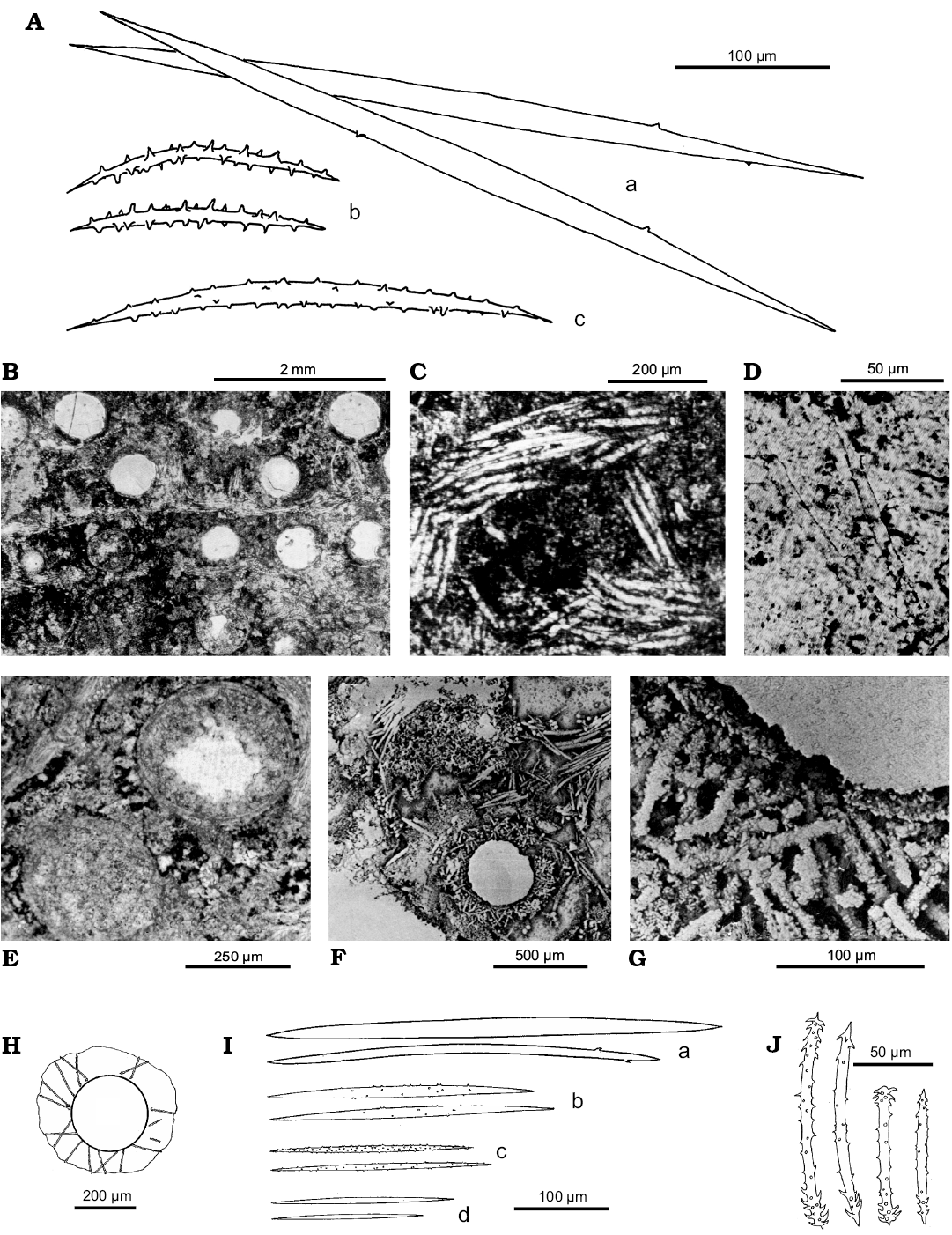
Fig. 13. Palaeospongilla chubutensis Ott and Volkheimer, 1972, Spongillida: Palaeospongillidae; and Spongilla patagonica Volkmer-Ribeiro and Reitner, 1991, Spongillida: Spongillidae; Aptian, Lower Cretaceous, 16 km NNW of Cerro Condor, Patagonia Chubut River Valley, Argentina. Spicules, skeleton architecture and gemmules, paratype MABA 24748. A. Drawings of megascleres (a), gemmuloscleres (b), and microscleres (c). B. Possible transversal section of several gemmules. C. Skeleton multispicular fibres or gemmular cage. D. Megascleres and microscleres. E. Naked gemmules. F. Armed gemmule with radial gemmuloscleres and multispicular main fibre. G. Close up on the gemmular theca. H. Gemmule. I. Megascleres (a, b), microscleres of different dimensional classes (c, d). J. Gemmuloscleres. A–D, modified from Ott and Wolkheimer (1972), E–G, modified from Volkmer-Ribeiro and Reitner (1991b), H–J, modified from Racek and Harrison (1975).
Spongillidae Gray, 1867
Lutetiospongilla heili Richter and Wuttke, 1999
Fig. 14.
Type horizon: Eocene.
Type locality: Messel oil shale, Germany.
References: Richter and Wuttke 1999; Richter and Baszio 2000, 2001, 2009; Richter et al. 2017.
Description (emended from Richter and Wuttke 1999; Richter and Baszio 2000, 2001, 2009).—Encrusting growth on submerged plants (stems, roots). Gemmules in carpets at the sponge’s basal portion directly on the substrate. Megascleres oxeas, slender (300–350 × 8–12 µm), straight to slightly curved, with entire surface covered by small spines. Microscleres not surely identifiable. Gemmules (800–1000 µm in diameter) with evident lumen (300–400 µm in diameter) and gemmular theca (200–250 µm in thickness) with radially arranged gemmuloscleres surrounded by a dense spicular cover of tangential megascleres irregularly arranged. Gemmuloscleres of two types. Larger gemmuloscleres shaped from strongyles to strongyloxeas almost straight to curved, coarsely spiny with spines more densely arranged at the tips, and extremely variable in size and shape (80–220 × 12–20 µm, shaft thickness at the middle); tips usually slightly inflated with tubercles/spines. Smaller gemmuloscleres birotules short, straight, slender, squat or with very variable shape (40–60 × 5–10 µm, shaft thickness at the thinnest point) with weak spines. Spicule tips inflated with strong spines, frequently with an apical spine, quite often as imperfect discs.
Remarks.—According to the authors, the small gemmuloscleres of Lutetiospongilla heili are similar to those of the genus Radiospongilla, although in the former they generally have a significantly slimmer shaft and spiny tips from inflated to imperfect rotules. The large gemmuloscleres, strongyles, despite considerable differences in length, are strictly radially arranged and densely packed in the gemmular theca. In addition to this “normal” gemmular morph other gemmules occasionally apparently contain significantly smaller gemmuloscleres arranged in multiple layers. Much more difficult to recognize is the original arrangement of small gemmuloscleres because they are always internal and, usually, relatively disorganized. Broken gemmules always show more or less radial gemmuloscleres.
Richter and Baszio (2000, 2001, 2009) presented new images of L. heili. The spicular complement shown in these more recent papers seems to not match those reported in the original description. These authors compare L. heili to numerous extant genera in which the gemmuloscleres are also arranged radially in the gemmular theca (i.e., Radiospongilla, Heteromeyenia, Corvospongilla, Anheteromeyenia, Corvomeyenia, Dosilia, and Acalle) concluding that Radiospongilla is the most similar to Lutetiospongilla. The taxon could be a mix of species and further studies are needed.
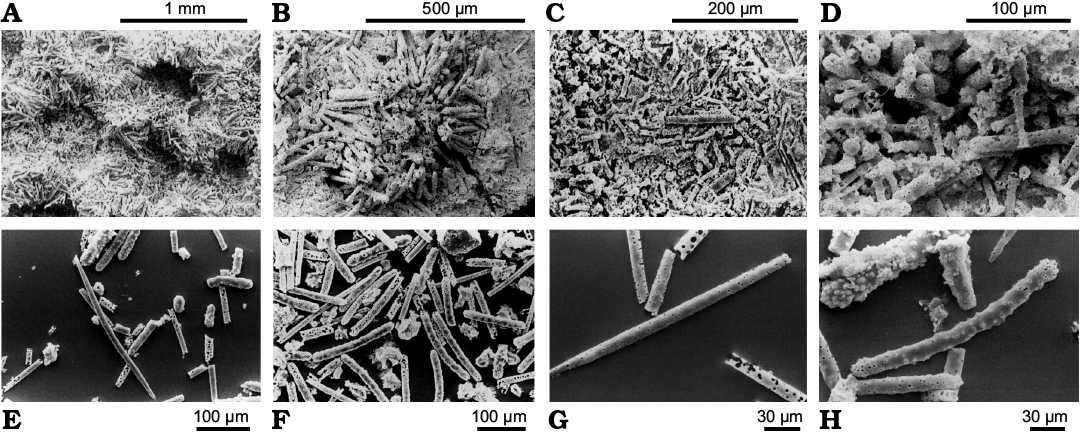
Fig. 14. Lutetiospongilla heili Richter and Wuttke, 1999, Spongillida: Spongillidae; Eocene, Messel oil shale, Germany. A. Holotype, SMF ME I 5143, carpet of closely bound gemmules with evident lumina and large radial gemmuloscleres. B. Single gemmular theca, firmly sessile on the surface of a woody plant, oblique view. Gemmular lumen (top right). C. Broken gemmule in the sediment with gemmular theca mainly formed of small more or less radially arranged gemmuloscleres. D. Broken-up lumen of a gemmule. Numerous mixed inner ends of large and small gemmuloscleres. E. Megasclere and large gemmuloscleres. F. Scattered sample with numerous huge gemmuloscleres. G. Megascleres originally finely spiny. Here, the pores are further apart and are narrower than in H. H. Gemmuloscleres from Messel with corrosion canals of original dense spines. SEM images. Modified from: A–D, Richter and Wuttke (1999); E, F, Richter and Baszio (2009); G, H, Richter and Baszio (2000).
Spongillidae Gray, 1867
Ephydatia kaiseri Rauff, 1926
Fig. 15.
Type horizon: (Pre) middle Eocene freshwater chert.
Type locality: Sinkhole south-west from fixed point 533, Pomona diamond field, Namib Desert, Namibia.
References: Rauff 1926; Arndt 1936; Manconi 2008.
Description (emended from Rauff 1926).—Spiculite of loose spicules with entire gemmules. Megascleres (many broken) oxeas of two dimensional classes, smooth, slightly bent, pointed, sometime with evident axial canal. Large oxeas (350 × 7–17 µm) maybe longer but broken. Short oxeas (up to 20 × 3.5 µm). The shorter and very thin can be young or larval spicules. Gemmules entire, with gemmuloscleres in natural arrangement. Gemmuloscleres birotules (44–65 µm in length) with smooth rotules (18–20 µm in diameter), with entire (not incised) margins, flat except for umbone, and smooth shaft (5–6 µm in thickness).
Remarks.—Ephydatia kaiseri is the only known fossil freshwater sponge from Africa. Rauff (1926) reports loose oxeas and gemmules with birotules in natural arrangement as confirmed by Arndt (1936: 17). The original paper reports that the deposit mainly consists of sponge spicules; in about 1–0.5 mm2 there are a minimum 70 of relicts. The spicules consist of chalcedony, however, some rare spicules do not show a double refraction. The variable length of birotules matches the dimensional spectrum of known species of extant Spongillidae belonging to the genus Ephydatia. The identification at the species level is confirmed by Manconi (2008). The Recent species Ephydatia fluviatilis var. capensis Kirkpatrick, 1907 reported from the Cape region (Valkenberg Vlei, South Africa) is in part similar to E. kaiseri sharing with it some birotules with smooth rotules, flat except for umbone, with margins not incised, and smooth shaft (see Manconi and Pronzato 2009: 133, figs. 163–165).
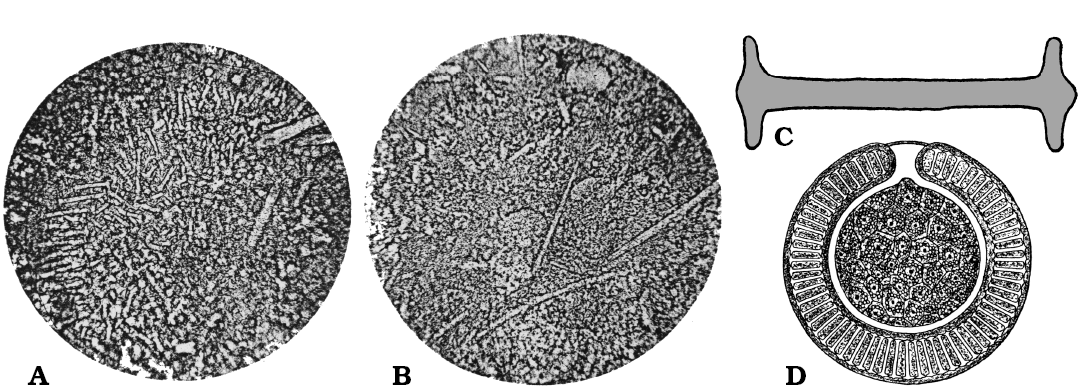
Fig. 15. Ephydatia kaiseri Rauff, 1926, Spongillida: Spongillidae; (pre) middle Eocene freshwater chert, Pomona diamond field, Namib Desert, Namibia. A. Grouped gemmuloscleres; originally enlarged 660×, the text states that particular birotules are 44–65 µm long. B. Megascleres; originally enlarged 175, the text states that spicules are 350 µm long. C. Birotule (gemmulosclere), originally enlarged 660×, the text states that spicule can be 44–65 µm long. D. Gemmule of a Recent species of Ephydatia (cross section). A, B, photographs; C, D, schematic drawings. Modified from Rauff (1926).
Spongillidae Gray, 1867
Ephydatia gutenbergiana (Müller, Zahn and Maidhof, 1982)
Fig. 16.
Type horizon: Middle Eocene.
Type locality: Messel, Germany.
References: Müller et al. 1982; Richter and Wuttke 1995; Richter and Baszio 2000, 2001, 2009; Richter et al. 2017.
Description (emended from Müller et al. 1982).—Megascleres oxeas of two dimensional classes (140–230 × 15–20 µm; 200–350 × 10–20 µm). Gemmuloscleres similar smaller oxeas (50–150 µm in length). Gemmules (structure interpreted as) with an extremely large diameter > 2 mm.
Redescription (emended from Richter and Wuttke 1995, 1999).—Unordered masses of gemmuloscleres heavily corroded and partially bounded together, and surrounded by secondary precipitation of dissolved silica. Absence of most of the rotules due to quite advanced corrosion processes. Spines as corrosion pits in the few spicules with rotules still recognizable.
Remarks.—According to the type material re-examination, the evaluation by Richter and Wuttke (1995, 1999) of the studied spicular complement is quite different from Müller et al. (1982). Gemmuloscleres found by the former authors in the holotype are birotules, whereas those by the latter are short spiny oxeas typical of Spongilla. Even the size of gemmules described by Müller et al. (1982) suggests that they cannot be considered true gemmules, as the largest known Spongillida gemmular diameter is only slightly larger than 1 mm. The new gemmuloscleres, found in the spiculite by Richter and Wuttke (1995, 1999) and Richter and Baszio (2009) are interpreted as birotules radially arranged in the gemmular theca of a species belonging to the genus Ephydatia. Indeed gemmuloscleres are spiny oxeas in Spongilla and birotules in Ephydatia. The new combination proposed by Richter and Wuttke (1995) shifting the species S. gutenbergiana to the genus Ephydatia is illustrated by quite convincing SEM images. The taxon could be a mix of species and further studies are needed.

Fig. 16. Ephydatia gutenbergiana (Müller, Zahn, and Maidhof, 1982), Spongillida: Spongillidae; middle Eocene, Messel, Germany. A. Five oxeas as represented in the original description. B. Entire gemmule. C. Skeletal network (line-like arranged megascleres). D. Megascleres. E. Gemmuloscleres, the transitional forms of the series a (bottom, right) could point out an origin of the birotules from oxeas with shortened shaft. F. Corroded gemmuloscleres. G, H. Two illustrations of gemmuloscleres that has been shifted from the genus Spongilla to the genus Ephydatia; insert in H shows gemmulosclere in phase contrast. I. Megasclere, original material with corrosion marks that are characteristic for Messel. Modified from: A, Müller et al. (1982); B, F, Richter and Wuttke (1999); G, H, Richter and Wuttke (1995); I, Richter and Baszio (2009).
Potamolepidae Brien, 1967
Potamophloios canadensis Pisera, Siver, and Wolfe, 2013
Fig. 17.
Type horizon: Lutetian, middle Eocene.
Type locality: Giraffe Kimberlite maar, Northern Canada (64°44’ N, 109°45’ W).
References: Pisera et al. 2013.
Description (emended from Pisera et al. 2013).—Megascleres. Strongyles with highly variable size (7.90–30.9 × 3.12–5.0 µm), smooth, thick, often curved, or otherwise slightly irregular, frequently with inflated tips. Microscleres (presumed) smooth strongyles. Gemmuloscleres not detected. No whole intact gemmules.
Remarks.—Pisera et al. (2013) report that a possible attribution to the genus Corvospongilla must to be excluded because of the lack of typical microscleres and gemmuloscleres belonging to this genus in all examined samples. Potamophloios canadensis is the oldest record belonging to the family Potamolepidae, of which the present geographic range is quite exclusive from Africa and South America with spot-like enclaves in the southern Nearctic Region, Thailand, and Oceanic Islands, i.e., Fiji and New Caledonia. Its presence in North Canada is consistent with the recent discovery of a living new potamolepid genus Cherokeesia Copeland, Pronzato, and Manconi, 2015 and species (Cherokeesia armata) from the Tennessee, and the fact that during the Eocene the climate of northern Canada and Nearctic Region was much warmer than today, as indicated by the presence of the redwood tree Metasequoia (Siver and Wolfe 2009).
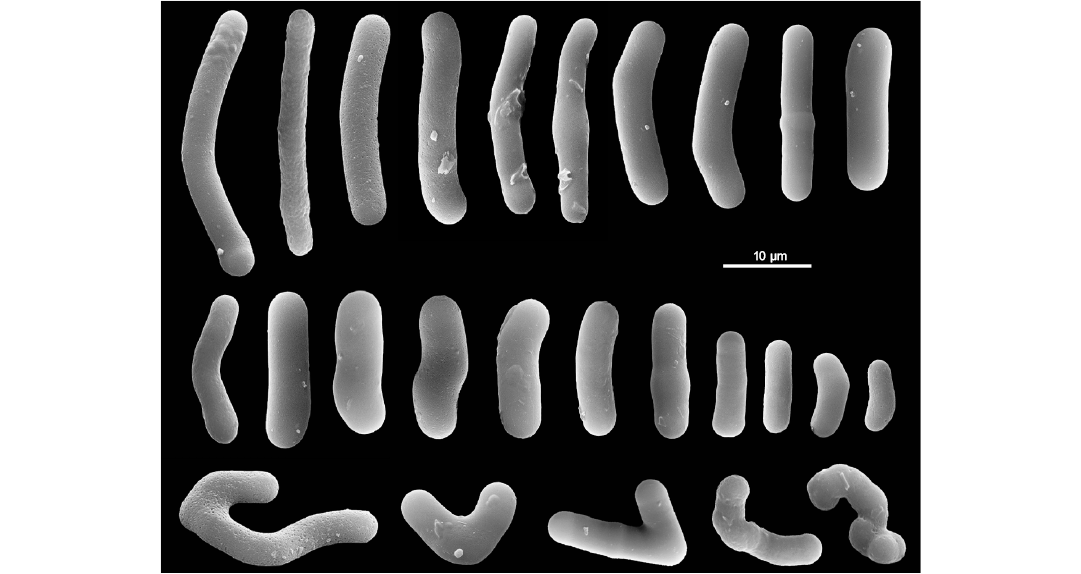
Fig. 17. Potamophloios canadensis Pisera, Siver, and Wolfe, 2013, Spongillida: Potamolepidae, ZPAL Pf23; Lutetian, middle Eocene, Giraffe Kimberlite maar, Northern Canada. Strongyles (gemmuloscleres) in a wide dimensional range. Malformed spicules (bottom row). SEM images. Modified from Pisera et al. (2013).
Spongillidae Gray, 1867
Ephydatia cf. facunda Weltner, 1895
Fig. 18.
Horizon: Lutetian, middle Eocene.
Locality: Giraffe Kimberlite maar, Northern Canada (64°44’ N, 109°45’ W).
References: Pisera et al. 2016.
Description (emended from Pisera et al. 2016).—Only disassociated spicules. Possible megascleres large oxeas (162.3–307 × 8.7–12.8 µm, usually 220–250 × 9–10 µm), straight to slightly curved, microspinose, with scattered small spines or tubercles, and the tips tapering to a sharp point. Some centrotylote forms present. Gemmuloscleres birotules with incised margins of both rotules and spiny shaft; spines (4–20, usually 6–12) scattered, large (extending in length to the rotule margin) and smooth with acute tips; rotules moderately smooth to strongly incised; at their center often rises a small rounded process (umbonate rotule).
Gemmuloscleres size highly variable (26–57 µm in length, mean 41.5 µm; shaft (not including spines) 5–11 µm in thickness mean 7.2 µm; rotulae diameter 15–28 µm mean 21.4 µm. Aberrant gemmuloscleres also present.
Remarks.—This species was recently very well described but for the sake of completeness this description is repeated here. Gemmuloscleres of E. cf. facunda are virtually identical, including their variability as in the extant species E. facunda, due to large time gap separating both forms, it was described as E. cf. facunda.
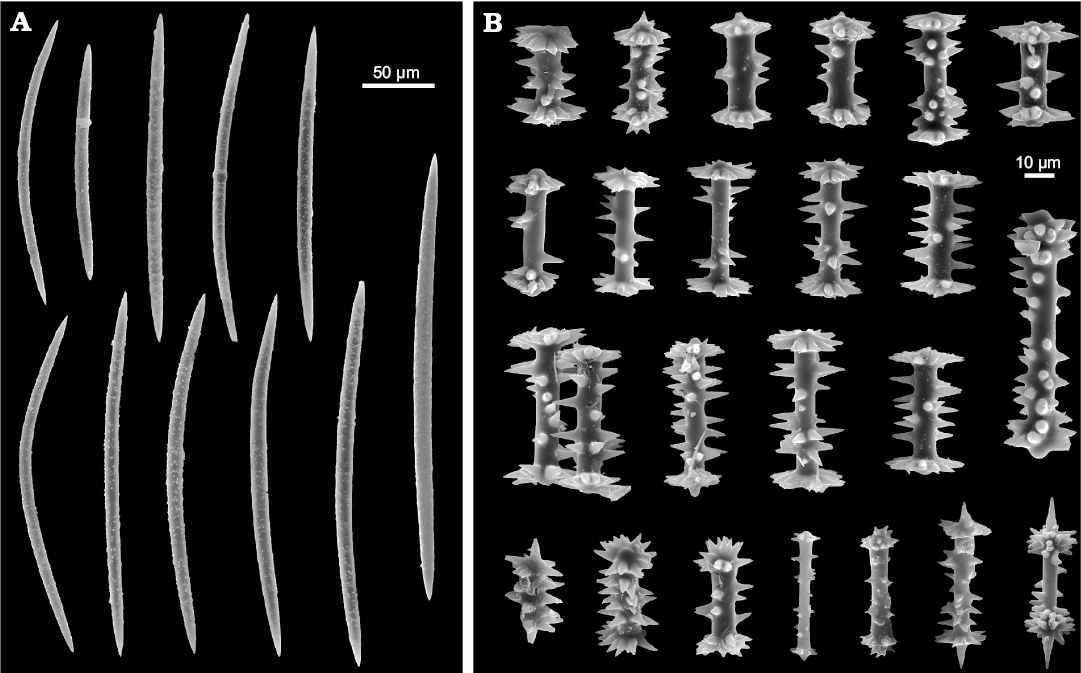
Fig. 18. Ephydatia cf. facunda Weltner, 1895, Spongillida: Spongillidae, ZPAL Pf23; Lutetian, middle Eocene, Giraffe Kimberlite maar, Northern Canada. A. Oxeas (megascleres). B. Birotules (gemmuloscleres). SEM images. Modified from Pisera et al. (2016).
Potamolepidae Brien, 1967
Oncosclera kaniensis Matsuoka and Masuda, 2000
Fig. 19.
Type horizon: Nakamura Formation, early Miocene.
Type locality: River bed on the Kiso River, Dota, Gifu Prefecture, Central Japan.
References: Matsuoka 1983; Matsuoka and Masuda 2000; Manconi et al. 2012.
Description (emended from Matsuoka and Masuda 2000).—Growth form encrusting (< 1 mm in thickness) on surfaces of bivalve shell and wood fragments. Surface even. Megascleres moderately small (100–179 × 7–15 µm) almost straight, from dominant, stout, cylindrical, spiny strongyles to less frequent spiny oxeas, covered with distinct spines at tips. Microscleres absent. Gemmules subspherical (ca. 500 µm in diameter) firmly adhering to basal portion of sponge body. Gemmuloscleres strongyles (23–100 × 4–7 µm) stout, variably curved with smooth concave area, inflated at middle, densely covered with distinct spines numerous at both tips; some aberrant polyfurcate spines.
Remarks.—Oncosclera kaniensis is assigned to the genus Oncosclera on the basis of its growth form and ornamentations of megasclere and gemmuloscleres. Authors note that this species is similar to some Recent species from Argentina, namely O. ponsi, O. atrata, and O. tonollii. The most similar is O. ponsi in spicular components, but it differs in having spiny oxeas as megascleres. O. kaniensis differs from O. atrata in having strongyles (gemmuloscleres) densely covered with distinct spines at both tips. It also differs from O. tonollii in having less spiny surface of gemmuloscleres and megascleres. O. kaniensis has gemmuloscleres similar to the Recent species O. jewelli and O. schubarti, but differs distinctly from the latter species in its spiny strongyles and oxeas as megascleres. Extant species belonging to the genus Oncosclera are reported from the whole tropical zone i.e., South America, Africa, South-East Asia, and Pacific Islands (see Manconi et al. 2012).
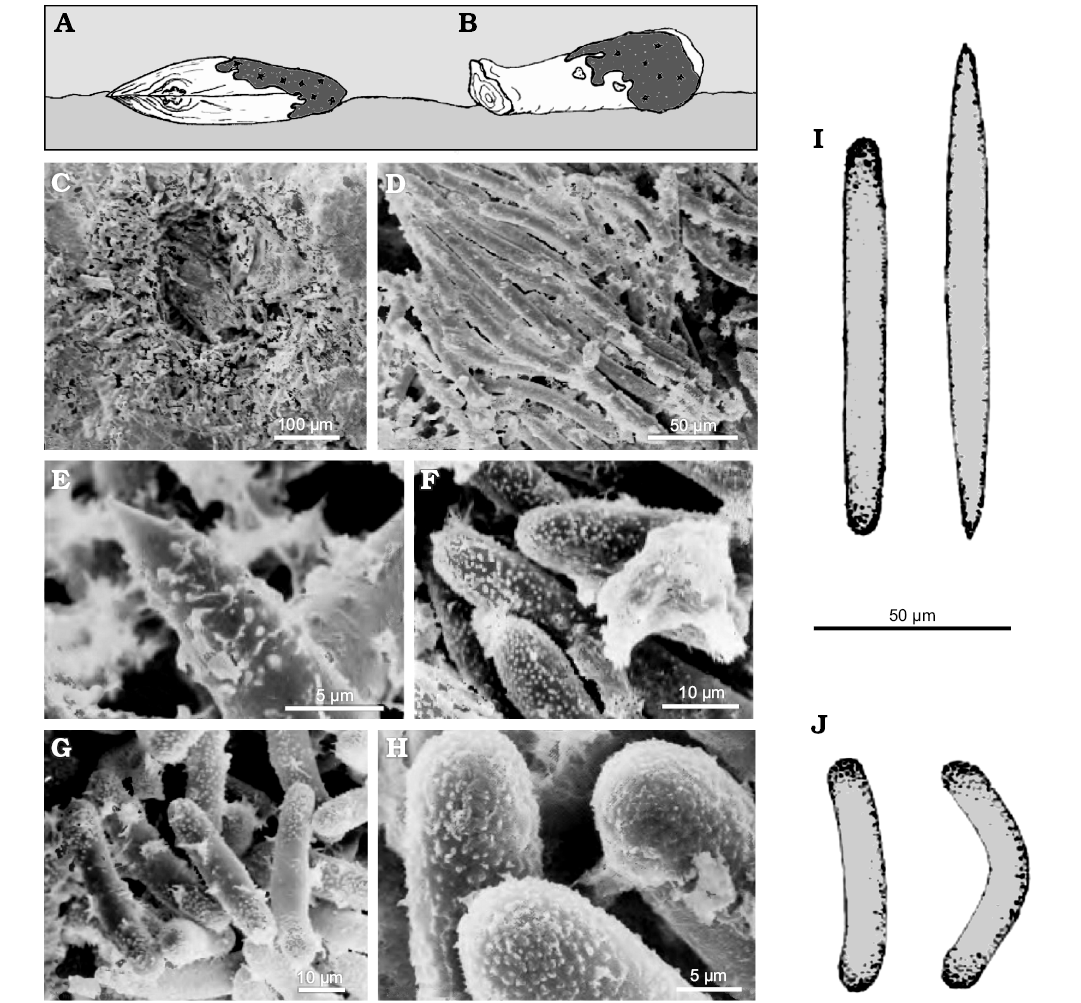
Fig. 19. Oncosclera kaniensis Matsuoka and Masuda, 2000, Spongillida: Potamolepidae, TMNH; Nakamura Formation, early Miocene, River bed on the Kiso River, Dota, Gifu Prefecture, Central Japan. A. Sponge encrusting a shell (Anodonta, Mollusca, Bivalvia). B. Sponge encrusting a wood fragment retaining its annual rings. C. Gemmule (cross section). D. Megascleres. E. Oxea tip (megasclere). F. Strongyles tips (megasclere). G. Strongyles (gemmuloscleres). H. Spiny tips of strongyles (gemmuloscleres). I. Strongyle and oxea (megascleres). J. Strongyles (gemmuloscleres). A, B, explanatory drawings; C–H, SEM images; I, J, drawings of the spicular complement. Modified from Matsuoka and Masuda (2000).
Spongillidae Gray, 1867
Ephydatia chileana Pisera and Sáez, 2003
Fig. 20.
Type horizon: Late Miocene.
Type locality: Quillagua Formation, Atacama region, Chile.
References: Pisera and Sáez 2003.
Description (emended from Pisera and Sáez 2003).—Three intact gemmules and many loose megascleres and gemmuloscleres preserved in a diatomite. No entire sponge bodies. Megascleres slightly curved, smooth oxeas (240–350 × 8–15 µm). Gemmules spherical to flattened (237–250 µm in diameter). Gemmuloscleres birotules (39–45 µm in length), with spiny shaft (5.5–7.2 µm in thickness) armed by a variable number (4–11) of strong, long conical spines of variable length. Rotules (usually 24.8–27.0 µm in diameter) with irregular margins, often deeply incised, and the resulting teeth (16 to 20–21 spines) not in the same plane vary greatly in length (ca. 11.0 µm at the longest); rotules upper surface, clear, flat, with a button-like central convexity (umbonate rotule). In one gemmule, many aberrant rotules bear a swelling in the middle of the shaft, along with rotules of normal morphology. Spines reduced to small tubercles on a bulbous-like termination of rotules and/or less numerous and irregularly developed.
Remarks.—The investigated material resembles the Recent cosmopolitan species Ephydatia fluviatilis but differs from it in having smaller diameters of gemmules, longer and thicker shafts of the gemmuloscleres, and larger diameter of rotules. E. chileana also has many long, conical spines on the gemmuloscleres’ shaft. The upper surface of the rotule is smooth in the investigated material, whereas in E. fluviatilis, it is often spiny to various degrees. Megascleres are of very similar size and smooth in E. chileana, whereas they are often microspinose in E. fluviatilis.

Fig. 20. Ephydatia chileana Pisera and Sáez, 2003, Spongillida: Spongillidae, ZPAL Pf12; late Miocene, Quillagua Formation, Atacama region, Chile. A. Gemmule (cross-section). B. Gemmular theca with radial gemmuloscleres bearing strong spines on the shaft (cross-section). C, D. Inner surface of the theca with proximal rotules of gemmuloscleres. E. Diatomite with loose oxeas. F. Drawings of loose megascleres from the sample with gemmules. A–E, SEM images. Modified from Pisera and Sáez (2003).
Spongillidae Gray, 1867
Ephydatia fossilis Traxler, 1894
Fig. 21.
Type horizon: Sarmatian, Miocene.
Type locality: lake sediments, limestone from Dubrovica deposits.
Other occurrences: Pliocene of Bory, Slovakia; lignite from Chambon, Central France; brackish deposit of Kevna Bremia, Romania.
References: Traxler 1894; Firtion 1944.
Description (emended from Traxler 1894).—Freshwater sponge spicules and freshwater diatoms. Megascleres oxeas very variable in size (160–350 × 5–20 μm), straight or slightly curved, gradually pointed, smooth or covered with small spines sometimes with a swelling in the middle; some skittle-like tips. Spicules short and thin, straight or curved, and sometimes with a swelling in the middle also present. Gemmuloscleres birotules stout, very variable in size and shape (41–69 µm in length, rotules 16–26 µm in diameter; shaft 12–16 × 6–8 µm). Rotules with indented margins (9–14 teeth), in places with central axial spine but, usually, with only a hemispherical protuberance (umbone). The different sizes and structures of the birotules could give the impression that they belong to different sponge species.
Description (emended from Firtion 1944).—The lignite from Chambon (France), includes freshwater sponge spicules in groups. No megascleres. Gemmuloscleres birotules (56–70 µm in length; ca. 2.5 times the diameter of the rotules) with shaft (5.6–7 µm in thickness), straight or weakly curved, sometime with a slight swelling. Rotules with indented margins, equal to sub-equal (22.3–25.5 µm in diameter). Shaft with numerous strong spines (6.1–7.6 µm in length), straight and perpendicular to axis. The diatomite of the brackish deposit from Kevna Bremia (Romania), includes spicules and particularly birotules stout, large straight or curved, with strong spines (64.36–69.02 µm in length, shaft 7.14–8.23 µm in thickness). Rotules (26.18–28.66 µm in diameter) equally developed and with incised margins.
Remarks.—Traxler (1894) reports that the spicular complement (oxeas and birotules) of this species (particularly birotules) is different from all freshwater sponges. Moreover fossil remains from the two deposits are very similar, although in the Bory sample birotules are more abundant. The most close living species to Ephydatia fossilis is E. robusta (Potts, 1888) previously Mejenia subdivisa. In the Firtion (1944) opinion, the spicules described by Traxler (1894) are comparable in all respects with the birotules of the Chambon lignite, and must be ascribed to E. fossilis.
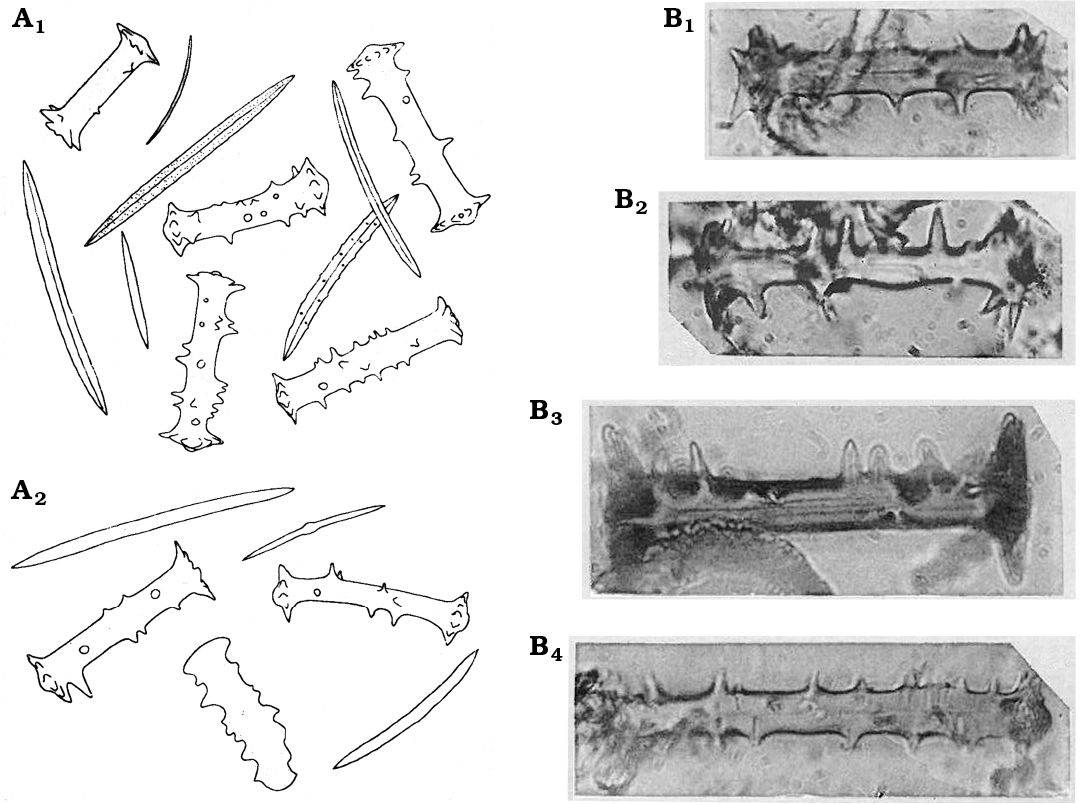
Fig. 21. Ephydatia fossilis Traxler, 1894, Spongillida: Spongillidae. A. Drawings of oxeas (megascleres) and birotules (gemmuloscleres). Originally not to scale; from the text it may be inferred that the birotules are between 41 and 67 µm long, and oxeas are 160 to 350 µm long; material from the Bory site (A1) and from the Dubrovica site (A2). B. Birotules with spiny shaft (gemmuloscleres); from Chambon, Central France. Birotules are 65–69 µm long. Modified from: A, Traxler (1894); B, Firtion (1944).
Spongillidae Gray, 1867
Spongillidae spp. sensu Williams 1985
Fig. 22.
Horizon: Miocene.
Locality: Lacustrine deposit at Oviatt Creek, northern Idaho, USA.
References: Williams 1985.
Description (emended from Williams 1985).—Spicular remains. Megascleres abundant, as smooth and spiny oxeas and strongyles (185–263 µm in length). Thickness not reported. Microscleres pseudobirotules (18–26 µm in length). Thickness of the shaft and pseudorotules diameter not reported. Gemmuloscleres of various types. Gemmuloscleres strongyles (40–105 µm in length) variably spiny. Thickness not reported. Gemmuloscleres birotules (24–34 µm in length). Thickness of the shaft and rotules diameter not reported.
Remarks.—A total of 28 spicules are described, illustrated and ascribed by Williams (1985) to the following genera: Anheteromeyenia, Corvospongilla, Eunapius, Radiospongilla, and Spongilla. In some cases an identification at species level is proposed (A. ryderi, E. fragilis, R. crateriformis, S. lacustris, and T. pennsylvanica) referring to Penney and Racek (1968) and to the help of A.A. Racek personally. The megascleres are not very useful for the identification of Spongillida; the majority (both living and fossil) bears monaxial megascleres, as smooth and/or spiny oxeas to strongyles. Also the spicule morphometries indicated by Williams (1985) are not diagnostic. Spicules identified as microscleres pseudobirotules are exclusive of the genera Corvoheteromeyenia, Corvomeyenia, and Corvospongilla. Monaxial gemmuloscleres are in agreement with the genera Eunapius, Radiospongilla, and Spongilla. Birotules could match the gemmuloscleres of the genus Anheteromeyenia, but some resemblance to the genera Ephydatia and Heteromeyenia also exists. The identification at the species level, and their identity with present taxa, seem hazardous considering rather poor material, but there is no doubt that several species/genera are present in this palaeofauna. The set of data from the Oviatt Creek, once again, evidences the high conservativeness of spicular morphotraits of freshwater sponges.
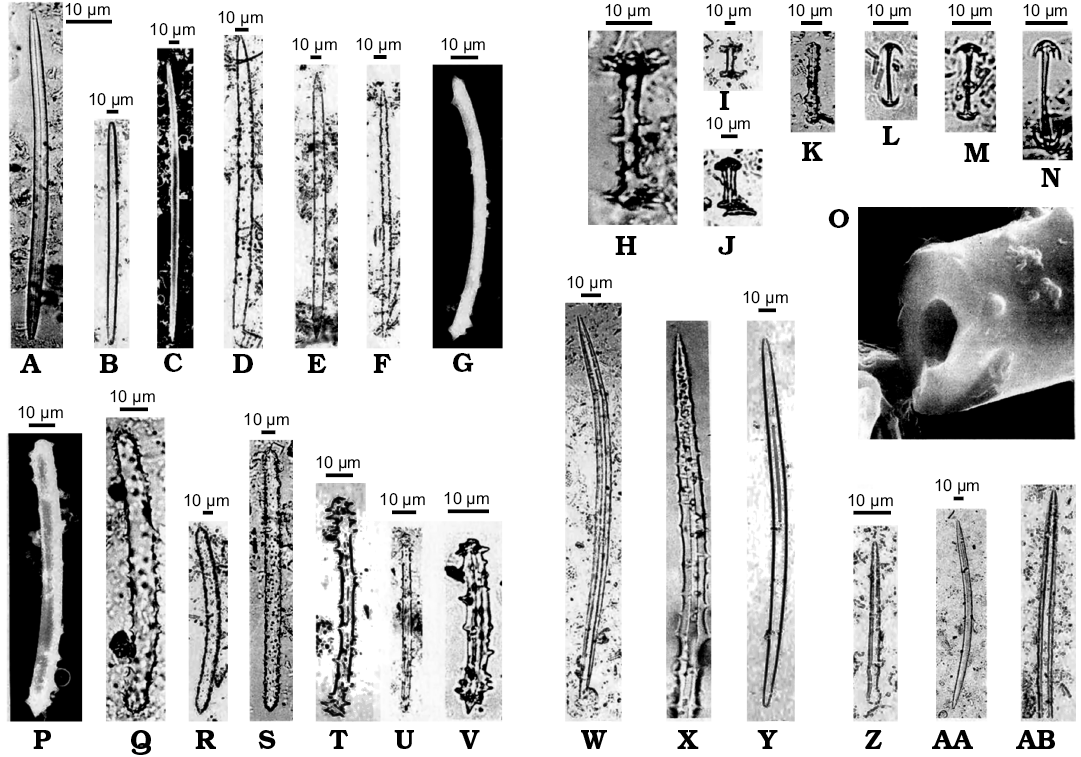
Fig. 22. Spicular remains of Spongillidae. Miocene, Lacustrine deposit at Oviatt Creek, northern Idaho, USA. Megascleres (A–F, S, W–AB), microscleres (L–N), and gemmuloscleres (G–K, P–V), ascribed originally to several genera: Anheteromeyenia, Corvospongilla, Eunapius, Radiospongilla, Spongilla, Trochospongilla; X, originally enlarged × 1200, AB, originally enlarged × 700. O. Axial canal of a broken spicule, originally enlarged × 5300. G, O, P, SEM images. Modified from Williams (1985).
Quaternary Spongillida
Records of subfossil Quaternary freshwater sponges that are mentioned in numerous papers (see Racek 1966; Harrison et al. 1979; Matsuoka 1983; Harrison and Warner 1986; Harrison 1988a, b, 1990; Frost 2001; Volkmer-Ribeiro et al. 2004; Volkmer-Ribeiro and Machado 2007; Parolin et al. 2008; Machado et al. 2012, 2014) and that raise no doubts about their taxonomic attribution are not considered in the present paper.
Discussion and conclusions
The critical review of fossil Spongillida we have performed on the basis of published data highlighted taxonomical incongruences among the different approaches recorded in the literature. Taxonomy of fossil Spongillida is based on taxonomy of extant forms.
There are few fossil records of Spongillida prior to the Quaternary. The oldest (Ediacaran) fossil putative freshwater sponge remains reported from China (Du et al. 2015) are not sponges, but probably testate amoebae, thus this record should be disregarded in discussion about freshwater sponge evolution.
The palaeontological data we have analyzed suggest that the most ancient freshwater sponge are Palaeozoic in age, precisely late Carboniferous (see Cayeux 1929; Schindler et al. 2008), but their freshwater setting is questioned by some authors (see Schultze 2009). Unfortunately the oldest occurrence of freshwater sponges from the Permo-Carboniferous of Europe (Schindler et al. 2008) can be only determined as freshwater sponge spicules, thus it remains unknown how the earliest known freshwater sponge spicular component of skeletal architecture was precisely developed, but it is obvious that those sponges had oxeas as megascleres and can be identified only as Spongillida. The same concerns Late Jurassic sponges from the USA reported by Dunagan (1999).
The Lower Cretaceous Spongilla purbeckensis (Hinde, 1883) from England is described solely on the basis of oxeas as megascleres, thus attribution only to the level of suborder Spongillida seems possible. On the contrary, the well preserved and described in detail Palaeospongilla chubutensis from South American Lower Cretaceous together with the Eocene Lutetiospongilla heili from Germany have affinities to Recent genera. The characteristic radial arrangement of spiny scleres in gemmules seems to be a conservative morphotrait in the adaptive radiation of the order Spongillida. The family Palaeospongillidae, erected on the absence of gemmuloscleres in the gemmular theca (only negative evidence) seems to be enigmatic because of the evidence that several species of Spongillidae, in different phases of their life cycle, produce also unarmed gemmules (Manconi and Desqueiroux-Faundez 1999).
The middle Eocene Ephydatia gutenbergiana from Germany is enigmatic due to poor preservation, and its attribution to the genus Ephydatia may be questioned, while (pre) middle Eocene Ephydatia kaiseri from southern Africa may belong to this genus but without certainty. On the other hand, the most recent report of Ephydatia cf. facunda from the middle Eocene of Northern Canada (Pisera et al. 2016) raises no doubts about its taxonomic placement. Lower Cretaceous Spongilla patagonica from Argentina is here considered a doubtful taxon, in agreement with previous studies (Racek and Harrison 1974; Manconi and Pronzato 2002) and its taxonomic position must yet be clarified.
Undoubted representatives of the genera Potamophloios and Oncosclera (both family Potamolepidae) are from the middle Eocene and early Miocene, respectively. We are aware, however, of yet undescribed highly diversified freshwater sponge assemblages (several species and genera) from the middle Eocene of Northern Canada (Pisera 2014; Pisera et al. 2016); this find may have important consequence to understand freshwater sponge evolution, but it would be inappropriate to discuss here these mostly unpublished data in detail.
From an evolutionary point of view Spongillida are considered monophyletic also in agreement with recent molecular studies (Morrow and Cardenas 2015). However morphological data indicate that their closest parent taxa are marine Haplosclerida (Manconi and Pronzato 2002, 2015, 2016a, b) living in salty to brackish waters with a wide range of salinity and able to produce gemmules, e.g., Chalinula loosanoffi (Fell 1974, 1978, 1995) whose gemmular theca is devoid of pneumatic layer and gemmuloscleres. After invading continental waters, gemmules evolved acquiring a pneumatic layer and specialized gemmuloscleres in the protective theca allowing cryptobiosis and overland dispersal (Manconi and Pronzato 2002, 2015, 2016a, b). Gemmules are the key structure for interpretation the evolutionary history and adaptive success of Spongillida and for their persistence and dispersal also in extreme stressfull ephemeral environments, from Arctic permafrost (e.g., North Siberia, North Canada) to alluvial lands (e.g., Pantanal, Brazil) and deserts (e.g., Tassili n’Ajjer, Algeria) (Manconi and Pronzato 2016a, b). As indicated by the fossil record discussed above, starting from the Mesozoic, freshwater sponges producing gemmules spread in all continents, radiating into the ca. 240 species presently known (Manconi and Pronzato 2008, 2015). The exceptionally preserved Palaeospongilla chubutensis (Early Cretaceous) exhibit a gemmule structurally identical to that of modern species. Also gemmules of all presently known Cenozoic fossil freshwater sponge species share several traits. These observations clearly indicate that freshwater sponges must have a much longer undisclosed evolutionary history as suggested by the pronounced morphological stasis of skeletal structures at least since Early Cretaceous.
Unfortunately, the very punctuated fossil record of freshwater fossil sponges (see Pisera 2004, 2006), that is typical for continental water deposits, is clearly highly incomplete, which hampers the detailed reconstruction of their evolutionary history. Also data on present geographic distribution of Spongillida are not exhaustive (see Manconi and Pronzato 2002, 2004, 2005, 2007, 2008, 2009; Pronzato and Manconi 2001; Manconi et al. 2008), but the palaeontological approach together with historical biogeography could help clarify when, where, and how colonisation processes occurred during the geological past. The present analysis of literature reports focusing on the age and morphotraits of fossil freshwater sponge remains suggests Carboniferous invasion on the Laurasia continent contemporarily with the invasion of continental waters by Ostracoda (Iglikowska 2014), Foraminifera and other taxa (Zatoń et al. 2012) and a successive expansion to the Gondwana. This picture may change considerably in the future as a result of new findings, that must be expected taking into account the incompleteness of their known fossil records and new yet undescribed Eocene fauna from Canada (Pisera et al. 2014).
Palaeontology is also crucial to calibrate the molecular trees to test phylogenetic hypotheses (Pisera 2004) although these fundamental data are not yet considered in the framework of molecular analyses (Manconi and Pronzato 2011). There are numerous reports of fossil sponge spicules from lacustrine sediments of Cretaceous and Cenozoic ages, that were never studied taxonomically, thus new studies to fill the gap in the past history of this taxon of Porifera are both possible and urgently needed.
Acknowledgements
This work was funded in part by the Regione Autonoma della Sardegna (project LR7/2007 RAS2012/CRP60215) and Fondazione Sardegna with support to RP, RM, and AP and from the National Science Center in Poland (grant project no. 2012/07/B/ST10/04186 to AP). We thank two anonymous reviewers for improving this paper.
References
Arndt, W. 1936. Die von Dr. A. Monard in Angola gesammelten Suswasserschwamme. Mit einem Uberblick uber die Spongilliden-fauna Africas nach dem gegenwartigen Stand unserer Kentnisse. Arquivos do Museu Bocage 7: 7–35.
Bitner, M.A. 1990. Middle Miocene (Badenian) brachiopods from the Roztocze Hills, south-eastern Poland. Acta Geologica Polonica 40: 129–157.
Bitner, M.A. and Pisera, A. 1979. Brachiopods from the Upper Cretaceous chalk of Mielnik (Eastern Poland). Acta Geologica Polonica 29: 67–88.
Cárdenas, P., Pérez, T., and Boury-Esnault, N. 2012. Sponge systematics facing new challenges. Advances in Marine Biology 61: 79–209. Crossref
Cayeux, L. 1929. Les Roches sédimentaires de France. Roches siliceuses. viii + 696 pp. Imprimerie nationale, Paris.
Chang, S., Feng, Q., Clausen, S., and Zhang, L. 2017. Sponge spicules from the lower Cambrian in the Yanjiahe Formation, South China: The earliest biomineralizing sponge record. Palaeogeography, Palaeoclimatology, Palaeoecology 474: 36–44. Crossref
Clements, R.G. 1967. Some notes on the Purbeck Beds. Proceedings of the Dorset Natural History and Archaeological Society 88: 43–44.
Copeland, J., Pronzato, R., and Manconi, R. 2015. Discovery of living Potamolepidae (Porifera: Spongillina) from Nearctic freshwater with description of a new genus. Zootaxa 3957: 37–48. Crossref
de Laubenfels, M.W. de 1955. Porifera. In: R.C. Moore (ed.), Treatise on Invertebrate Palaeontology, Part E, Archaeocyatha and Porifera, E21–E112. University of Kansas, Lawrence and Geological Society of America, Boulder.
Du, W., Wang, X.L., and Komiya, T. 2015. Potential Ediacaran sponge gemmules from the Yangtze Gorges area in South China. Gondwana Research 28: 1246–1254. Crossref
Dunagan, S.P. 1999. A North American freshwater sponge (Eospongilla morrisonensis new genus and species) from the Morrison Formation (Upper Jurassic), Colorado. Journal of Paleontology 73: 389–393. Crossref
Erpenbeck, D. and Wörheide, G. 2007. On the molecular phylogeny of sponges (Porifera). Zootaxa 1668: 197–126.
Fell, P.E. 1974. Diapause in the gemmule of the marine sponge Haliclona loosanoffi, with a note on the gemmule of Haliclona oculata. Biological Bulletin 147: 333–351. Crossref
Fell, P.E. 1978. Variation in the time of annual degeneration of the estuarine sponge, Haliclona loosanoffi. Estuaries 1: 261–264. Crossref
Fell, P.E. 1995. Deep diapause and the influence of low temperature on the hatching of the gemmules of Spongilla lacustris (L.) and Eunapius fragilis (Leidy). Invertebrate Biology 114: 3–8. Crossref
Fell, P.E. 1993. Porifera. In: K.G. Adiyodi and R.G. Adiyodi (eds.), Reproductive Biology of Invertebrates VI, A, Asexual Propagation and Reproductive Strategies, 1–44. IBH Publishing Co., New Delhi.
Firtion, F. 1944. Spongilles et Chrysostomatacees d’un lignite de la formation cineritique du Lac Chambon(Puy-de-Dome). Bulletin de la Société Géologique de France 5 (14): 331–346.
Frisone, V., Pisera, A., and Preto, N. 2016. A highly diverse siliceous sponge fauna (Porifera: Hexactinellida, Demospongiae) from the Eocene of north-eastern Italy: systematics and paleoecology. Journal of Systematic Palaeontology 19: 949–1002. Crossref
Frost, T.M. 2001. Freshwater sponges. In: J.P. Smol, H.J.B. Birks, and W.M. Last (eds.), Tracking Environmental Change Using Lake Sediments. Volume 3: Terrestrial, Algal, and Siliceous Indicators, 253–262. Kluwer Academic Publishers, Dordrecht.
Gehling, J.G. and Rigby, J.K. 1996. Long expected sponges from the Neoproterozoic Ediacarafauna of South Australia. Journal of Paleontology 70: 185–195. Crossref
Geyer, O.F. 1962. Über Schwammgesteine (Spongiolith, Tuberolith, Spiculit und Gaizit). In: O.F. Geyer (ed.), Hermann-Aldinger-Festschrift, 51–59. Schweizerbart, Stuttgart.
Greensmith, J.T. 1978. Textbook of Petrology. 2: Petrology of the Sedimentary Rocks. 6th Edition. 241 pp. Allen and Unwin, London.
Harrison, F.W. 1988. Utilization of freshwater sponges in paleolimnological studies. In: J. Gray (ed.), Paleolimnology: Aspects of Freshwater Paleoecology and Biogeography, 387–397. Elsevier, Amsterdam. Crossref
Harrison, F.W. 1988. Utilization of freshwater sponges in paleolimnological studies. Palaeogeography, Palaeoclimatology, Palaeoecology 62: 387–397. Crossref
Harrison, F.W. 1990. Freshwater Sponges. In: G.B. Warner (ed.), Methods in Quaternary Ecology. Geoscience Canada Reprint Ser. 5, 75–80. Geological Association of Canada, Toronto.
Harrison, F.W. and Warner, B.G. 1986. Fossil freshwater sponges (Porifera: Spongillidae) from Western Canada: An overlooked group of Quaternary paleoecological indicators. Transactions of the American Microscopical Society 105: 110–120. Crossref
Harrison, F.W., Gleason, P.J., and Stone, P.A. 1979. Paleolimnology of Lake Okeechobee, Florida: An analysis utilizing spicular components of freshwater sponges (Porifera: Spongillidae). Notulae Naturae of the Academy of Natural Sciences of Philadelphia 454: 1–6.
Hinde, G.J. 1883. Catalogue of the Fossil Sponges in the Geological Department of the British Museum (Natural History). 248 pp. Taylor & Francis, London.
Hooper, J.N.A., Van Soest, R.W.M., and Pisera, A. 2011. Phylum Porifera Grant, 1826. In: Z.-Q. Zhang (ed.), Animal biodiversity: An outline of higher-level classification and survey of taxonomic richness. Zootaxa 3148: 1–237.
Iglikowska, A. 2014 Stranded: The conquest of fresh water by marine ostracods. Paleontological Research 18: 125–133. Crossref
Kirkpatrick, R. 1907. Notes on two species of African freshwater sponges. Annals and Magazine of Natural History 20: 523–525. Crossref
Machado,V.S., Volkmer-Ribeiro, C., and Iannuzzi, R. 2012. Inventary of the sponge fauna of the cemitério Paleolake, Catalão, Goiás, Brazil. Anais da Academia Brasileira de Ciências 84: 17–34. Crossref
Machado,V.S., Volkmer-Ribeiro, C., and Iannuzzi, R. 2014. Late Pleistocene climatic changes in Central Brazil indicated by freshwater sponges. International Journal of Geosciences 5: 799–815. Crossref
Manconi, R. 2008. The genus Ephydatia (Spongillina: Spongillidae) in Africa: a case of Mediterranean vs Southern Africa disjunct distribution. Biogeographia 29: 19–28. Crossref
Manconi, R. and Desqueiroux-Faundez, R. 1999. Freshwater sponges (Porifera, Spongillidae) from the Lake of Geneva, Switzerland. Revue Suisse de Zoologie 106: 571–580. Crossref
Manconi, R. and Pronzato, R. 1994. Spongillids of Mediterranean islands. In: R.W.M. Van Soest, T.M.G. Kempen, and J.C. Braekman (eds.), Sponges in Time and Space, 333–340. Balkema, Rotterdam.
Manconi, R. and Pronzato, R. 2002. Spongillina new suborder, Lubomirskiidae, Malawispongiidae n. fam., Metaniidae, Metschnikowiidae, Palaeospongillidae, Potamolepiidae, Spongillidae. In: H.J.N. Hooper and R.W.M. Van Soest (eds.), Systema Porifera: A Guide to the Classification of Sponges, Vol. 1, 921–1019. Kluwer Academic/Plenum Publishers, New York. Crossref
Manconi, R. and Pronzato, R. 2004. The genus Corvospongilla Annandale (Haplosclerida, Spongillina, Spongillidae) with description of a new species from eastern Mesopotamia, Iraq. Archiv für Hydrobiologie, Supplement, Monographic Studies 151: 161–189.
Manconi, R. and Pronzato, R. 2005. Freshwater sponges of the West Indies: discovery of Spongillidae (Haplosclerida, Spongillina) from Cuba with biogeographic notes and a checklist for the Caribbean area. Journal of Natural History 39: 3235–3253. Crossref
Manconi, R. and Pronzato, R. 2007. Gemmules as a key structure for the adaptive radiation of freshwater sponges: A morpho-functional and biogeographical study. In: M.R. Custódio, G. Lôbo-Hajdu, E. Hajdu, and M. Muricy (eds.), Porifera Research: Biodiversity, Innovation, Sustainability, Serie Livros 28, 61–77. Museu Nacional, Rio de Janeiro.
Manconi, R. and Pronzato, R. 2008. Global diversity of sponges (Porifera: Spongillina) in freshwater. Hydrobiologia 595: 27–33. Crossref
Manconi, R. and Pronzato, R. 2009. Atlas of African freshwater sponges. Studies in Afrotropical Zoology 295: 1–214.
Manconi, R. and Pronzato, R. 2011. Suborder Spongillina (freshwater sponges). In: M. Pansini, R. Manconi, and R. Pronzato (eds.), Porifera I. Calcarea, Demospongiae (partim), Hexactinellida, Homoscleromorpha. Fauna d’Italia, Vol. XLVI, 341–366. Calderini-Il Sole24Ore, Bologna.
Manconi, R. and Pronzato, R. 2015. Chapter 8 Phylum Porifera. In: J. Thorp and D.C. Rogers (eds.), Thorp and Covich’s Freshwater Invertebrates, Vol. I: Ecology and General Biology. 4th Edition, 133–157. Elsevier, London.
Manconi, R. and Pronzato, R. 2016a. Chapter 3 Phylum Porifera. In: J. Thorp and D.C. Rogers (eds.), Thorp and Covich’s Freshwater Invertebrates, Vol. II: Keys to Nearctic Fauna, 4th Edition, 39–83. Elsevier, San Diego.
Manconi, R. and Pronzato, R. 2016b. How to survive and persist in temporary freshwater? Adaptive traits of sponges (Porifera, Spongillida). A review. Hydrobiologia 782: 11–22. Crossref
Manconi, R., Murgia, S., and Pronzato, R. 2008. Sponges from African inland waters: the genus Eunapius (Haplosclerida, Spongillina, Spongillidae). Fundamental and Applied Limnology (Archiv für Hydrobiologie) 170: 333–350. Crossref
Manconi, R., Ruengsawang, N., Ledda, F.D., Hanjavanit, C., and Sangpradub, N. 2012. Biodiversity assessment in the Lower Mekong Basin: First record of the genus Oncosclera (Porifera: Spongillina: Potamolepidae) from the Oriental Region. Zootaxa 3544: 41–51.
Manconi, R., Ruengsawang, N., Vannachak, V., Hanjavanit, C., Sangpradub, N., and Pronzato, R. 2013. Biodiversity in South East Asia: an overview of freshwater sponges (Porifera: Demospongiae: Spongillina). Journal of Limnology 72 (s2): 313–326. Crossref
Marra, M.V., Cadeddu, B., Giacobbe, S., and Manconi, R. 2013. Porifera of Lake Faro (Sicily): gemmules of a sponge epibiont on Mytilus galloprovincialis. Biologia Marina Mediterranea 20: 140–141.
Matsuoka, K. 1983. Pleistocene freshwater sponges (Porifera: Spongillidae) from Katata Formation of the Kobiwako Group, Shiga Prefecture, Central Japan. The Journal of Earth Sciences 31: 1–16.
Matsuoka, K. and Masuda, Y. 2000. A new potamolepid freshwater sponge (Demospongiae) from the Miocene Nakamura Formation, central Japan. Palaeontological Research 4: 131–137.
Melis, P., Riesgo, A., Taboada, S., and Manconi, R. 2016. Coping with brackishwater: A new species of cave-dwelling Protosuberites (Porifera: Demospongiae: Suberitidae) from the Western Mediterranean and a first contribution to the phylogenetic relationships within the genus. Zootaxa 4208: 349–364. Crossref
Morrow, C. and Cárdenas, P. 2015. Proposal for a revised classification of the Demospongiae (Porifera). Frontiers in Zoology 12: 1–27. Crossref
Müller, W.E.G., Zahn, R.K., and Maidhof, A. 1982 Spongilla guttenbergiana n. sp. ein Süswasserschwamm aus dem Mittel-Eozane von Messel. Senckenbergiana lethaea 63: 465–472.
Ott, E. and Volkheimer, W. 1972. Palaeospongilla chubutensis n. g. et n. sp. Ein Süsswasserschwamm aus der Kreide Patagoniens. Neues Jahrbuch für Geologie und Paläontologie Abhandlungen 140: 49–63.
Parolin, M., Volkmer-Ribeiro, C., and Stevaux, J.C. 2007. Sponge spicules in peaty sediments as palaeoenvironmental indicators of the holocene in the upper Paraná river, Brazil. Revista Brasileira de Paleontologia 10: 17–26. Crossref
Penney, J.T. and Racek, A.A. 1968. Comprehensive revision of a worldwide collection of freshwater sponges (Porifera: Spongillidae). Proceedings of the United States National Museum 272: 1–184. Crossref
Petit, G. and Charbonnier, S. 2012. Fossil sponge gemmules, epibionts of Carpopenaeus garassinoi Charbonnier n. sp. (Crustacea, Decapoda) from the Sahel Alma Lagerstätte (Late Cretaceous, Lebanon). Geodiversitas 34: 359–372. Crossref
Pisera, A. 1997. Upper Jurassic siliceous sponges from the Swabian Alb: taxonomy and paleoecology. Palaeontologia Polonica 57: 1–216.
Pisera, A. 1999. Postpaleozoic history of the siliceous sponges with rigid skeleton. Memoires of the Queensland Museum 44: 473–478.
Pisera, A. 2004. What can we learn about siliceous sponges from paleontology. Bollettino dei Musei e degli Istituti Biologici dell’Università di Genova 68 (for 2003): 55–69.
Pisera, A. 2006. Palaeontology of sponges—a review. Canadian Journal of Zoology 84: 242–261. Crossref
Pisera, A. 2010. A high diversity Middle Eocene freshwater sponge fauna from the Giraffe Pipe crater lake, Canada. In: Ancient Animals New Challenges. Book of Abstracts VIII World Sponge Conference 2010, 93. Girona.
Pisera, A. and Saez, A. 2003. Paleoenvironmental significance of a new species of freshwater sponge from the Late Miocene Quillagua Formation (N Chile). Journal of South American Earth Studies 15: 847–852. Crossref
Pisera, A. and Tabachnick, K. 2014. The sponge genus Laocoetis Pomel, 1972 (Hexactinosida, Craticulariidae): a living fossil? In: C.V. Rubinstein, C.A. Marsicano, and B.G. Waisfeld (eds.), The History of Life: A View from the Southern Hemisphere, Abstract Volume, 4th International Paleontological Congress, Mendoza Sept. 28 – Oct. 3, 2014, 782. CONICET, Mendoza.
Pisera, A., Manconi, R., Siver, P., and Wolfe, A.
2016. The sponge genus Ephydatia from the
Middle Eocene: environmental and evolutionary significance. Palaeontologische
Zeitschrift 90: 673–680. Crossref
Pisera, A., Rützler, K., Kaźmierczak, J., and Kempe, S. 2010. Sponges in an extreme environment: suberitids from quasi-marine Satonda Island crater lake (Sumbawa, Indonesia). Journal of Marine Biological Association of the United Kingdom 90: 203–212. Crossref
Pisera, A., Siver, P., and Wolfe, A. 2014. Middle Eocene lake deposits from the Giraffe crater, Northern Canada: a window on freshwater sponge evolution. In: C.V. Rubinstein, C.A. Marsicano, and B.G. Waisfeld (eds.), The History of Life: A View from the Southern Hemisphere, Abstract Volume, 4th International Paleontological Congress, Mendoza Sept. 28 – Oct. 3, 2014, p. 595. CONICET, Mendoza.
Potts, E. 1888. Contributions towards a synopsis of the American forms of fresh-water Sponges with descriptions of those named by other authors and from all parts of the world. Proceedings of the Academy of Natural Sciences of Philadelphia 39 (for 1887): 158–279.
Pronzato, R. and Manconi, R. 1994. Life history of Ephydatia fluviatilis: a model for adaptive strategies in discontinuous habitats. In: R.W.M. Van Soest, Th.M.G., Van Kempen, and J.C. Braekman (eds.), Sponges in Time and Space, 327–331. Balkema, Rotterdam.
Pronzato, R. and Manconi, R. 2001. Atlas of European freshwater sponges. Annali del Museo Civico di Storia Naturale di Ferrara 4: 3–64.
Racek, A.A. 1966. Spicular remains of freshwater sponges. In: U.M. Cowgill, G.E. Hutchinson, A.A. Racek, C.E. Goulden, R. Patrick, and M. Tsukada (eds.), The History of Laguna de Petenxil: A Small Lake in Northern Guatemala. Memoirs of the Connecticut Academy of Arts and Sciences 17: 78–83.
Racek, A.A. and Harrison, F.W. 1975. The systematic and phylogenetic position of Palaeospongilla chubutensis (Porifera: Spongillidae). Proceedings of the Linnean Society of New South Wales 99: 157–165.
Rauff, H. 1926. Über prämitteleozäne fossilfuhrende Süswasser-Hornsteine aus der Namib. In: E. Kaiser (ed.), Die Diamantenwuste Südwest-Afrikas, Bd. 2, 160–166. D. Deimer, Berlin.
Rezvoi, P.D., Zhurarleva, I.T., and Koltun, V.M. 1971. Porifera. In: Y.A. Orlov (ed.), Fundamentals of Palaeontology. II Porifera, Archeocyata, Coelenterata, 18–127. Vermes. Israel Program for Scientific Translation, Jerusalem.
Richter, G. and Baszio, S. 2000. Zur Taphonomie von Spongilliden (Porifera)—I. Korrosionserscheinungen an fossilen Skleren der Grube Messel. Senckenbergiana lethaea 80: 445–461.
Richter, G. and Baszio, S. 2001. Zur Taphonomie von Spongillidenskleren (Porifera)—II. Aktuopaläontologische Untersuchungen. Senckenbergiana lethea 81: 59–69.
Richter, G. and Baszio, S. 2009. Geographic and stratigraphic distribution of spongillids (Porifera) and the leit value of spiculites in the Messel Pit Fossil Site. Paleobiodiversity and Palaeoenvironments 89: 53–66. Crossref
Richter, G. and Wuttke, M. 1995. Der Messel Süsswasser-Kieselschwamm Spongilla gutenbergiana, eine Ephydatia. Natur und Museum 125: 134–135.
Richter, G. and Wuttke, M. 1999. Lutetiospongilla heili n. gen. n. sp. und die eozäne Spongillidefauna von Messel. Courier Forschung-Institute Senckenberg 216: 183–195.
Richter, G., Baszio, S., and Wuttke, M. 2017. Discontinuities in the microfossil record of middle Eocene Lake Mesel: clues for ecological changes in lake’s history? Paleobiodiversity and Palaeoenvironments 97: 295–314. Crossref
Rigby, J.K. (ed.) 2004. Porifera revised. In: R.L. Kaesler (ed.), Treatise on Invertebrate Paleontology. Part. E Porifera, Revised. Vol. 3: Porifera (Demospongea, Hexactinellida, Heteractinida, Calcarea), 1–872. Geological Society of America, Boulder and University of Kansas, Lawrence.
Schindler, T., Wuttke, M., and Poschmann, M. 2008. Oldest record of freshwater sponges (Porifera: Spongillina) spiculite finds in the Permo-Carboniferous of Europe. Paläontlogische Zeitschrfit 82: 373–384. Crossref
Schultze, H.P. 2009. Interpretation of marine and freshwater paleoenvironments in Permo-Carboniferous deposits. Palaeogeography, Palaeoclimatology, Palaeoecology 291: 126–136. Crossref
Siver, P.A. and Wolfe, P.A. 2009. Tropical ochrophyte algae from the Eocene of northern Canada: a biogeographical response to past global warming. Palaios 24: 192–198. Crossref
Traxler, L. 1894. Ephydatia fossilis, eine neu Art der fossilen Spongilliden. Földatni Közlöny 24: 234–237.
Van Soest, R.W.M. Van, Boury-Esnault, N., Hooper, J.N.A., Rützler, K., Voogd de, N.J., Alvarez Glasby, B., Hajdu, E., Pisera, A., Manconi, R., Schoenberg, C., Janussen, D., Tabachnick, K.R., Klautau, M., Picton, B., Kelly, M., and Vacelet, J. 2017. World Porifera Database. Available online at http://www.marinespecies.org/porifera.
Vodrážka, R. and Crame, A. 2011. First fossil sponge from Antarctica and its paleobiogeographical significance. Journal of Paleontology 85: 48–57. Crossref
Volkmer-Ribeiro, C. and Machado, V.S. 2007. Freshwater sponges (Porifera, Demospongiae) indicators of some coastal habitats in South America: redescriptions and key to identification. Iheringia, Série Zoologia 97: 157–167. Crossref
Volkmer-Ribeiro, C. and Reitner, J. 1991. Renewed study of the type material of Palaeospongilla chubutensis Ott and Volkheimer (1972). In: J. Reitner and H. Keupp (eds.), Fossil and Recent Sponges, 121–133. Springer, Berlin.
Volkmer-Ribeiro, C., de Ezcurra, D., and Parolin, M. 2007. Spicules of the freshwater sponge Ephydatia facunda indicate lagoonal palaeoenvironment at the Pampas of Buenos Aires Province, Argentina. Journal of Coastal Research 50: 449–452.
Volkmer-Ribeiro, C., Marques, D.M., De Rosa-Barbosa, R., and Machado, V.S. 2004. Sponge spicules in sediments indicate evolution of coastal freshwater bodies. Journal of Coastal Research 39: 469–472.
Volkmer-Ribeiro, C., Marques, D.M., De Rosa-Barbosa, R., and Machado, V.S. 2006. Sponge spicules in sediments indicate evolution of coastal freshwater bodies. Journal of Coastal Research 39: 469–472.
Waldschmidt, W.A. and Leroy, L.W. 1944. Reconsideration of the Morrison Formation in the type area, Jefferson County, Colorado. Geological Society of America Bulletin 55: 1097–1114. Crossref
Weert, W.H. de 2002. Family Chalinidae Gray, 1867. In: J.N.A. Hooper and R.W.M. Van Soest (eds.), Systema Porifera: A Guide to the Classification of Sponges, Vol. 1, 852–873. Kluwer Academic/Plenum Publishers, New York. Crossref
Weltner, W. 1895. Spongillidenstudien III. Katalog und Verbreitung der bekannten Süsswasserschswämme. Archiv für Naturgeschichte 61: 114–144.
Williams, J.L. 1985. Spicular remains of freshwater sponges from a Miocene lacustrine deposit in Northern Idaho. In: C.J. Smiley, A.E. Leviton, and M. Berson (eds.), Late Cenozoic History of the Pacific Northwest, 349–355. Allen Press, Lawrence.
Yen, T. 1952. Molluscan fauna of the Morrison Formation. U.S. Geological Survey Professional Paper 233B: 21–47.
Young, J.T. 1878. On the occurence of a freshwater sponge in the Purbeck Limestone. Geological Magazine 5: 220–221. Crossref
Zatoń, M., Vinn, O., and Tomescu, A.M.F. P. 2012. Invasion of freshwater and variable marginal marine habitats by microconchid tubeworms—an evolutionary perspective. Geobios 45: 603–610. Crossref
Zimmerle, W. 1991. Stratigraphic distribution, lithological paragenesis, depositional environment and diagenesis of fossil siliceous sponges in Europe. In: J. Reitner and H. Keupp (eds.), Fossil and Recent Sponges, 554–577. Springer, Berlin. Crossref
Appendix 1
Fossil freshwater sponge taxa (Porifera, Demospongiae, Spongillida). Morphotraits of the spicular complement and gemmules.
Matris gemmuleanalogi Du, Wang, and Komiya, 2015
Not Porifera
Spongillida gen. et sp. indet. sensu Cayeux, 1929
Monaxial siliceous spicules (200–250 × 10–20 µm)
Spongillida gen. et sp. indet. sensu Schindler et al., 2008
Smooth oxeas with traces of axial canal (379–1250 µm to 300–900 µm in length)
Spongillidae spp. sensu De Laubenfels, 1955
Freshwater siliceous sponges identified as: Spongilla, Meyenia, and Heteromeyenia. No more data.
Eospongilla morrisonensis Dunagan, 1999
Two small specimens (10–15 mm) of freshwater sponges.
Megascleres. Strongyles (dominant) and oxeas, smooth (180–300 × 20–35 µm).
Microscleres. Not detected.
Gemmules. Not detected
Gemmuloscleres. Not detected.
Spongilla purbeckensis Young, 1878
Megascleres. Oxeas (450 × 15–25.5 µm) fusiform, acerate, slightly curved, thickest in the centre, and gradually tapering towards the tips (Hinde 1883). Spicules minutely tubercled according to Young (1878).
Microscleres. Not detected.
Gemmules. Not detected.
Gemmuloscleres. Not detected.
Palaeospongilla chubutensis Ott and Volkheimer, 1972
Megascleres. Oxeas of two types. Oxeas abruptly pointed, stout (230–540 × 10–22 µm) smooth, with rare spines. Oxeas sharply pointed, abundant, slender, finely spiny except at tips (180–308 × 4–14 µm).
Microscleres. Oxeas abruptly pointed, spiny (179–190 × 3–4 µm) to smooth (77–147 × 3–10 µm). Absent following Volkmer–Ribeiro and Reitner (1991).
Gemmuloscleres. Oxeas to strongyles (70–168 × 3.5–8 µm) slender, with scattered spines, more dense and recurved at tips. Absent following Volkmer-Ribeiro and Reitner (1991).
Gemmules. Subspherical (483–644 µm in diameter, 20 µm inner layer, 100 µm pneumatic layer) single or grouped, of two types, with armed and unarmed gemmular theca. Unarmed with cage of spiny megascleres, bilayered theca, foramen simple. Armed with monolayer of variably arranged gemmuloscleres, thick trilayered theca, nipple-like foramen. Only unarmed gemmules following Volkmer-Ribeiro and Reitner (1991).
Spongilla patagonica Volkmer-Ribeiro and Reitner, 1991
Megascleres. Oxeas (210–260 × 7–10 µm), slim, gradually sharp pointed, smooth.
Microscleres. Oxeas (100–110 × 9–10 µm) straight to bent, spiny.
Gemmule. Subspherical (470 µm in diameter, 20 µm inner layer, 100 µm pneumatic layer) with spicules randomly oriented and loosely packed in the thick theca, sometimes protruding tips. Foramen single, nipple-like
Gemmuloscleres. Strongyles (70–80 × 8 µm) short, slightly curved, strongly spiny, with star-like tips.
Lutetiospongilla heili Richter and Wuttke, 1999
Megascleres. Oxeas (300–350 × 8–12 µm), spiny, slender with scattered, fine spines.
Microscleres. Not surely identifiable.
Gemmuloscleres. Two types. Large strongyles (80–220 × 12–20 µm) very variable. Small strongyles to pseudo-oxeas (40–60 × 5–10 μm) very variable, short, slender, squat.
Gemmules. Subspherical (800–1000 µm in diameter; lumen 300–400 µm in diameter) with armed theca (200–350 µm in thickness).
Ephydatia kaiseri Rauff, 1926
Megascleres. Oxeas (350 × 7–17 µm) smooth to slightly tubercled; short oxeas (20 × 3.5 µm) .
Microscleres. Absent.
Gemmules. Fragments of intact gemmules with birotules in natural arrangement.
Gemmuloscleres. Birotules (44–65 µm in length, rotules 18–20 µm in diameter, shaft 5–6 µm in thickness).
Ephydatia gutenbergiana (Müller, Zahn, and Maidhof, 1982)
Megascleres. Oxeas spiny to rarely smooth of two size classes (140–230 × 10–35 µm; 200–350 × 10–20 µm).
Microscleres. Absent.
Gemmule. Structure interpreted as gemmule > 2 mm (?) in diameter, lumen 50–150 µm in diameter (?).
Gemmuloscleres. Oxeas to birotules with corroded rotules (50–150 µm in length).
Potamophloios canadensis Pisera, Siver, and Wolfe, 2013
Megascleres. Strongyles (7.90–30.9 × 3.12–5.0 µm) smooth, thick, with highly variable size, slightly irregular, frequently with inflated tips.
Microscleres. Not detected.
Gemmules. Whole intact gemmules not detected.
Gemmuloscleres. Not detected.
Ephydatia cf. facunda Weltner, 1895
Megascleres. Only disassociated spicules. Possible large oxeas (162.3–307 × 8.7–12.8 µm, usually 220–250 × 9–10 µm) sharply pointed straight to slightly curved, with scattered small spines or tubercles. Some centrotylote present.
Microscleres. Not detected.
Gemmuloscleres. Birotules (26–57 µm in length, average 41.5 µm; rotulae diameter 15–28 µm average 21.4 µm; shaft 5–11 µm in thickness, average 7.2 µm not including spines) with incised margins and spiny shaft; scattered, large spines (4–20, usually 6–12) with acute tips; rotules often umbonate, moderately smooth to strongly incised. Aberrant forms also present.
Oncosclera kaniensis Matsuoka and Masuda, 2000
Megascleres. Strongyles dominant, stout, to occasional oxeas (100–179 × 7–15 µm), almost straight, covered with distinct spines at both tips.
Microscleres. Absent.
Gemmules. Subspherical (ca. 500 µm in diameter), firmly adhering to substrata.
Gemmuloscleres. Strongyles (23–100 × 4–7 µm), stout, variably curved, inflated at middle, densely covered with distinct bent spines numerous at both tips.
Ephydatia chileana Pisera and Sáez, 2003
Megascleres. Oxeas slightly curved, smooth (240–350 × 8–15 µm).
Microscleres. Absent.
Gemmules. Subspherical to hemispherical (237–250 µm in diameter).
Gemmuloscleres. Birotules (39–45 µm in lenght), with spiny shaft (5.5–7.2 µm in thickness), with strong, long conical spines (4–11) of variable length. Rotules (24.8–27 µm) irregularly, often deeply incised, and the resulting teeth (16 to 20–21) of variable length (max. 11 µm).
Ephydatia fossilis Traxler, 1894
Traxler, 1894. Megascleres. Oxeas (160–350 × 5–20 µm) very variable, smooth to spiny.
Microscleres. Absent.
Gemmules. Not detected.
Gemmuloscleres. Birotules stout (41–69 µm in length; rotules 16–26 µm in diameter) with macrospiny shaft (12–16 × 6–8 µm). Rotules with indented margins (9–14 teeth).
Firtion, 1944. From Chambon (France)
Megascleres. Not detected.
Gemmuloscleres birotules straight or weakly curved (56–70 µm in length); shaft (5.6–7 µm in thickness) with strong spines (6.1–7.6 µm in length); equal to sub-equal rotules with indented margins (22.3–25.5 µm in diameter).
From Kevna Bremia (Romania)
Gemmuloscleres birotules stout (64.36–69.02 µm in length, shaft 7.14–8.23 µm in thickness), straight or curved, with strong spines. Rotules (26.18–28.66 µm in diameter) equally developed and with incised margins.
Spongillidae spp. Spicular remains of freshwater sponges Williams, 1985
Anheteromeyenia ryderi (Potts, 1882)
Megascleres. Not observed.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Microbirotules with spiny shaft, rotules flat to slightly umbonate, with slightly incised margins.
Anheteromyenia cf. ryderi (Potts, 1882)
Megascleres. Not observed.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Microbirotules with stout cylindrical smooth shaft, rotules flat to slightly umbonate, with slightly incised margins.
?Anheteromeyenia ryderi (Potts, 1882)
Megascleres. Oxeas, abnormal, fusiform bearing numerous small spines except at tips.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
Corvospongilla sp.
Megascleres. Not detected.
Microscleres. Micro-pseudobirotules straight to slightly curved with smooth shaft and 4–5 terminal hooks.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
?Corvospongilla sp.
Megascleres. Not observed.
Microscleres. Micro-pseudo birotules straight to slightly curved with smooth shaft and 4–5 terminal, very small and stubby hooks.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
Eunapius fragilis (Leidy, 1851)
Megascleres. Not observed.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Strongyles to strongyloxeas, stout, slightly curved, strongly spiny.
Radiospongilla crateriformis (Potts, 1882)
Megascleres. Not observed.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Strongyles, slender, slightly to strongly curved with variable number of conical spines more dense at the tips to form pseudorotules.
Radiospongilla cf. crateriformis (Potts, 1882)
Megascleres. Hastate sharply pointed oxeas with sparse microspines; tips smooth.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
?Radiospongilla crateriformis (Potts, 1882)
Megascleres. Oxeas, hastate sharply pointed, sparsely microspiny with few or no spines at tips.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
?Radiospongilla cantonensis (Gee, 1929)
Megascleres. Oxeas, slender, fusiform and nearly straight, with numerous acute spines, except at the tips where spines are sparse to absent.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
Spongilla lacustris (Linnaeus, 1759)
Megascleres. Not observed.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Oxeas stout to slender slightly to strongly curved with sparse,stout recurved spines.
?Trochospongilla pennsylvanica (Potts, 1882)
Megascleres. Strongyles to oxeas, slender, slightly curved with small, sharp spines.
Microscleres. Not observed.
Gemmules. Not observed.
Gemmuloscleres. Not observed.
Acta Palaeontol. Pol. 62 (3): 467–495, 2017
https://doi.org/10.4202/app.00354.2017