

Ontogeny, muscle scars, colour pattern and predation marks in a Silurian orthoceratid cephalopod
ŠTĚPÁN MANDA and VOJTĚCH TUREK
Manda, Š. and Turek, V. 2025. Ontogeny, muscle scars, colour pattern and predation marks in a Silurian orthoceratid cephalopod. Acta Palaeontologica Polonica 70 (3): 517–541.
Muscle scars, embryonic shell, colour pattern and intraspecific variation are known together in a very limited number of straight shelled cephalopods, leading to uncertainty in the classification of the subclass Orthoceratoidea. The newly described genus Lepidoceras from the Silurian of Bohemia, interpreted as a demersal orthoceratid similar to Kionoceras, shows all relevant diagnostic characters. The earliest late Wenlock populations of Lepidoceras have a weakly curved endogastric shell, but the shell becomes increasingly more curved throughout the Ludlow. The sculpture is characterized by distinctive longitudinal lirae or ridges, the number of which is variable and maintained during ontogeny. The embryonic shell of Lepidoceras is one of the largest in orthoceratids; it is conical or weakly curved in shape beginning with a smooth non-accretion initial shell. Hatching is manifested by growth walls in some specimens; hatchlings were apparently demersal. Adults are usually characterized by mature modifications of the shell: septal crowding and shell wall thickening. A high proportion of adult specimens indicates low mortality in palaeopopulations. Healed repairs indicate a lower predation pressure, which decreases after the juvenile stage. Extensive shell repair in juveniles demonstrates high regenerative capacity. Orthoceratomorph muscle scars found only in adults show a pair of dorsal retractor imprints that gradually expanded laterally. The colour pattern consists of irregular transverse bands, which have never been detected in orthoceratids before. The pattern, which is similar to that in curved oncocerids, is probably an expression of adaptive convergence of this feature in both groups. Shell pathologies, which have been observed in juvenile orthoceratids for the first time, are rare; an extensive pathology in a mature specimen illustrates limit of survival in orthoceratids. Lepidoceras is an example of a demersal orthoceratid entering a niche inhabited predominantly by multiceratoids.
Key words: Cephalopoda, Orthoceratida, ontogeny, variability, palaeoecology, predation.
Štěpán Manda [stepan.manda@geology.cz; ORCID: https://orcid.org/0000-0002-4939-2063 ], Czech Geological Survey, Klárov 3, 118 21 Praha 1, Czech Republic.
Vojtěch Turek [vojtech.turek@nm.cz; ORCID: https://orcid.org/0000-0001-7366-8229 ], National Museum, Department of Palaeontology, Václavské náměstí 68, 115 79 Praha 1, Czech Republic.
Received 28 January 2025, accepted 2 June 2025, published online 10 September 2025.
Copyright © 2025 Š. Manda and V. Turek. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The low number of cephalopod species, for which all significant morphological characters and their intraspecific variation are known, has limited a better understanding of early Paleozoic orthoceratoid phylogeny and auteocology. The diagnoses of many families and genera are based on a combination of characters of different genera or species placed in a given taxon. Adaptive convergence or parallel evolution in the shell form of early Paleozoic cephalopods is thus difficult to detect. Therefore, there is currently no general consensus regarding the classification of non-ammonoid cephalopods with external shells, especially orthoceratoids (Dzik 1984; King and Evans 2019; Niko et al. 2020; Pohle et al. 2022). The common adaptive convergence of orthoceratid shells reinforces the importance of early ontogenetic development for classification, but this is limited because early growth stages are known for only a small number of species (e.g., Kröger 2006; Kröger and Isakar 2006; Manda and Turek 2024). The majority of known early growth stages cannot be identified with taxa, which are defined based on post-juvenile shells (e.g., Ristedt 1968, 1971).
Understanding of predator-prey interactions and their evolution throughout the early Paleozoic is limited by the lack of studies on shell repair and cephalopod associations. There are almost no data documenting how common healed injuries are in individual species, how their frequency varies among associations and through the early Paleozoic (Kröger 2004, 2011; Klug 2007). The Silurian cephalopod fauna of the Prague Basin is known for its species richness and good preservation (Barrande 1865–1877), yet the number of species for which all diagnostic characters or at least the most important ones are known, is small, especially in orthoceratids (Turek and Manda 2012). Any study based on material from historical collections is somewhat limited by collecting biases. The cephalopod shells from limestones in the collections have been carefully removed from the rock matrix and their faunistic and taphonomic context is thus lost (Stridsberg 1985). The apical portion in smaller orthoceratid shells is almost always broken off.
The orthoceratid Lepidoceras gen. nov. from the middle Silurian of the Prague Basin is described herein. The material studied comes from historical collections, mainly the collection of Joachim Barrande, supplemented by the findings of one of the authors (ŠM) providing biostratigraphic control and taphonomic context. Endogastric, moderately expanding shell, distinct reticulate sculpture and thin siphuncle clearly differentiate the new genus Lepidoceras from all co-occurring taxa and make it possible to describe its morphological variability. The type species of Lepidoceras, Cyrtoceras lepidum, was described by Barrande (1866) and has since been mentioned only by Dzik (1984), who placed it in the family Sactoceratidae Troedsson, 1926.
The new findings of the apical parts of the shell allowed the study of the whole ontogeny from the embryonic shell to the shell with mature modification, which is only possible in a limited number of orthoceratid species (e.g., Barrande 1870, 1877; Marek 1971; Kolebaba 1977). In several individuals, muscle imprints are preserved and allow to study variability in the extent of retractors. Orthoceratoid muscle scars have so far been documented only in some species and based on single specimens (Mutvei 1957; Marek 1971; Kröger et al. 2005; Turek and Manda 2012).
The good preservation of relatively complete shells with the shell wall allowed the quantification of sublethal shell repairs. The pattern of shell coloration described here in Lepidoceras is new in orthoceratids, which are usually coloured by longitudinal bands. A pathological shell is also described here; the degree of the malformation is among the most striking yet known in early Paleozoic cephalopods (Kobluk and Mapes 1989; Kröger et al. 2009; Manda and Turek 2015).
Institutional abbreviations.—CGS SM, Czech Geological Survey, Prague, Czech Republic; MCZ, the Shary’s collection of the Museum of Comparative Zoology, Harvard, USA; NM L, palaeontological collection of the National Museum, Prague, Czech Republic.
Nomenclatural acts.—This published work and the nomenclatural act it contains have been registered in ZooBank urn:lsid:zoobank.org:act:47BD78E7-F127-48E5-8B91-152D0A55557A.
Geological setting
The Paleozoic of the Barrandian area (Central Bohemia) represents the sedimentary cover of the pre-Cadomian Teplá-Barrandian Unit of the Bohemian Massif. The unmetamorphosed Silurian rocks are preserved in the central part of the Prague Synform, SW of Prague. The Prague Synform formed during the Variscan orogeny which substantially modified the sedimentary and volcanic infill of the former Early Ordovician–Middle Devonian marine basin, called the Prague Basin (for summary see Havlíček 1981, 1998). The facies distribution, the geometry of basin infill and the geochemistry of the volcanic rocks indicate a rather continuous marine deposition in an intra-plate linear rift basin divided into several sub-basins by synsedimentary faults (Kříž 1998b). There is no indication of a continental shoreline after the early Middle Ordovician. The preserved Silurian rocks represent an erosional remnant of the off-shore part of the basin without a significant amount of continental siliciclastic input (Kříž 1998b).
The Silurian part of the Prague Basin infill consists of five formations. The herein studied material comes from the upper part of the Motol Formation and the Kopanina Formation, the lower boundary of which coincides with base of the Ludlow. The upper part of the Motol (Homerian, Wenlock) and the lower part of the Kopanina (Gorstian, Ludlow) formations consist of volcano-sedimentary and shale dominated facies. Volcanic activity formed several bathymetric highs of a largely submarine volcanic archipelago with shallow-water domains surrounded by off-shore laminites (Kříž 1998b). The largest volcano, the Svatý Jan Volcanic Centre, occasionally reached above the sea level as documented by subaerial basalts, plant remains and spores occurring in sediments close to the island shore (Kříž 1992; Bek et al. 2022). A basalt sheet, formed in the lower part of the Kopanina Formation across the Svatý Jan Volcanic Centre in the total area of about several square kilometres, emerged above sea level and forms an island from earlier Ludlow until latest Přídolí (Kříž 1998b). Cephalopod fossils are known from several sites, usually from local limestone beds within volcano-sedimentary facies. Several local benthic communities inhabited upper and lower slopes of the volcanic archipelago (Havlíček 1995; Kříž 2008; Mergl 2024) and the cephalopod assemblages are also differentiated (ŠM unpublished data). Cephalopod limestones are recurrent, time-specific facies, consisting of skeletal accumulations of current-oriented cephalopod shells; the shells were deposited in areas, where the surface currents reach the bottom (Ferretti and Kříž 1995; Kříž 1998a; Histon 2012). Cephalopod limestones form thin beds in distal volcano-sedimentary facies. The cephalopod limestones contains a specific bivalve-dominated benthic community, as well as a specific cephalopod assemblage in the Lundgreni, Nilssoni, and lower and upper Scanicus biozones (Kříž 1998a, 1999; ŠM unpublished data). Shales that deposited in an oxygen depleted environment contain abundant pelagic cephalopods accompanied by very occasional stray migrants from shallow water environment (Turek 1983; Manda et al. 2019).
The upper part of the Kopanina Formation (Ludfordian, Ludlow) consists of carbonate and shale-dominated facies. The carbonate facies occur on former volcanic bottom highs. The shallow, light-coloured limestones pass basin-ward into grey muddy limestones. The carbonate facies rapidly changes laterally, progradation or flooding of carbonate platforms reflects sea level oscillations and local subsidence or uplift of basinal segments or their parts. The shallow water domains were surrounded by off-shore shales (Kříž 1998b; Manda and Kříž 2006). Cephalopod limestones form up to several meters’ thick succession deposited on a distal carbonate platform (Braník type of Ferretti and Kříž 1995). Locally, the cephalopod limestones deposition persisted throughout most of the Ludfordian (Kříž 1998a); the low thickness and faunal succession indicates condensation and a gap in sedimentation in the mid-Ludfordian (Manda and Kříž 2006). Thin beds of cephalopod limestones occur also in the shallow water succession (Kosov type of Ferretti and Kříž 1995). The cephalopod fauna is diverse and most taxa are known from the cephalopod limestones. Cephalopods are missing in the shallow-water subtidal limestones. There is a succession of cephalopod assemblages across the Ludfordian; a distinct assemblage occurs in the Leintwardinensis, Inexpectatus, Kozlowskii, upper Latilobus-balticus and Fragmentalis zones (Kříž 1998a; ŠM unpublished data). The cephalopod faunas of the distal and proximal carbonate facies are different; pelagic and nektonic forms disappear towards the shore (ŠM unpublished data). Off-shore shales contain cephalopod faunas resembling those from the Wenlock and lower Ludlow (e.g., Štorch et al. 2014).
The orientation of graptolite rhabdosomes and cephalopod shells indicate a stable surface current system directed from the south to the northwest, considering present structural position of the Prague Synform (Turek 1983; Ferretti and Kříž 1995). The gradual transition from shallow water limestones to off-shore shales and benthic fauna in shales suggests that the maximum depth of the basin of about 200 metres was attained (e.g., Manda et al. 2019).
Material and methods
The studied material originates mostly from Barrande’s (1865–1877) collection. The material from the Motol and the lower part of the Kopanina formations was completed by material collected by the author (ŠM) in the last decades. Field revision of Barrande’s (1865–1877) localities enabled precise age determination of historical collections. Specimens from cephalopod limestone of the upper part of the Kopanina Formation come, with the exception of a single new specimen, from Barrande’s (1865–1877) localities embracing several sites within the Ludfordian, Ludlow, and lowermost Přídolí strata; the exact age determination is difficult in these specimens. A single, recently collected specimen from the lithologically distinct, light-coloured, rusty cephalopod limestone bed exposed in Koněprusy, Velký vrch (Big Hill) road cut enabled to determine age of specimens from Barrande’s (1865–1877) locality Konieprus e2. The studied material is generally well preserved.
The fundamental morphological terminology follows that of Teichert (1964). The terms “exogastric” and “endogastric” are used as defined by Teichert (1964). According to that definition, the endogastric shell has the venter on the concave side. In Lepidoceras, the venter is determined by the hyponomic sinus on the concave side of the shell and the dorsal furrow on the convex side of the shell.
The longitudinal lirae on the shell surface of Lepidoceras correspond in extent to the distinctly raised growth lines of Pohle et al. (2022). Longitudinal ridges differ from lirae in a raised profile protruding from the surface of the shell; the width of the ridges roughly equals or exceeds their height (Manda and Turek 2025). Pohle et al. (2022) called this sculptural element ribs, but ribs form a number of growth lines or lirae, while longitudinal ridges are a single narrow raised sculptural element on which no other sculptural element can be distinguished (Fig. 1). Longitudinal lirae can turn into ridges during shell growth. The transverse thickening of the shell wall that is not expressed on the internal mould is termed growth walls. Teichert (1964) also considered growth walls to be a less common type of annulation, but the distinction between the growth walls and annulation is useful for describing the ontogeny of sculpture. Pohle et al. (2022: appendix 1: 2) referred to growth walls by the term ribs, but in ammonites and nautilids the term ribs usually corresponds to what is called annulation in orthoceratoids. Therefore, the use of the term rib in orthoceratoids is confusing.
It is difficult to express the degree of curvature of the Lepidoceras shell because the apical part of the shell is preserved only exceptionally. The curvature of the shell is expressed here by the ratio of the shell diameter (SD) to the shell height (SH); this ratio is lower than it would be in completely preserved shells. In curved shells, the shell diameter equals shell height plus the projected distance to the apex. The main measured parameters are shown in Fig. 1, measurements are listed in Table 1.
The terminology of muscle scars follows Sweet (1959), Teichert (1964), and King and Evans (2019), while the terminology of annular elevation is after Mutvei (1957) and Mutvei et al. (1993).
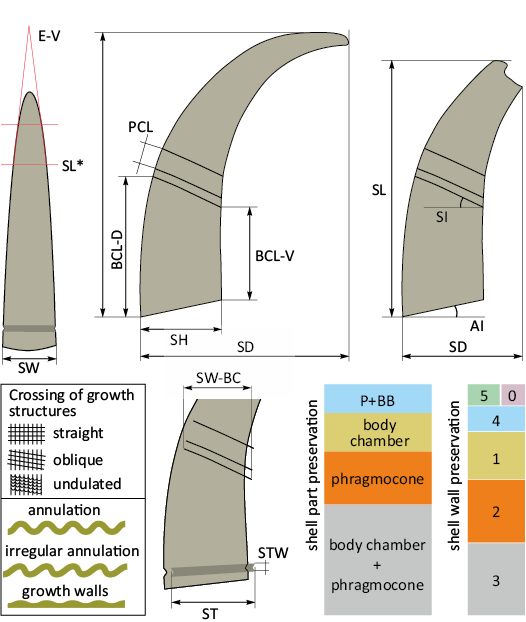
Fig. 1. Schematic drawing explaining the shell morphology of Lepidoceras lepidum (Barrande, 1866); the diagrams in the right lower corner show proportional representation of shell parts and shell wall preservation in the material studied; for categories see Material and methods. Abbreviations: AI, aperture inclination; BCL-D, body chamber length dorsally; BCL-V, body chamber length ventrally; E-V, shell expansion ventrally (in degree); SD, shell diameter; SL, shell length; SL*, shell lenght of complete shell; SH, shell height; SI, suture inclination, P+BB, phragmocone with base of body chamber; PCL, phragmocone chamber length; ST, shell wall thickening at aperture, shell height position of maximal thickening; STW, shell wall thickening width; SW, maximum shell width; SW-BC, shell width at the base of the body chamber. All dimensions in mm.
Table 1. Inventory numbers (type specimens in bold), locality, age, measurements and taphonomic characteristic of Lepidoceras lepidum. Abbreviations: AI, aperture inclination (in degrees); AP, aperture; B, body chamber; BB, base of the body chamber; BCL-D, body chamber length dorsally; BCL-V, body chamber length ventrally; E-L, shell expansion laterally (in degree); E-V, shell expansion ventrally (in degree); n, not observed; nd, not developed; NL, number of longitudinal lirae/ridges around shell circumference; P, phragmocone; p, present; SC, septal crowding; SD, shell diameter; SH, maximum shell height; SH-BC, shell height at the base of the body chamber; SI, suture inclination (in degrees); SL, shell length; SP, shell preservation; SR, shell repair; ST, shell wall thickening at aperture, shell height position of maximal thickening; STW, shell wall thickening width; SW, maximum shell width; SW-BC, shell width at the base of the body chamber; un, unmeasurable.
|
Specimen |
Locality |
Age |
SP |
SL |
SD |
SH |
SW |
BCL-D |
BCL-V |
SH-BC |
SW-BC |
E-L |
E-V |
AP |
NL |
SI |
AI |
ST |
STW |
SC |
SR |
|
NM L 646 |
Butovice |
Nilssoni |
P+BB |
30.9 |
18 |
9.8 |
9.8 |
|
|
|
|
9 |
15 |
|
42 |
4 |
|
|
|
? |
p |
|
NM L 647 |
Butovice |
Nilssoni |
P+B |
20.6 |
15 |
7.2 |
7.2 |
11 |
8.2 |
5.4 |
5.4 |
8 |
13 |
p |
|
8 |
|
nd |
nd |
? |
p |
|
NM L 13820 |
Lochkov |
Ludfordian |
P+BB |
33.1 |
30.4 |
11.3 |
11.3 |
|
|
|
|
10 |
8 |
|
|
|
|
|
|
? |
p |
|
NM L 13821 |
Vohrada |
Ludfordian |
P+BB |
37.0 |
17 |
10.0 |
|
|
|
10 |
|
|
11 |
|
|
|
|
|
|
? |
p |
|
NM L 13822 |
Butovice |
Nilssoni |
P+B |
37.9 |
17.2 |
|
11.2 |
|
15 |
|
11 |
|
9 |
p |
|
|
|
11 |
2.6 |
? |
n |
|
NM L 13823 |
Butovice |
Nilssoni |
P+B |
36.2 |
19.5 |
11.9 |
11.9 |
18 |
16 |
11 |
11 |
8 |
13 |
p |
|
17.5 |
|
12 |
2.1 |
p |
n |
|
NM L 13824 |
Butovice |
Nilssoni |
P+B |
51.9 |
13 |
11 |
10.8 |
19 |
|
|
|
7 |
8 |
p |
|
|
|
10 |
1.9 |
? |
n |
|
NM L 15381 |
Lochkov |
Ludfordian |
P+B |
59.9 |
13 |
11.9 |
|
20.6 |
|
9.2 |
|
5 |
|
p |
|
|
|
11 |
1.2 |
? |
n |
|
NM L 15382 |
Kozel |
Lundgreni |
P+B |
46.0 |
11.9 |
11.9 |
11.9 |
20.2 |
18.5 |
8.9 |
8.9 |
10 |
10 |
p |
|
7.2 |
|
12 |
1.8 |
p |
n |
|
NM L 15385 |
Butovice |
Nilssoni |
P+B |
42.0 |
11.4 |
9.8 |
9.8 |
17.1 |
15 |
8.9 |
8.9 |
8 |
6 |
p |
28 |
14 |
2 |
9.2 |
2.1 |
p |
p |
|
NM L 63557 |
Kozel |
Lundgreni |
P+BB |
30.9 |
8.9 |
7 |
7 |
|
|
7 |
7 |
10 |
|
|
|
|
|
|
|
? |
p |
|
NM L 63559 |
Butovice |
Nilssoni |
P+B |
37.3 |
13.0 |
11.6 |
11.2 |
20 |
18 |
10.9 |
10.1 |
7 |
9 |
p |
35 |
|
|
11 |
3.8 |
nd |
p |
|
NM L 63560 |
Lochkov |
Ludfordian |
P |
31.0 |
17 |
12 |
|
|
|
|
|
14 |
16 |
|
42 |
|
|
|
|
? |
n |
|
NM L 63562 |
Vyskočilka |
Lundgreni |
P+B |
44.9 |
|
10 |
9.9 |
|
|
8.80 |
8.8 |
10 |
16 |
|
42 |
16.5 |
|
|
|
? |
n |
|
NM L 63563 |
Butovice |
Nilssoni |
P+BB |
21.7 |
10.1 |
8.1 |
8.1 |
|
|
7 |
7 |
17 |
18 |
|
|
16 |
|
|
|
? |
n |
|
NM L 63564 |
Butovice |
Nilssoni |
P+B |
29.8 |
11.9 |
|
7.9 |
9.7 |
|
6.5 |
6 |
10 |
10 |
p |
|
11 |
|
nd |
nd |
nd |
n |
|
NM L 63566 |
Butovice |
Nilssoni |
P+BB |
32.0 |
15.0 |
8.6 |
8.5 |
|
|
8.6 |
8.5 |
|
10 |
|
|
17 |
|
|
|
nd |
n |
|
NM L 63567 |
Butovice |
Nilssoni |
P+B |
35.4 |
16.9 |
12.9 |
12.9 |
19.6 |
13.8 |
11.1 |
11.1 |
13 |
13 |
p |
34 |
|
|
nd |
nd |
nd |
n |
|
NM L 63568 |
Butovice |
Nilssoni |
P+B |
45.0 |
10.4 |
9.8 |
9.8 |
20 |
17 |
8.1 |
8 |
7 |
7 |
p |
45 |
|
4 |
9.7 |
2.5 |
? |
n |
|
NM L 63569 |
Konieprus |
Ludfordian |
P |
27.9 |
22.0 |
12 |
10.1 |
|
|
|
|
|
17 |
|
|
|
|
|
|
? |
n |
|
NM L 63570 |
Kosorz |
Ludfordian |
P |
32.2 |
17.0 |
10.5 |
10.5 |
|
|
|
|
14 |
13 |
|
|
4 |
|
|
|
nd |
n |
|
NM L 63571 |
Butovice |
Nilssoni |
P+B |
39.0 |
19 |
10.4 |
10.4 |
14.1 |
11.1 |
10.1 |
10.1 |
11 |
15 |
p |
|
18 |
5 |
10 |
0.9 |
? |
n |
|
NM L 63572 |
Butovice |
Nilssoni |
P |
31.9 |
9.3 |
9 |
9 |
|
|
9 |
9 |
12 |
16 |
|
38 |
14.3 |
|
|
|
? |
n |
|
NM L 63573 |
Butovice |
Nilssoni |
P |
22.6 |
12.2 |
8.2 |
8 |
|
|
|
|
|
14 |
|
|
|
|
|
|
? |
n |
|
NM L 63574 |
Kozel |
Lundgreni |
P+B |
30.0 |
12.0 |
12 |
12 |
21.2 |
19.9 |
9 |
9 |
|
|
p |
30 |
2 |
4 |
nd |
nd |
? |
n |
|
NM L 63575 |
Butovice |
Nilssoni |
B |
18.0 |
10.0 |
10 |
10 |
18 |
14 |
7.9 |
7.8 |
13 |
13 |
p |
|
21 |
10 |
nd |
nd |
? |
n |
|
NM L 63576 |
Butovice |
Nilssoni |
P+B |
26.8 |
10.2 |
10.9 |
10.9 |
|
|
10 |
10 |
13 |
13 |
|
42 |
10 |
|
|
|
nd |
n |
|
NM L 63577 |
Butovice |
Nilssoni |
P+BB |
25.8 |
17.5 |
7.1 |
7 |
|
|
7.1 |
7 |
14 |
15 |
|
52 |
|
|
|
|
nd |
n |
|
NM L 63578 |
Butovice |
Nilssoni |
P+B |
21.9 |
8.4 |
7.4 |
7.4 |
9.9 |
8 |
6.9 |
6.9 |
17 |
13 |
p |
|
16 |
2 |
nd |
nd |
nd |
n |
|
NM L 63579 |
Butovice |
Nilssoni |
P+BB |
25.0 |
|
8.4 |
8.2 |
|
|
|
|
14 |
10 |
|
43 |
|
|
|
|
? |
n |
|
NM L 63580 |
Butovice |
Nilssoni |
P |
16.0 |
7.6 |
5 |
3 |
|
|
|
|
15 |
15 |
|
40 |
|
|
|
|
? |
n |
|
NM L 63581 |
?Ischadites pit |
Lundgreni |
B |
32.0 |
16.2 |
16 |
|
31 |
27.9 |
11.70 |
11 |
16 |
13 |
p |
60 |
7.5 |
|
nd |
nd |
? |
n |
|
NM L 63582 |
Kozel |
Nilssoni |
B |
25.6 |
|
|
9.5 |
25.6 |
23 |
|
|
|
|
|
|
|
|
nd |
nd |
? |
n |
|
NM L 63583 |
Kozel |
Lundgreni |
P+B |
41.0 |
11.5 |
10.3 |
10.3 |
17.8 |
16.1 |
8.9 |
8.6 |
8 |
7.5 |
p |
|
|
7 |
9.9 |
1 |
? |
n |
|
NM L 63584 |
Butovice |
Nilssoni |
P+B |
26.1 |
10.2 |
9.9 |
9.8 |
16.9 |
11.3 |
7.6 |
7.6 |
9 |
8 |
p |
40 |
14 |
|
9.3 |
2 |
nd |
p |
|
NM L 63585 |
Butovice |
Nilssoni |
P+B |
37.9 |
14 |
12 |
11.1 |
24 |
|
9 |
8.4 |
9 |
10 |
p |
37 |
12.6 |
9 |
11 |
3.4 |
nd |
n |
|
NM L 63586 |
Butovice |
Nilssoni |
P |
28.1 |
8 |
7.1 |
7.1 |
|
|
|
|
12 |
14 |
|
46 |
|
|
|
|
? |
n |
|
NM L 63587 |
unknown |
Ludfordian |
P |
25.0 |
14 |
8.9 |
|
|
|
|
|
|
|
|
|
|
|
|
|
nd |
n |
|
Specimen |
Locality |
Age |
SP |
SL |
SD |
SH |
SW |
BCL-D |
BCL-V |
SH-BC |
SW-BC |
E-L |
E-V |
AP |
NĽ |
SI |
AI |
ST |
STW |
SC |
SR |
|
NM L 63588 |
unknown |
Ludfordian |
P |
25.0 |
22 |
10 |
10 |
|
|
|
|
11 |
11 |
|
45 |
|
|
|
|
nd |
n |
|
NM L 63589 |
unknown |
Ludfordian |
P+BB |
29.1 |
13.6 |
10.8 |
9.8 |
|
|
|
|
16 |
15 |
|
39 |
|
|
|
|
? |
n |
|
NM L 63591 |
?Kozel |
Lundgreni |
P+B |
42.6 |
11.1 |
11.1 |
10.9 |
|
17.2 |
9.2 |
8.8 |
9 |
9 |
p |
31 |
|
|
11 |
1.7 |
? |
p |
|
NM L 63592 |
Kozel |
Nilssoni |
P |
21.1 |
8.4 |
7.3 |
7.3 |
|
|
|
|
15 |
13 |
|
|
|
|
|
|
? |
n |
|
MCZ 132450 |
Butovice |
Nilssoni |
P+B |
33 |
30.1 |
12.8 |
12.8 |
17.1 |
15 |
10.9 |
10.9 |
10 |
7 |
p |
46 |
11 |
|
12 |
2.8 |
nd |
n |
|
MCZ 162299 |
Bubovice |
Lundgreni |
P+B |
31.9 |
9.6 |
9.4 |
9.2 |
13.3 |
12.4 |
7 |
6.8 |
10 |
10 |
p |
29 |
16 |
0 |
nd |
nd |
nd |
p |
|
CGS SM 197 |
Butovice |
Nilssoni |
P+B |
34.1 |
9.8 |
7.9 |
7.7 |
13.3 |
12 |
7.1 |
6.9 |
11 |
9 |
p |
39 |
|
|
nd |
nd |
? |
n |
|
CGS SM 450 |
Lištice 759, bn. 2A |
Lundgreni |
B |
14.2 |
|
8 |
7 |
13 |
12.1 |
6.1 |
5.9 |
|
9 |
p |
34 |
|
|
nd |
nd |
? |
n |
|
CGS SM 454 |
Butovice |
Nilssoni |
P |
18.9 |
20.3 |
|
6.5 |
|
|
6.2 |
|
17 |
17 |
|
37 |
|
|
|
|
? |
n |
|
CGS SM 455 |
Lištice 759, bn. 2A |
Lundgreni |
B |
32.0 |
|
15.7 |
15.6 |
30.2 |
26.1 |
10.9 |
|
12 |
10 |
p |
|
13 |
8 |
16 |
2.6 |
? |
p |
|
CGS SM 461 |
Lištice 759, bn. 2A |
Lundgreni |
B |
25.0 |
|
14.7 |
13.1 |
24.2 |
|
|
10.4 |
|
8 |
p |
|
|
|
|
|
? |
n |
|
CGS SM 462 |
Lištice 759, bn. 2A |
Lundgreni |
B |
26.4 |
|
14.3 |
|
26.2 |
25.1 |
11 |
10.9 |
9 |
8 |
p |
|
5 |
|
|
|
? |
n |
|
CGS SM 463 |
Lištice 759, bn. 2A |
Lundgreni |
P |
25.0 |
|
11.1 |
11.1 |
|
|
|
|
9 |
|
|
24 |
7 |
|
|
|
nd |
n |
|
CGS SM 464 |
Bubovice |
Nilssoni |
P+B |
31.0 |
12.2 |
11.5 |
11 |
|
|
|
|
|
|
|
|
|
|
un |
un |
p |
n |
The classification of shell repairs, minor, moderate, massive, is adopted from Bond and Sounders (1989). Minor damages span one to five growth lines (depending on spacing), but extend for only a short distance laterally. The repair is recorded in only a few growth lines immediately succeeding the injury. Moderate damages penetrate as much as 15% of the aperture. Such damages were probably somewhat traumatic, in that they would have required localized dedication of the mantle to generate repair, but the activity of the organism was probably not otherwise affected. Massive damages probably represent traumatic events to the living organism, because a substantial portion of the animal’s soft parts would have been exposed (for details see Bond and Sounders 1989: 418, 419). The proportion of preserved shell wall is expressed in six categories (0–5), where 0 represents the internal mould and 5 the fully preserved shell. The intermediate four categories include shell wall preserved to one-quarter (1, 25%), one-half (2, 50%), or three-quarters (3, 75%) of the preserved surface; category 4 includes individuals with shell preserved between three-quarters and fully preserved shell wall.
We recognize here four basic categories of shell preservation: body chamber + phragmocone, isolated phragmocone, isolated body chamber, phragmocone + base of the body chamber. Complete shell consists of a phragmocone with tip of apex and body chamber with preserved aperture. The number of specimens in each category indicates the degree of shell fragmentation. For each specimen, the preservation of aperture is indicated. The preserved aperture indicates that the specimen died in this growth stage. This is an implication for mortality in palaeopopopulations, i.e., proportionally high number of juvenile specimens with preserved aperture indicates high mortality of juveniles.
Graptolite biozone chart is used as defined by Štorch (2023).
Systematic palaeontology
Subclass Orthoceratoidea Teichert, 1967
Order Orthoceratida Kuhn, 1940
Family Dawsonoceratidae Flower, 1962
Genus Lepidoceras nov.
Zoobank LSID: urn:lsid:zoobank.org:act:47BD78E7-F127-48E5-8B91-152D0A55557A.
Type species: Cyrtoceras lepidum Barrande, 1866, Silurian of Bohemia; monotypic.
Etymology: The genus name is derived from the species name of the type species, lepidum meaning pleasant in Latin.
Diagnosis.—As for the type species, by monotypy.
Remarks.—The shell morphology of Lepidoceras corresponds to diagnostic features of the family Dawsonoceratidae as emended by Kröger and Isakar (2006). The presence of annulation is one of the typical characters of the Dawsonoceratidae. However, intraspecific variation exists in the type species of the genotype Dawsonoceras annulatum (Sowerby, 1818). In addition, annulation may be well developed, suppressed or eventually entirely missing in the evolutionarily younger representatives (Dawsonoceras obsoletum Barrande, 1874, in Horný 1956) from the Prague Basin (Barrande 1874; Horný 1956; Manda and Turek 2015, 2024). The annulation in Lepidoceras is usually developed in a short segment of juvenile shells but an indication of annulation may also occur in later ontogenetic stages. The changing distance of growth lines in the early shell of Lepidoceras and occasionally also in later stages resembles a similar pattern observed in the annulated shell of dawsonoceratids (Manda and Turek 2024). A reduction/suppression of annulation is a newly acquired character of Lepidoceras.
Dzik (1984) suggested a phyletic link between Cyrtoceras lepidum Barrande, 1866 (here the type species of Lepidoceras gen. nov.) and the pseudorthoceratid Lyecoceras Mutvei, 1957, but the comparison of the muscle scars and embryonic shells in the species of Lyecoceras (Mutvei 1957) and L. lepidum rules such a link out.
Lepidoceras differs from the very similar genus Serpaglioceras Gnoli & Serventi, 2008 (middle Silurian of the peri-Gondwana, Carnic Alps) in having a thinner siphuncle with less expanded connecting rings, short septal necks, and poorly developed annulation. Another similar orthoceratid, the Silurian Metaspyroceras Foerste, 1932 (Laurentia, northeast USA), differs from Lepidoceras in having well-developed annulation running obliquely to the sutures and a slightly constricted aperture. Many species were assigned to the genus Metaspyroceras in the past, but none of them have the similar shell shape and narrowing of the apertural part as the type species; the genus Metaspyroceras is probably monospecific. All three of these genera represent specialized cephalopods with small shells and limited geographic distribution that diverged from the ancestral lineage of dawsonoceratids.
Unlike Lepidoceras, the shell of Calorthoceras Chen in Chen et al., 1981, is long and slowly expanding, usually straight in the post-juvenile stages. The number of radial ribs increases as the shell grows. The shape of the embryonic shell of Lepidoceras corresponds to that of Calorthoceras (Ristedt 1968; Manda and Turek 2024). Both taxa share non-constricted, slightly curved, blunt, endogastric or conical embryonic shell. Fine longitudinal lirae appear already in the embryonic stage and pass into more pronounced ridges in the juvenile stage. Lepidoceras and Calorthoceras share well developed longitudinal ridges, the number of which markedly varies. In both genera, the reticulate sculpture is also variable, i.e., the distance of growth lines changes, forming growth sequences with densely and sparsely packed growth lines. In both genera, longitudinal ridges are occasionally intercalated with fine longitudinal lirae. Lepidoceras probably diverged from Calorthoceras.
The shell apex of Lepidoceras resembles the apical part of Kionoceras Hyatt, 1884, from which it is distinguished by the thinner siphuncle, the smooth non-accretion initial shell and the constant number of undifferentiated radial ridges. The shell cross section in Lepidoceras is never polygonal and the annular elevation is narrow. Both genera are endogastric, and in Kionoceras there are also specimens with a variably curved shell, especially in the apical part (Barrande 1868). The early stages of Kionoceras and Lepidoceras probably competitively filled the same niche as the juvenile stages of nautilids (Manda and Turek 2019). Lepidoceras and Kionoceras co-occur at all localities of the upper Wenlock and lower Ludlow strata, but Kionoceras is more widespread in this interval and its record is more continuous, which indicates Lepidoceras might have been a specialist.
Stratigraphic and geographic range.—Upper Wenlock (Homerian)–upper Ludlow (Ludfordian) of Prague Basin (Bohemia).
Lepidoceras lepidum (Barrande, 1866)
Figs. 2–9.
1866 Cyrtoceras lepidum Barr.; Barrande 1866: pl. 157: 31–34, pl. 168: 1–11.
1866 Cyrtoceras acies Barr.; Barrande 1866: pl. 168: 12–15.
1866 Cyrtoceras fragile Barr.; Barrande 1866: pl. 168: 16–21.
1867 Cyrtoceras lepidum Barr.; Barrande 1867: 637, 638.
1867 Cyrtoceras fragile Barr.; Barrande 1867: 636.
1867 Cyrtoceras acies Barr.; Barrande 1867: 473.
1868 Orthoceras mimus Barr.; Barrande 1868: pl. 279: 9–16.
1874 Orthoceras mimus Barr.; Barrande 1874: 202, 203.
1984 Cyrtoceras lepidum Barrande; Dzik 1984: 125, 127, fig. 49.
Type material: Lectotype of Cyrtoceras lepidum Barrande, 1866, designated herein: NM L 646, Butovitz (Barrande 1866: pl. 157: 31–33; refigured here as Fig. 5A). Paralectotypes: NM L 647, Butovitz (Barrande 1866: pl. 157: 34); NM L 13820, Lochkov (Barrande 1866: pl. 168: 1–3; refigured here as Fig. 2F); NM L 13821, Vohrada (Barrande 1866: pl. 157: 4–5); NM L 13822, Butovitz (Barrande 1866: pl. 157: 6–9); NM L 13823, Butovitz (Barrande 1866: pl. 157: 9, 10). Types of O. mimus Barrande, 1868: NM L 15384, Kozel (Barrande 1868: pl. 279: 15, 16; refigured here as Fig. 3B), NM L 15381, Lochkov (Barrande 1868: pl. 279: 9, 10); NM L 15382, Kozel (Barrande 1868: pl. 279: 11; refigured here as Fig. 3A); NM L 15383, Kozel (Barrande 1868: pl. 279: 12; refigured here as Fig. 3C); NM L 15384, Kozel (Barrande 1868: pl. 279: 13). All from Silurian of the Prague Basin, Czech Republic.
Type locality: Czech Republic, Central Bohemia, Praha-Butovice, Na břekvici (Butovitz).
Type horizon: Middle Nilssoni Zone, Gorstian, Ludlow, Kopanina Formation.
Material.—Seventy-four specimens (including type material): NM L 646, 647, 13820–13824, 15381–15385, 63557, 63559–63589, 63591, 63592, 63652, 63655, 63657; MCZ 11009, 132450, 162299; CGS SM 197, 446–466, all from upper Wenlock (Homerian)–upper Ludlow (Ludfordian) of Central Bohemia, Czech Republic (for localities see chapter Stratigraphic and geographic range).
Four categories of preservation are recognised. Relatively complete shells with broken apex represent 48% of the specimens (n = 29). Isolated phragmocones make up 23% (N = 22) of the studied shells. Phragmocones with preserved base of the body chamber and isolated body chambers are less frequent, 17% (n = 9) and 13% (n = 6) of all specimens, respectively (Fig. 1). The aperture is preserved in more than half of the studied shells. The shell wall is usually well preserved, nearly half of the studied shells retained more than half of the shell wall with visible sculpture.
Emended diagnosis.—Orthoceratid with variously curved endogastric shell, cross section circular or slightly compressed. Apex gradually expanding without constriction, longitudinal lirae appear already on the embryonic shell. Body chamber short. Sculpture variable, consists of a combination of longitudinal lirae/ridges and growth lines/ridges; the distance of longitudinal elements is higher than that of transversal elements. Annulation either not developed, or slightly indicated or restricted to the early juvenile stage. Siphuncle subcentral, thin, tubular, shifted ventrally, septal necks cyrtochoanitic. Colour pattern irregular with prevailing undulated, transverse bands.
Description.—The shell is endogastric, very slightly to strongly curved (Fig. 2); the ratio of shell diameter/shell height is 8–30.4 (median 13, n = 42). The shell is more curved dorsally than ventrally. The shell expansion is variable, usually decreases with shell growth. The angle of expansion (laterally) ranges from 5° to 17° (median 10°). The ventral angle of expansion varies from 6° to 18° (median 11°). The angle of expansion is the same laterally and dorso-ventrally in less than one third of the specimens (n = 13); the number of shells expanding more rapidly dorso-ventrally or laterally is equal; the maximum difference in lateral and ventral expansions in the same specimen is 6°. The ratio of the shell height at the aperture and the shell height at the body chamber base ranges from 1 to 1.4 (median 1.2, n = 32).

Fig. 2. General morphology of the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Motol (D) and Kopanina (A–C, E, F) formations. A. NM L 63589, locality unknown (SW part of Praha), Ludfordian, Ludlow, dorsal (A1, see reduced longitudinal lines dorsally), ventral (A2) and lateral (A3) views. B. NM L 63588, locality unknown (SW part of Praha), Ludfordian, Ludlow, lateral view (see Fig. 10C). C. NM L 63585, Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, dorsal (C1) and lateral (C2) views. D. NM L 63591, Kozel (i.e., Lištice 759), Lundgreni Zone, Homerian, Wenlock, lateral (D1), dorsal (D2), and dorsolateral (D3) views, two healed repairs h1, h2, arrows indicate the position of repairs on the shell edge. E. MCZ 11009 (see also Fig. 7B), Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, lateral view, a detail showing wrinkle structure on internal mould (E1) and two massive healed repairs close to the aperture (E2). F. Most curved specimen, NM L 13820, paratype (Barrande 1866: pl. 168: 1–3), Praha-Lochkov, Ludfordian, Ludlow, apical-dorsal (F1) and lateral (F2) view. All specimens coated with ammonium chloride. Scale bars 5 mm.
The shell wall is thin. It consists of the wrinkle layer (Fig. 2E1), nacreous layer and delicate outer prismatic layer; the maximum shell thickness is 0.3 mm. The wrinkle structures are very delicate, anastomosing, with about 12 ridges per millimetre; the wrinkles are perpendicular to the shell axis and oblique to the suture (Fig. 3E1). The outer shell layer is commonly exfoliated giving the shell a smooth look (e.g., Fig. 5B2). The shell cross section is circular or very slightly compressed, Barrande’s (1866) form Cyrtoceras acies (Barrande, 1866).
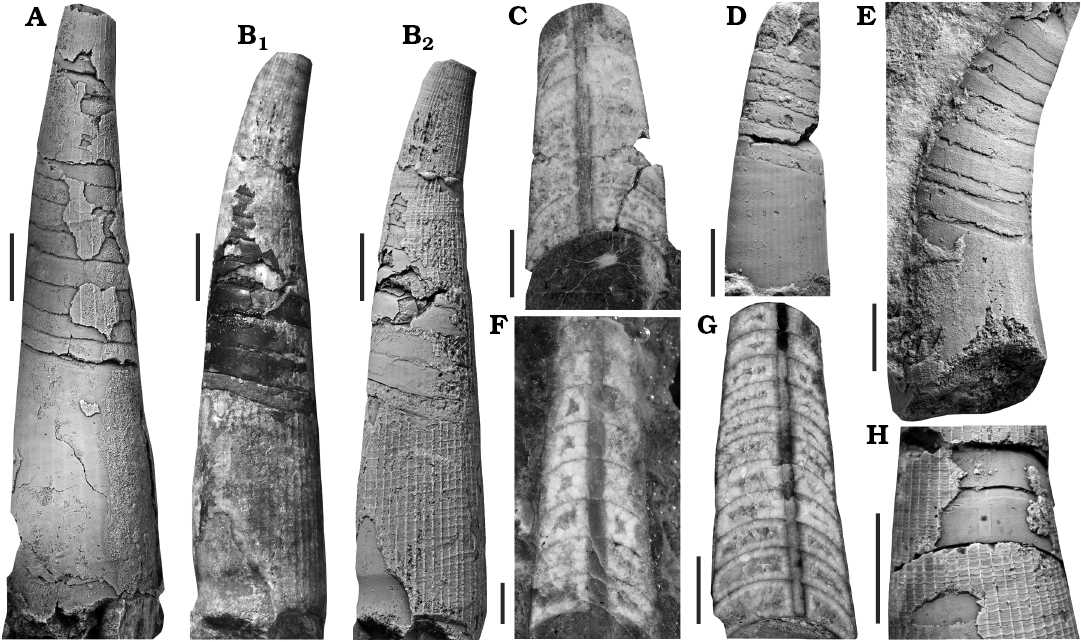
Fig. 3. Orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Motol (A, C, D, F) and Kopanina (B, E, G, H) formations. A. NM L 15382, paralectotype (Orthoceras mimus, Barrande 1868, pl. 279: 11), Kozel (i.e., Lištice 759), Lundgreni Zone, Homerian, Wenlock, lateral view. B. NM L 15385, type (Orthoceras mimus Barrande, 1868: pl. 279: 15, 16), Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, lateral view (uncoated and coated with ammonium chloride). C. NM L 15383, type (Orthoceras mimus, Barrande 1868: pl. 279: 12), Kozel (i.e., Lištice 759), Lundgreni Zone, Homerian, Wenlock, lateral median section, left side. D. CGS SM 452, Lištice 759 (bed no 2a), Lundgreni Zone, Homerian, Wenlock, lateral median section, right side. E. NM L 63578, Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, lateral view. F. CGS SM 455, Lištice 759 bed no 2a), Lundgreni Zone, Homerian, Wenlock, lateral median section, right side. G. CGS SM 464, Praha-Butovice, Na břekvici (bed no 10), Nilssoni Zone, Gorstian, Ludlow, lateral view. H. NM L 63584, Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, dorsal view. All specimens coated with ammonium chloride, except for B1, C, F, G. Scale bars 5 mm, except F 1 mm.
The siphuncle is circular in cross section, thin, subcentral and shifts towards the venter during ontogeny (Fig. 3C, F, G). The ratio between the diameter of the siphuncle and the shell height and is about 0.1. The thin-walled connecting rings are very slightly convex, more vaulted dorsally. The septal necks are cyrtochoanitic. Intrasiphonal deposits are unknown. Phragmocone chambers are short; the chamber length is three to five times lower than the respective shell height. The last chamber may be shortened. The chamber length in the early growth stage is relatively longer than in the later growth stages. Cameral deposits are unknown. Septa are moderately concave with the maximum depth sub-centrally, slightly shifted towards the dorsum. The mural part of the septum is short. The dorsal furrow on convex shell side is straight, formed by two parallel lines, in apex the furrow is very narrow, later the furrow width is about 1/13 of the shell width. The ventral furrow was not observed. Sutures are straight, oblique, with an angle of inclination between 2° and 21°, the median is 13° (angle of inclination increases with increasing curvature). A dorsal shallow lobe can be indicated in the early stage (Fig. 6A).
The body chamber is short. The ratio between dorsal and ventral body chamber length ranges from 1 to 1.5 (median 1.1 which is also the most common value found in 13 specimens; n = 22). The ratio between the body chamber length (dorsally) and the shell height is 1.4–2 (median 1.7, n = 25). The length of the body chamber increases linearly during ontogeny. The shell wall thickening is parallel to the aperture. The aperture is oblique, inclined dorsally with an angle of 2–11°. The hyponomic sinus, if present, is wide and very shallow. The annular elevation of the orthoceratomorph type is situated around the base of the body chamber (Fig. 4). Ventrally, the elevation is narrow and its width slightly fluctuates. The width of the elevation increases dorsally and reaches its maximum mid-dorsally; in one specimen, the width increases mid-ventrally (Fig. 5E). A shallow lobe may be developed mid-dorsally in the adoral band of the elevation. The adapical margin of the elevation is slightly bent towards the aperture at the contact with the dorsal furrow, or perhaps in one case towards the apex. The maximum width of the elevation is ca. 1.1 mm. The onset of the widening of the elevation varies from dorsolateral to ventrolateral. The widening of the elevation is either abrupt, or gradual towards the middle of the dorsal side. The elevation bears a pair of retractor muscle impressions separated in the middle of the dorsal side by a narrow crest. The retractor scars disappear between the dorsolateral and ventrolateral side.
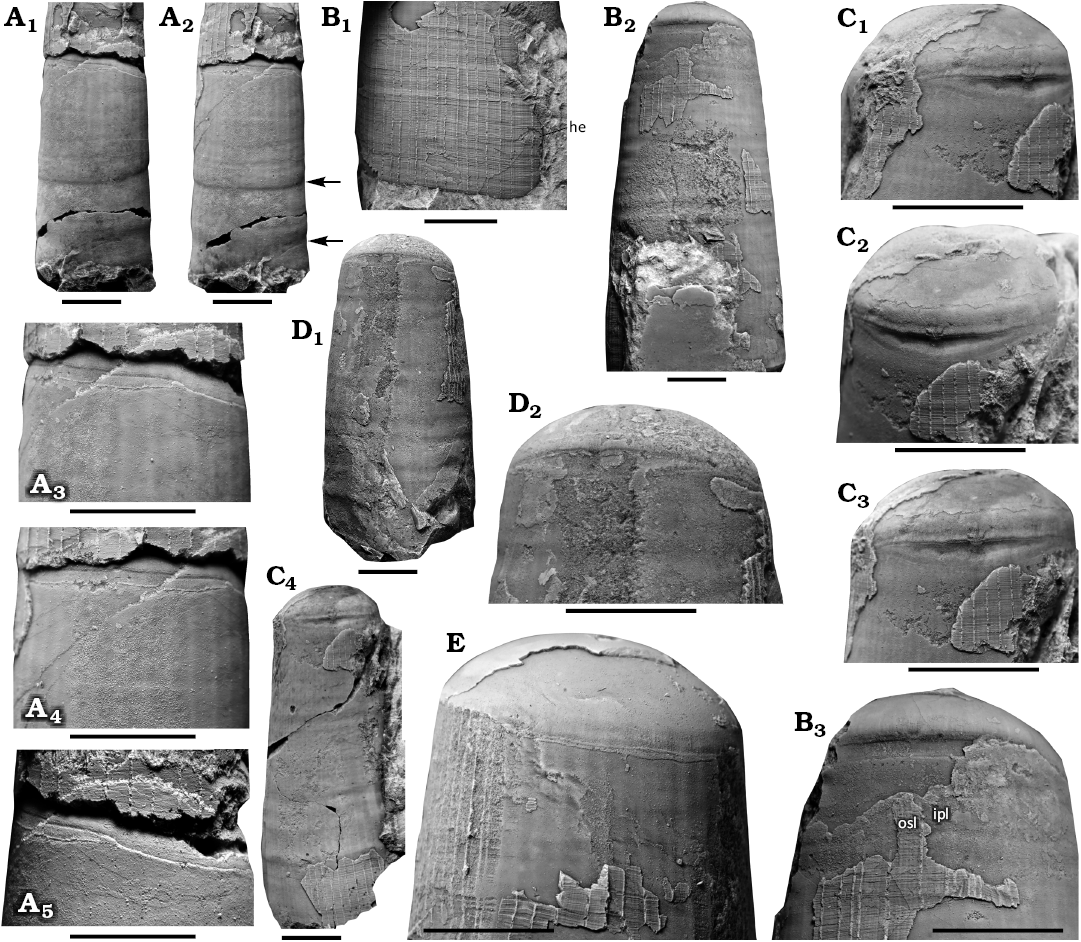
Fig. 4. Muscle scars of the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Motol Formation; Kozel (A, C) and Lištice 759, bed no 2a (B, D, E), Lundgreni Zone, Homerian, Wenlock. A. NM L 63583, lateral (A1) and dorsal (A2) views, detail of muscle scars laterally (A3), dorsolaterally (A4) and ventrolaterally (A5); arrows indicate two shell wall thickenings. B. CGS SM 463, dorsal view (B2), detail of muscle scars (B3) and ventrolateral detail of aperture (B1) showing hyponomic sinus; osl (outer shell layer), ipl (inner shell layer). C. NM L 63582, detail of muscle scars, dorsolateral (C1) and dorsal (C2, C3). D. CGS SM 458, dorsal view (D1) and detail of muscle scars (D2). E. CGS SM 461, ventrolateral detail of muscle scars. All specimens coated with ammonium chloride. Scale bars 5 mm.
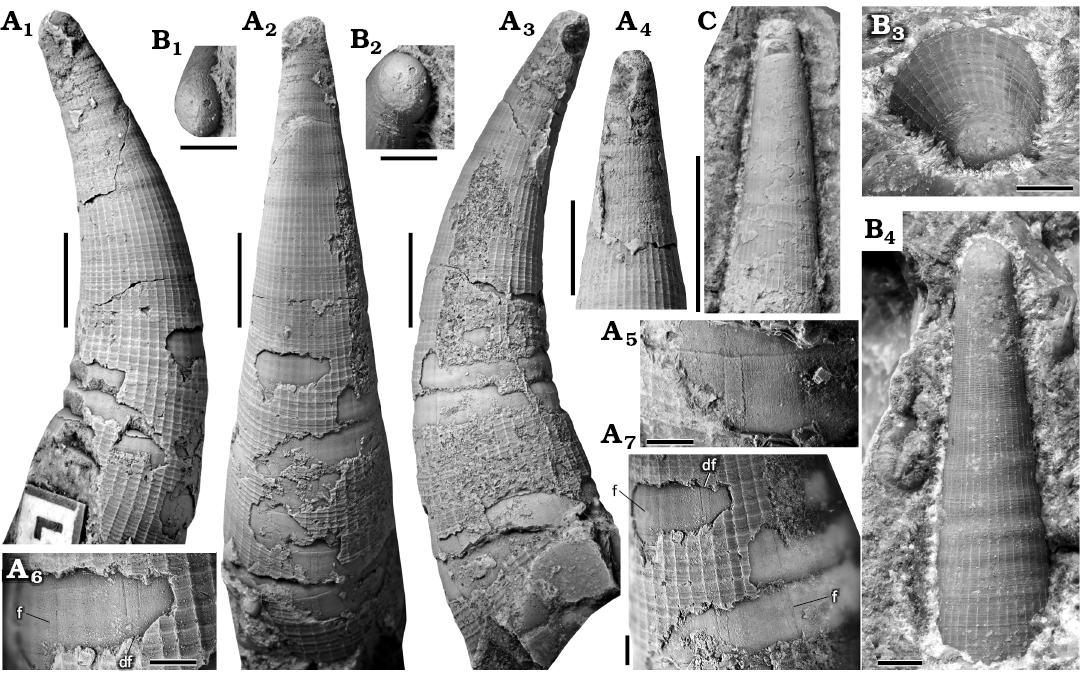
Fig. 5. Juvenile shell of the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Motol (D) and Kopanina (A–C) formations. A. NM L 646, lectotype (Barrande 1868, pl. 157: 31–34), left lateral (A1), dorsal (A2), right lateral (A3), ventral (A4) view, details (A5–A7) of the dorsal furrows (df) and two parallel furrows (f), Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow. B. NM L 63657 (see Fig. 8B), apical (slightly inclined towards the dorsal side B1, ventral side B2 and lateral side B3) and lateral view (B4), Tachlovice, Prostřední mlýn, section 713, Nilssoni Zone. Gorstian, Ludlow, C. CGS SM 466 (see Fig. 7E), lateral view, Praha-Butovice, Na břekvici (bed no 10), Nilssoni Zone, Gorstian, Ludlow, lateral view. All specimens coated with ammonium chloride. Scale bars 5 mm, except A5–A7, B, 1 mm.
The reticulate sculpture is highly variable. It consists of a combination of longitudinal lirae/ridges and the usually more densely spaced and less distinct transversal growth lines and ridges. Their crossing is straight and perpendicular, straight and oblique, or undulating (U-shaped growth ridges intercalated between longitudinal elements). The number of longitudinal ridges around the entire circumference of the shell varies between 24 and 60 (n = 29, Q1: 34, median: 39, Q3: 42.5). Their number is retained during the whole shell growth. Sometimes one or several finer longitudinal ridges are intercalated between the more distinct ridges. The longitudinal ridges can be suppressed or disappear dorsally in the subadult-adult shell, e.g., in some Ludfordian specimens (Fig. 2F) of Cyrtoceras fragile Barrande, 1866. The density and prominence of growth ridges varies markedly. During growth, the shell portions with more or less distinct growth structures are sometimes present. Low, transversal growth walls or annuli can be developed in any post-juvenile growth stage of the shell. Typically, four growth walls in the shell portion close to the apex are developed.
The smooth initial portion of the shell at the tip of the apex is slightly elliptical, dorsoventrally elongated with the maximum length of about 1 mm (Figs. 5, 6). There is a relief fold at the margin of the initial shell and growth lines and fine longitudinal ridges appear concentrically around the smooth initial shell area. The apical part of the shell is conical or slightly curved. The dimensions and morphological changes are summarised in Table 2. The shape of the shell apex and sculpture varies within a small range. A short part of the shell with four, more or less elaborate growth walls is usually present in the apical part of the shell; in one specimen the walls are missing (Fig. 6C).

Fig. 6. Juvenile shell of the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Kozel (i.e., Lištice 759), Lundgreni Zone, Homerian, Wenlock, Motol Formation (A, C) and Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, Kopanina Formation (B, D, E). A. CGS SM450, dorsal view of a juvenile body chamber, longitudinal ridges already expressed on the internal mould. B. MCZ 11009, lateral view (B1) and detail of the apex (B2). C. NM L 63557, lateral view (C1) and detail of the apex (C2), see also Fig. 3E. D. NM L 63655, lateral view. E. CGS SM 466, lateral view (see Fig 6B). All specimens coated with ammonium chloride. Scale bars 5 mm, except B2, C2, 1 mm.

Fig. 7. Shell pathology in the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Praha-Butovice, Na břekvici (A) and Prostřední mlýn, section 713 (B), Nilssoni Zone, Gorstian, Ludlow, Kopanina Formation. A. CGS SM 197, right lateral (A1, A5), ventral (A2), left lateral (A3, A6), and dorsal (A4, A7) views, complete specimens and marked details. Abbreviations: a, aperture; ar, anastomosing ridges close to small pits; csd, changing shell growth direction at the aperture; fll, zone with several fine longitudinal lirae of second order intercalated between two adjacent first order lines; ill, irregular undulated growth of longitudinal lirae; is, irregular sculpture appearance; mt, maximum shell thickness; p, pit; sp, small pits; ss, anomalous smooth sculpture; swi1, slightly increased thickness of shell wall; swi2, rapidly increased thickness of the shell wall; wli, wrinkle layer indication in pathologic shell; wd, disappearance of wrinkle layer and traces of longitudinal ridges on internal mould. B. NM L 63657, detail of the shell apex with irregular growth lines indicated by arrows, dorsal side, appearance of growth wall corresponds to hatching (h) (see Fig. 6B). All specimens coated with ammonium chloride. Scale bars 5 mm.
Table 2. Dimensions (in mm) and morphology of a juvenile of Lepidoceras lepidum. Abbreviations: AE, angle of expansion; DI, diameter of initial shell laterally; SD, shell diameter; SH, shell height, SH1: at SL 2 mm, SH2: at SL 4 mm, SH3: at SL 6 mm; SL, shell length.
|
Specimen |
SL |
SH |
SD |
DI |
AE |
SH1 |
SH2 |
SH3 |
Sudden changes in morphology |
|
MCZ 11009 Butovice |
44 |
6.4 |
14.6 |
1.07 |
6° |
1.7 |
1.77 |
2.07 |
1. appearance of growth lines and longitudinal lirae; SH: 1.34; SL: 0.44 2. strengthening of growth lines, number of lines per millimetre decreases from 28 to 13; SH: 1.76; SL: 2.89 3. appearance of growth walls; SH: 1.78; SL: 4.16 (four walls) 4. disappearance of growth walls; SH: 2.07; SL: 6.38 |
|
NM L 63655 Butovice |
26.63 |
6.25 |
8.25 |
≤1.6 |
10.8° |
≥1.94 |
≥2.38 |
≥2.86 |
1. appearance of growth walls; SH: 2.38; SL: ≤3.38 (four walls) 2. disappearance of growth walls, strengthening of growth lines; SH: 3.13; SL: 6.75 |
|
NM L 646 Butovice |
30.9 |
15 |
18 |
≤2 |
13° |
≥3.07 |
≥3.49 |
≥4.13 |
1. appearance of growth walls; SH: 3.2; SL: ≤4.8 (four faint growth walls) 2. increasing shell expansion (ventr), strengthening of growth lines; SH: 4.47; SL: ≤8.93 3. disappearance of growth walls, appearance of hyponomic sinus; SH: 4.8; SL: ≤10.27 |
|
NM L 63657 Tachlovice |
9.9 |
2.62 |
2.62 |
1.06 |
10° |
1.51 |
1.89 |
2.28 |
1. appearance of growth walls, strengthening of growth lines, decreasing spacing of growth lines; SH: 2.4; SL: 5 (four walls) 2. disappearance of growth walls; SH: 2.54; SL: 7.48 |
|
NM L 63557 Kozel |
30.9 |
7 |
8.9 |
1.31 |
22° |
2.03 |
2.63 |
2.86 |
1. appearance of longitudinal lirae ventrally; SH: 0.18; SL: 1.25 2. appearance of irregular growth lines dorsally; SH: 1.9; SL: 1.37 3. appearance of regular growth lines; SH: 2.26; SL: 2.3 4. decreasing expansion (to 11°), appearance of longitudinal lirae all around the shell, fide of growth lines; SH: 2.34; SL: 2.68 5. strengthening of growth lines; SH: 2.68; SL: 4.9 |
|
CGS SM 461 Butovice |
22.8 |
6 |
13 |
≥1 |
10.9 |
1.5 |
1.9 |
2.4 |
1. appearance of faint growth walls; SH: 2.2; SL: 5 (four walls) 2. disappearance of growth walls, a break in shell curve, strengthening of growth sculpture; SH: 2.6; SL: 7.2 |
The shell colouration has been observed in six specimens, where it is present over the entire surface in all ontogenetic stages. The colour pattern is irregular, bilaterally asymmetrical and different in each specimen. The proportion of pigmented and non-pigmented shell is roughly equal. Irregular and poorly distinguishable spots are seen on one juvenile shell (Fig. 10E). The post-juvenile part of the shell shows irregular transverse colour bands of variable width. Changes in band width are more pronounced in weakly curved shells (Fig. 9B, D, E) than in more curved shells with a more regular colour pattern (Fig. 9A, C). Transverse bands are occasionally connected by longitudinal or transverse pigmented zones. In one case, the dorsal strips are joined to form a broad longitudinal stripe flanked laterally by a longitudinal narrow, nonpigmented zone (Fig. 9C1).
Remarks.—The vast majority of the studied specimens come from two localities in Praha-Butovice: Na břekvici section 584 and Beroun-Lištice section 579, where the species occurs in a thin part of the section consisting of a few beds. The shell preservation (no encrustation and corrosion, very fine preservation of delicate growth structures) indicates a rather rapid deposition at both sites. The relatively high intraspecific variation of the species thus rather reflects a variability of palaeopopulations than an effect of condensation in the sedimentary record.
Barrande (1866–1874) recognised four species, which are all synonymised herein. Orthoceras mimus and Cyrtoceras lepidum differ in the shell form. The former species possesses a slightly curved shell and the latter one a more curved shell. However, there is continuous transition in the shell form between both co-occurring taxa. Cyrtoceras acies differs from the co-occurring C. mimus in having a slightly laterally compressed shell in the post-juvenile growth stage. However, this only represents the extreme case of subcircular cross section visible in several studied specimens (Table 1). Cyrtoceras fragile differs from C. lepidum by the suppression and eventual disappearance of longitudinal ridges on the dorsal side of the adult growth stage. This feature is slightly developed in more specimens from the upper Ludlow and is considered an intraspecific variation of evolutionarily younger populations.
Stratigraphic and geographic range.—Most of the studied specimens come from historical collections; the localities where the presence of the species has been recently confirmed are indicated below by an asterisk.
Motol Formation, Wenlock, Homerian, Lundgreni Biozone: Praha-Řeporyje, Arethusina Gorge, NW slope, section 687*, lower part of the biozone (a bed of cephalopod limestone embedded in shale, Kříž et al. 1993), SE slope of the gorge, upper part of the biozone (limestone beds with Aulacopleura, after Prantl 1936). Barrande’s locality Bubovitz (Bubovice, exact site unknown, skeletal limestone, Havlíček 1995). Beroun-Lištice, U cestičky, section 759*, Barrande’s locality Kozel (skeletal limestones; Kříž 1992; Manda 1996). Beroun-Lištice, Herinky, Barrande’s pit*, limestone with Plicocyrtia Community and overlaying tuffites (Havlíček 1995). Barrande’s locality Lodenitz (Loděnice, Špičatý vrch-Černidla, Ischadites pit, skeletal limestone, Kříž 1992). Praha-Malá Chuchle, Vyskočilka locality (Wiskocilka in Barrande, limestone with juvenile molluscs; Vodička and Manda 2019, interval of beds 11 to 19). Tachlovice, Prostřední mlýn, section 713 (Barrande’s locality Tachlowitz, skeletal muddy limestone, Kříž et al. 1993).
Lower part of the Kopanina Formation, Gorstian, Ludlow, middle Nilssoni Biozone: Praha-Butovice, Na břekvici section 584*, beds 10, 11 (cephalopod limestone, Kříž et al. 1993, Barrande’s locality Butovitz).
Upper part of the Kopanina Formation (the cephalopod limestone biofacies, see Kříž 1998), Ludfordian, Ludlow: historical collection material come from Barrande’s (1865–1877) localities Dlauha hora (Beroun, Dlouhá hora), Konieprus* (Koněprusy), Kosorz (Kosoř), Hinter Kopanina (Praha-Zadní Kopanina), Lochkow (Praha-Lochkov), and Wohrada (Praha-Malá Ohrada); Inexpectatus inexpectatus Biozone: Koněprusy, road-cut near Velký vrch (light coloured cephalopod limestone).

Fig. 8. Healed shell repairs in the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Kozel, Lundgreni Zone, Homerian, Wenlock, Motol Formation (C, D) and Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, Kopanina Formation (A, B, E, F). A. NM L 63584, ventrolateral view. B. NM L 63559, dorsal view (B3) and details of a shell repair around the shell circumference in right lateral (B1), left lateral (B2), and ventral (B4) views and of several repairs close to the aperture (B5). C. NM L 63592, dorsal (C1), lateral (C2), and ventral (C3) views; rr (reduction of longitudinal ridges after massive shell injury), he1, 2 (two healed repairs). D. NM L 63557, lateral (D1) and dorsal (D2) views. E. NM L 63586, dorsal view. F. NM L 63577, ventrolateral view, arrow indicates a small injury. All specimens coated with ammonium chloride. Scale bars 5 mm, except B3, 10 mm.

Fig. 9. Shell coloration in the orthoceratid cephalopod Lepidoceras lepidum (Barrande, 1866), Silurian of the Prague Basin (Czech Republic, Central Bohemia), Motol Formation (E) and Kopanina Formation (A–D). A. NM L 63587, locality unknown (SW part of Praha), Ludfordian, Ludlow, dorsal (A1), lateral (A2), and ventrolateral (A3) views. B. NM L 63586, Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, lateral (B1), dorsal (B2), and ventral (B3) views. C. NM L 63588 (see Fig. 3B), locality unknown (SW part of Praha), Ludfordian, Ludlow, dorsal (C1), lateral (C2), and ventral (C3) views. D. NM L 63652, Praha-Butovice, Na břekvici, Nilssoni Zone, Gorstian, Ludlow, lateral view. E. NM L 63557 (see Fig. 7C), Kozel (i.e., Lištice 759), Lundgreni Zone, Homerian, Wenlock, dorsolateral (E1), lateral (E2), and ventrolateral (E3) views. Scale bars 5 mm.
Results and discussion
Evolutionary path of shell form.—Most orthoceratids, including the stratigraphically oldest species (Evans 2007), have a straight shell and a tubular siphuncle, and are considered to have been pelagic cephalopods (Kröger et al. 2009). Demersal orthoceratids have also evolved and often possessed an annulated shell (Marek 1971; Holland 1984; Manda and Turek 2015). Orthoceratids with a slightly curved shell are relatively common but more strongly curved shells have evolved only rarely (e.g., Dzik 1984; Kröger and Isakar 2006). The best-known case is the Silurian demersal orthoceratid Cyrtocycloceras Foerste, 1936, with an annulated, moderately curved shell (Marek 1971). Lepidoceras has a stratigraphic range of about seven million years. During that time, populations consisting of specimens with a slightly curved and finally strongly curved shell (Figs. 2, 10) developed. The body chamber/phragmocone ratio is a prominent factor in the shell hydrostatic properties (Petermann et al. 2019). The mode of preservation (fragmentation) does not allow an accurate measurement of the ratio between the length of the phragmocone and the length of the living chamber. However, considering the best-preserved shells, the ratio is about 1.5. The actual length of the phragmocone was probably greater in more curved shells than in slightly curved shells. Unlike the shape of the shell, proportions of the living chamber show very little variance (see ratios shell height at the aperture/shell height at the base of the body chamber, dorsal body chamber length/ventral body chamber length, shell height at the aperture/body chamber length; Table 1, Fig. 11).
Although there has been an increase in the shell curvature of L. lepidum throughout Ludlow, there are still specimens with a less curved shell. The question is whether this is a phenotypic (response to the environment) or genetic issue. The Silurian species of Cyrtocycloceras from the Prague Basin always have a very similar shell shape, but in this case the shape of the shell determines the swimming pattern, i.e., it allows rocking movement of the shell (Marek 1971). Cyrtocycloceras ibex (Sowerby in Murchison, 1839), on the other hand, is characterized by a variable shell curvature (Holland 2007). Similarly, in the related species Dawsonocerina dulce (Barrande, 1868) from the Ludlow/Přídolí boundary interval in the Prague Basin, the shell is curved in variable ways (Barrande 1868). For example, the material from about 40 cm thick set of cephalopod limestone beds, Praha-Lochkov, Nad ubikacemi section, contains specimens with straight to moderately curved shells (ŠM unpublished data). Lepidoceras exhibits highest variation in shell curving among orthoceratids.
Specimens of Lepidoceras lepidum with the most curved shell are convergent in shell form with co-occurring oncocerids with slowly expanding curved shells. This is a case of orthoceratids entering the morphospace usually occupied by demersal oncocerids. The limited geographic dispersion of Lepidoceras lepidum, along with its locally high abundance in a specific community and its absence in oxygen depleted facies with pelagic fauna also corresponds with the distribution pattern of demersal cephalopods.
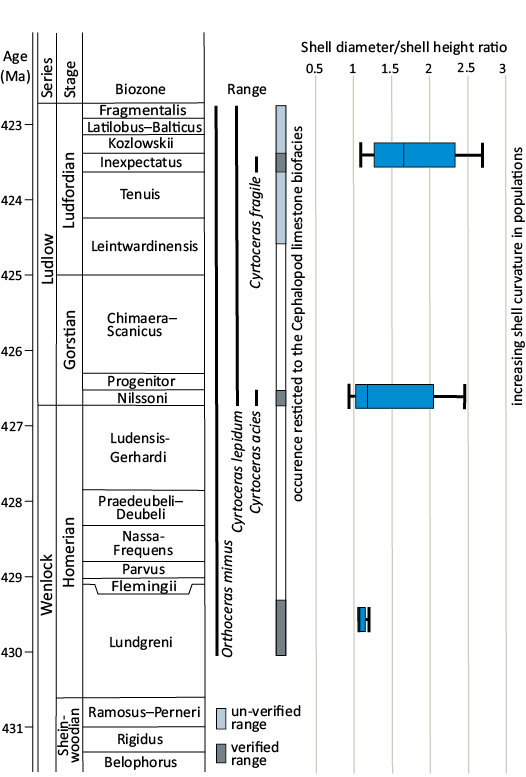
Fig. 10. Range of Lepidoceras lepidum (Barrande, 1866) and former Barrande’s (1866, 1867) taxa in the Silurian of the Prague Basin and shell diameter/shell height ratio.
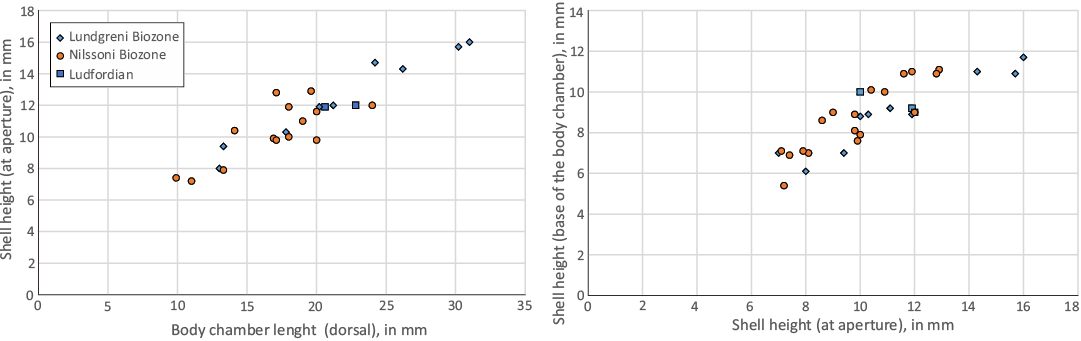
Fig. 11. Ratio of dorsal body chamber length/shell height and shell height/shell height at the base of the body chamber in Lepidoceras lepidum.
Apex, non-accretion initial shell.—The cicatrix region (cicatrix sensu Erben and Flajs 1975), well known in Nautilus, represents the initial part of the shell and is formed by secondary calcification of an initial organic plate/disc. Thus, this region does not show growth structures but there is a central bar and grove/s and a relief fold on the margin of cicatrix region (e.g., Arnold et al. 1987; Mutvei and Doguzhaeva 1997). Cicatrix region of similar type is known to have developed in actinocerids (Doguzhaeva et al. 1999; Kröger and Mapes 2007) and pseudorthoceratids where it is one of the diagnostic characters (e.g., Niko et al. 2020; King and Evans 2019). In the present concept, orthoceratids are distinguished from pseudorthoceratids by the absence of cicatrix and radial divisions of annulosiphonate deposits. Somewhat lost in the present discussion is the fact that Ristedt (1971) also described cicatrix in species that are currently classified as orthoceratids (see also Erben and Flajs 1975). Ristedt (1971: 168) described apex of Silurian Merocycloceras declivis Ristedt, 1968 “with a large smooth cicatrix area on initial calotte clearly delimited by marginal furrow and growth lines”. Ristedt (1971) distinguished several other types of apex tips and non-accretion initial shells without growth lines in orthoceratids (“Schalenanfang” or “Initialkalotte”). For bactritids, Doghuzayeva (2002) used the term primordial dome. However, the smooth apex of the initial chamber encircled by the growth lines, the so-called “bald spot”, is also known in Devonian ammonoids (for summary see Klofak et al. 1999). In orthoceratids, the margin of the smooth area may or may not coincide with the relief fold. Unlike the nautilids, however, the orthocerids do not have a central bar. We refer to this non-accretion portion of the shell by the term non-accretion initial shell.
The very tip (initial portion of the shell) of the apex in Lepidoceras lepidum is very slightly convex, dorsoventrally elongated, and smooth without any growth structures (Figs. 5B, 6C). The size of the non-accretion initial shell is variable with a dorsoventral diameter between one and two millimetres (Table 2). Length of the initial shell of Lepidoceras lepidum is moderately higher than in closely related Calorthoceras butovitzense Chen in Chen et al., 1981 (Manda and Turek 2024). The non-accretion initial shell of the apex is poorly known in orthoceratids probably due to the very small size of the apex and the smooth surface of the juvenile shell in many species. The variable size of the non-accretion initial shell in orthoceratids is consistent with the size variation of cicatrix region in pseudorthoceratids (cf. Blind 1988; Kröger and Mapes 2004) and nautilids.
Early shell form variability.—The understanding of the rate of evolution and intraspecific variation of juvenile orthoceratids is limited by the fact that most juvenile orthoceratids cannot be assigned to species based on specimens in adult/subadult growth stage. In Lepidoceras lepidum, juvenile individuals can be unambiguously identified by specific sculpture. The five juvenile specimens available (Table 2; Figs. 5, 6) suggest high variation in shell shape, size and sculpture. The shape and overall dimensions of the embryonic shell in Lepidoceras lepidum are not uniform. The embryonic shell is conical and either straight, or slightly endogastrically curved. The annulation on the early juvenile shell is not always developed, and the size of the annuli also varies. Similar shape variability of the embryonic shell is documented (Manda and Turek 2024) in the closely related Silurian orthoceratid Calorthoceras Chen in Chen et al., 1981. Generally, the intraspecific variability in embryonic shells of Silurian orthoceratids is poorly known, and there are species for which only moderate variability in the shape and size has been documented (e.g., Serpagli and Gnoli 1977). Kolebaba (1973) described forty forms of orthoceratid embryonic chambers from a very thin bed at a single locality (Praha-Pankrác, upper Lundgreni Zone, lower Homerian, Wenlock), a number far exceeding the number of orthoceratid species known from upper Wenlock of the Prague Basin (ŠM unpublished data). This also suggests a high intraspecific variation in the early shell formation rather than a higher diversity than currently known and detectable by subadult and adult shells only. The virtual absence of transitional forms between straight conical and curved embryonic shells and between annulated and non-annulated juvenile shells might indicate embryonic shell polymorphism in Lepidoceras lepidum. However, the data are still too limited at this point to further evaluate this possibility.
Hatching.—On the Lepidoceras lepidum shell, hatching is presumably manifested by a change in sculpture, most commonly by the appearance of annulation (Figs. 5, 6B, D; Table 2). However, the change in sculpture may be very subtle (Fig. 6C).
The shell of a freshly hatched individual of Lepidoceras lepidum reached a height (i.e., dorsoventral diameter) of about 2.5 mm and a length between 4–5 mm. Cephalopods with the embryonic shell/egg size of Lepidoceras lepidum have been suggested to be planktonic or nektobenthic (e.g., De Baets et al. 2012). Juvenile orthoceratids have so far been considered planktonic (Hewitt 1988; Kröger et al. 2009). In Lepidoceras, the shell shape is conical with a relatively large angle of expansion and in one case even resembles the shape of the juvenile shells of oncocerids. Pelagic orthoceratids, on the other hand, have shells with a lower angle of expansion (e.g., Ristedt 1968). The complete juvenile shells of Lepidoceras lepidum with preserved body chamber, which document juvenile mortality, are usually found together with adults. In one case, a juvenile shell comes from a locality from which no other Lepidoceras lepidum specimen is known. No juvenile Lepidoceras lepidum specimens have been found in association with juvenile orthoceratids in oxygen-depleted facies. The demersal mode of life of Lepidoceras lepidum juveniles would also correspond to the geographically limited occurrence of the genus so far known only from the Prague Basin. In summary, all indications are that the juveniles were nektobenthic, similar to adults. The shell of the early juvenile stage is usually characterized by four growth walls; a hyponomic sinus develops shortly after the disappearance of the last growth wall.
The juvenile shell of Lepidoceras lepidum is one of the largest among orthoceratids and probably the largest among post-Ordovician orthoceratids (Ristedt 1971; Aubrechtová et al. 2020). The size of the embryonic shell, i.e., egg and hatchling, of Lepidoceras lepidum moderately increased in comparison with the type species of Dawsonoceras (Kröger and Isakar 2006) and Calorthoceras, the assumed ancestor of Lepidoceras (Manda and Turek 2024). Thus, in Lepidoceras lepidum, there is an increase in egg size and a simultaneous decrease in the shell size, including the length of the living chamber, compared to its relatives. This implies a reduction in fertility and an increase in incubation time to a rate comparable to some K-strategy oncocerids. Thus, a correlation between adult shell size and egg size cannot be considered for orthoceratids. Lepidoceras lepidum is another indication that the evolution of reproductive strategies in cephalopods is rather complex (see Rocha et al. 2001; Pohle et al. 2020).
Mature modification, adult shell size range, and mortality.—Lepidoceras lepidum exhibits three morphological features considered to be mature modifications (Fig. 12): septal approximation, apertural shell thickening (Klug et al. 2015) and eventually the presence of muscle scars (Davis et al. 1996). Septal crowding and shell wall thickening might also result from pathologies (e.g., Hoffmann and Keupp 2015), but there is no indication of such an origin in the material studied here.
Septal crowding was observed in three specimens (Figs. 3A–C, 13). However, it is not present in three specimens with shell thickening. In one case, a shortened phragmocone chamber is followed by a chamber of standard length (Fig. 3E). Documentation of septal approximation in orthoceratids is very limited (e.g., Kröger and Landing 2007; the Middle Ordovician Gangshanoceras Zou, 1988). Septal crowding probably occurred in only some individuals and may have been followed by at least one non-shortened phragmocone chamber. Septal crowding itself is therefore not sufficient to identify the adult growth stage (see Beck et al. 2021).
Shell wall thickening at the aperture was found in shells with a width between 9.2 and 15.6 mm (median 11 mm, n = 16, Table 1). The thickening was found immediately at the apertural edge or at distances of up to 4.2 mm from the apertural margin (Figs. 2D, 3A, B, 12). The width of the thickening varies between 0.9 to 3.8 mm (median 2.1 mm). In one specimen, two wall thickenings were observed in close proximity (Fig. 4A).
There is no general agreement on the preservation of muscle scars being an indicator of maturity, particularly in nautilids (Davis et al. 1996; Klug et al. 2015). Muscle attachment is known in pre-adult stages in oncocerids and discosorids (Barrande 1865–1877; Manda 2008; Manda and Turek 2009a). Muscle scars were observed in five specimens of Lepidoceras lepidum, four of which also showed the shell wall thickening (apertures of the other are broken off). The scars were observed only in adult specimens. Muscle scars preservation linked with a fully-grown specimen was observed also in the orthoceratid Cyrtocycloceras semitectum (Barrande, 1866) (Marek 1971). There is no known case of retractor muscle imprints in immature orthoceratids. The presence of muscle impressions may therefore indicate maturity in orthoceratids.
Shell wall thickening and septal crowding are not terminal adult modifications but show a degree of flexibility. If they really represent mature modifications and indicate maturity, the difference between the shell height of the smallest mature and the largest mature shell is six millimetres (maximum measured shell height is 15.7 mm). Environmental-related variation in adult shell size is well known in multiceratoids (e.g., Silurian Pentameroceras and Phragmoceras; Barrande 1865, Stridsberg 1985, Manda 2008) but has not yet been investigated in orthoceratoids.
Of the thirty shells with preserved aperture (Table 1), nearly all have a shell height above 6 mm. More than half of the specimens fall in the shell height interval 9.6–12.8 mm. Only one shell belongs to a juvenile. Sixteen shells show an adult modification. Overall, this implies relatively low mortality in the populations (Fig. 12). Given the preservation of the shells, significant taphonomic bias can be ruled out.
The shell height (Fig. 1) of the mature shells with preserved aperture varies between 10–16 mm, which indicates the size variability of the fully-grown shell (Fig. 12). Intraspecific variation in shell size is well known in oncocerids (e.g., Pentameroceras Hyatt, 1884; Stridsberg 1985) and discosorids (e.g., Phragmoceras Broderip in Murchison, 1839; Barrande 1865) with an adult modification (constricted aperture). The smallest specimens can be three times smaller than the largest. Lepidoceras is the first orthoceratid with documented variance in the fully-grown shell size (Fig. 12).
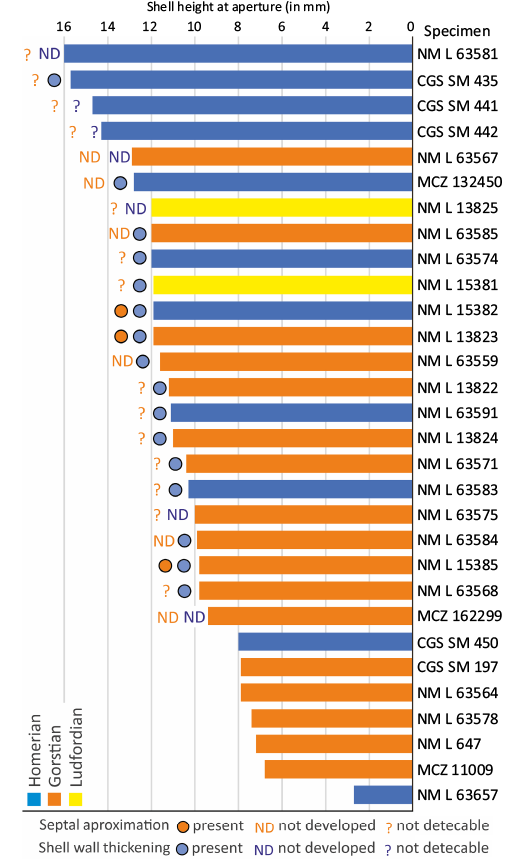
Fig. 12. Mature modifications in Lepidoceras lepidum, all specimens with preserved aperture included.
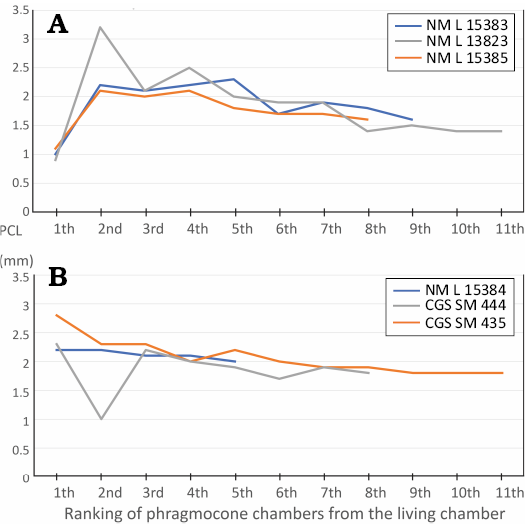
Fig. 13. Distance of septa calculated from last chamber in specimens of Lepidoceras lepidum with narrowing of the last chamber (A) and in specimens without narrowing of the chamber but with thickening of the shell at the aperture (B).
Muscle attachment.—In orthoceratids, the annular elevation is developed around the base of the body chamber. It consists of an unpaired (or paired), low anterior lobe (or lobes) on the dorsal side corresponding to unpaired (or paired) retractor imprints (Mutvei 1957, 2002; Marek 1971; Kröger et al. 2005). The development of muscle attachments during ontogeny is unknown and intraspecific variability of muscle scars is poorly known.
Mutvei (1957: text-fig. 7e; 1964: figs. 3c, 6a–c; 2002) pointed out that the retractor muscles in orthoceratids did not form a roof over the ventral mantle cavity as in nautilids and tarphyceratids. The retractor contractions could therefore not have been used to expel water from the mantle cavity to allow jet-powered swimming. Mutvei (2002) also suggested that the area of the orthoceratoid retractor muscle scars relative to the size of the living chamber is often smaller than in Nautilus and tarphyceratids. However, the studies of Mutvei (1957, 2002) concern muscle scars in straight orthoceratids with long body chambers. The living chamber of Lepidoceras lepidum is relatively short but the annular elevation corresponds to that of orthoceratids with long body chambers. The body chamber of Lepidoceras lepidum is conical or slightly curved, its dorsal side is considerably longer that the ventral (oblique, dorsally expanded aperture, inclined septa). The retractor muscles are thus longer than the mantle cavity. The shape of the body chamber and its position in life produced an aperture that was oriented obliquely towards the shorter ventral side. The dorsal retractor muscles were thus relatively close to the ventral side of the mantle cavity. Consequently, the contraction of the retractor muscle expelled water from the mantle cavity more effectively than in the case of straight orthoceratids with a long body chamber. The swimming ability was probably slightly improved in the curved endogastric shell compared to the slowly expanding straight shell. This may correspond to increasing of the shell curvature in L. lepidum. The species also exhibits well developed and relatively broad dorsal furrow, which according to Kröger et al. (2005) represents the most important zone for the attachment of the palliovisceral ligament.
Until now it has been assumed that retractor imprints on the annular elevation are established in a shape that does not change in adults. However, Lepidoceras shows an extension of the retractor scars. Retractors on the annular elevation were initially located in the middle of the dorsal side (about a quarter of the circumference of the base of the living chamber) and gradually extended laterally to more than half the circumference of the shell (Fig. 4). The muscle impressions are only known in adults. The growth slowed down upon maturation and the soft body probably remained longer in the same body chamber and became more firmly attached, which resulted in the expansion of the muscle attachment site. The gradual enlargement and lateral extension of the retractors certainly led to an increase in the propulsion efficiency.
We do not question the planktic life style of orthoceratids with a straight shell and a long living chamber (Mutvei 2002; Kröger et al. 2005). Lepidoceras lepidum is an example of an orthoceratid in which a change in shell morphology led to an adaptation to a demersal mode of life. The Silurian species of Cyrtocycloceras are other examples of a demersal orthoceratids (Marek 1971; Holland 1984) whose muscle scars do not differ in position and development from those of Lepidoceras lepidum. However, the well-developed hyponomic sinus of Cyrtocycloceras clearly demonstrates a relatively efficient jet propulsion.
Mid-dorsal furrow.—The mid-dorsal furrow is developed on the convex side of the shell of L. lepidum and along with the shallow hyponomic sinus on the opposite side demonstrates the endogastric character of the shell (Fig. 3H). Like in other orthoceratids, the mid-dorsal furrow is formed by an interrupted, paired, shallow groove on the internal mould. The furrow width relatively increases slightly during ontogeny and forms an elliptical depression on the inner surface of the shell (second type of preservation of Flower 1939: fig. 2b; Fig. 8E).
In cephalopods, the dorsal furrow originates from the inner part of the calcified shell apex, in line with the inner dorsal termination of the central linear depression of the cicatrix (Schindewolf 1933). The dorsal furrow in orthoceratids originates in the same position in the initial chamber (Ristedt 1971; Kolebaba 1973) and therefore synchronously with the ventral furrow appearing on the opposite side of apex (Chirat and Boletzky 2003; Kröger et al. 2005). In one specimen, morphological structures resembling the dorsal furrow are also developed dorsolaterally on both sides of the dorsal furrow in a symmetrical position (Fig. 5A). One groove is developed on the right side and two closely adjacent grooves are present on the left side. This demonstrates that the conditions leading to the formation of the dorsal furrow may have occasionally partially occurred in a symmetrical position at other locations on the margin of the developing initial shell. A parallel morphological structure corresponding to the ventral furrow has also been described in a Silurian oncocerid (Manda and Turek 2009a: fig. 7i).
Shell colouration.—Shell colouration was observed in six out of the 69 specimens retaining the shell wall and is recorded from the entire stratigraphic range of the species (Fig. 9). The colouration in embryonic shells is poorly preserved but in juvenile and adult shells, it is well recorded. This is one of the best documented shell colourations in the Orthoceratoidea.
The colour pattern described in the Ordovician, Silurian and Early Devonian orthoceratoids consists of regular longitudinal bands (Kobluk and Mapes 1989; Kröger et al. 2009; Turek and Manda 2012; Manda and Turek 2015; Kröger and Aubrechtová 2019). Regular chevron pattern appears in the Middle Devonian orthoceratids/pseudorthoceratids and is documented also from the Carboniferous (Kobluk and Mapes 1989). Shell colouration in the form of regular transverse bands is known in a single Carboniferous, flattened, straight specimen (Foerste 1930). Irregular transverse bands are described in a straight shell fragment from the earliest Devonian of the Prague Basin (Turek 2009). However, the taxonomic position of the two latter specimens is uncertain (?Orthoceratida, Pseudorthoceratida, Actinoceratida, Astroviida). The irregular, transversely oblique colour bands in Lepidoceras lepidum thus represent the first record of this type of colour pattern in the Orthoceratoidea. The colour pattern of L. lepidum corresponds to the shell colouration of some morphologically similar Silurian oncocerids (Turek and Manda 2020; Fig. 14). The moderately irregular transverse colour bands have been observed in the most curved adult shells of L. lepidum, as well as in some oncocerids (a more strongly curved shell generally exhibits more regular colour bands that a less curved shell; Turek and Manda 2020). The most irregular colouration is present in slightly curved shells and the embryonic and juvenile shell of L. lepidum, which is also a feature observed in some moderately expanded and curved Silurian oncocerids (Turek and Manda 2020). The demersal orthoceratid Lepidoceras acquired the shell colouration characteristic of demersal multiceratoids as a result of adaptive convergence. This is the second case of convergence of shell colouration and shell form between orthoceratoids and multiceratoids (Fig. 14). The first case is a Silurian oncocerid with a slowly expanding and moderately curved shell exhibiting longitudinal bands like orthoceratoids (Manda and Turek 2009b).
The course of the colour bands, their width and shape are highly variable in L. lepidum, as in some Silurian oncocerids with a slightly curved shell. The variability of the colour pattern in L. lepidum is exceptional among Ordovician and Silurian orthoceratoids, which show a low variability of colouration restricted to the number of longitudinal bands and their width. However, the number of orthoceratoids with known colour pattern is limited so far.
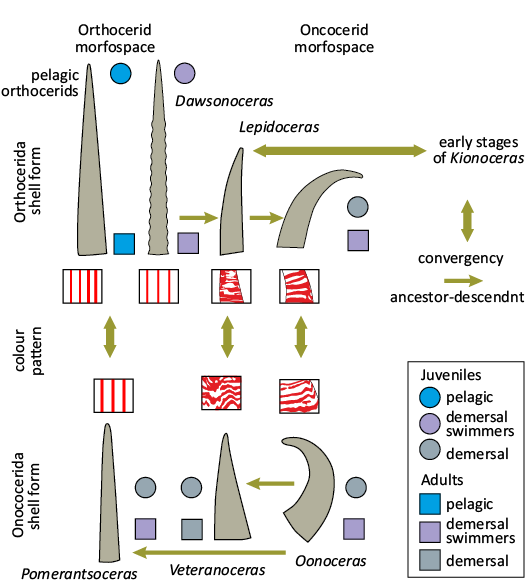
Fig. 14. Convergences in coloration and shell shape between Silurian orthoceratids, oncoceratids, and pseudorthoceratids.
Variability in longitudinal sculpture.—Longitudinal sculpture appeared in numerous orthoceratids. The density of longitudinal sculptural elements is often used as a diagnostic character at the genus level, even when only a small number of specimens is available.
Longitudinal ridges were calculated in 29 specimens of Lepidoceras lepidum. The number of these elements does not change during ontogeny including embryonic stage. In one case, two longitudinal ridges were lost after a massive, repaired shell damage (Fig. 8C1). The number of longitudinal ridges varies from 24 to 60 (Fig. 15, Q1 = 34, median = 39, Q3 = 42.5) and shows a unimodal and slightly skewed right distribution, when plotted on a histogram (Fig. 15). Thus, there is a preference for a relatively lower number of longitudinal ridges rather than a higher number. The greatest range in the number of longitudinal lirae/ridges is shown by the upper Wenlock specimens. The average number of longitudinal lirae/ridges increases slightly in the Ludlow compared to the Wenlock. The variability in the number of ribs is smallest in the evolutionarily youngest specimens (Ludfordian, late Ludlow), and the number of ribs corresponds to the average number in preceding populations. The width and height of the longitudinal lirae/ridges is not related to the number of lirae/ridges, so individuals with fewer ridges do not have these elements more pronounced. No correlation was found between the number of longitudinal lirae/ridges and the origin of the respective specimens from shallower or deeper water environments (limestone facies). In summary, the above findings indicate that the number of the longitudinal sculptural elements cannot be considered a reliable diagnostic feature. A variable number of longitudinal lirae/ridges has also been documented in the Silurian Calorthoceras, where it has been used as a character diagnostic for several species (Chen et al. 1981). However, in the latter genus, the differences in the number of longitudinal lirae/ridges clearly represent intraspecific variation (Manda and Turek 2024).
The appearance of distinct sculpture has been attributed to shell strength improvement, anti-predatory adaptation, shell hydrodynamics refinement, and camouflage (Chamberlain 1993; Barskov 2018; Jaitly et al. 2022). Regardless of its adaptive function, there wasn’t a strong selection pressure for a particular number of longitudinal lirae/ridges in Lepidoceras lepidum.
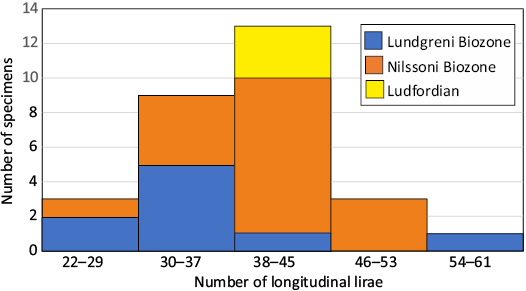
Fig. 15. Number of specimens of Lepidoceras lepidum in categories by number of longitudinal lirae.
Healed shell damages.—A healed shell damage was found in 18 of 74 individuals (23%, Fig. 16, Table 3). Nine specimens had more than one injury. Minor damages were found in ten individuals, moderate in seven and massive in six.
Three types of shell repair were recognised. The first type (Figs. 2E, 8B) is characterised by an interruption of the shell secretion associated with a displacement of longitudinal ridges (number of observed repairs, n = 6). The interruption may be accompanied by a gentle constriction of the shell. The second type (Fig. 8C, E) represents a symmetrical, more or less deep, U-shaped breakage of the shell (n = 19). The third type (Fig. 2F) represents a broad, crested breakout of the shell followed by irregular growth lines gradually filling the breakout (n = 9). The combined shell repair (Figs. 2E, 8C, D) is represented by a combination of the first type with the second or third type (n = 3). One specimen exhibits a specific combined shell repair around the shell circumference manifested as transverse narrow break in the shell secretion with two U-shaped repairs located dorsally and ventrally (Fig. 8C). Most healed shells exhibit a single shell damage, number of specimens displaying two, three, or four damages decrease; the exception being the apertural region of one adult specimen, which shows seven damages (Fig. 8E). Multiple massive repairs on the same specimen were not observed.
The distribution of shell damages on the shell surface is difficult interpret. A limited number of shell repairs occur on the dorsal side, while minor damages are most common on the ventral and ventrolateral sides. The types of healed damages found in L. lepidum are consistent with those found in other Silurian cephalopods from the Prague Basin and early Paleozoic cephalopods in general (Kröger 2004, 2011; Klug 2007). However, combined shell damages with deeper, narrow breaks on the opposite sides of the shell are rare in the Silurian of the Prague Basin and, if they occur at all, are mainly documented in Přídolí.
Table 3. Quantification of shells with a healed repair in relation to the shell wall preservation.
|
Shell wall preservation category |
0 |
1 |
2 |
3 |
4 |
5 |
|
Number of specimens |
3 |
15 |
20 |
23 |
8 |
4 |
|
Number of specimens with an injury |
0 |
3 |
3 |
8 |
3 |
0 |
|
Specimens with a repair in % |
0 |
20 |
15 |
35 |
38 |
0 |
|
Total number of repairs |
0 |
4 |
5 |
17 |
9 |
0 |
The number of damages in relation to the shell height varies. Most damages occur in the 6.4–9.6 mm height interval, while moderate and massive damages are rare as the shell height increases (Fig. 16, Table 3). One specimen (Fig. 6B) possessing extensive healed juvenile damages defines the limits of survival after an extensive shell breakage. This indicates that the maximum tolerated shell loss is about 25%, similar to the modern-day Nautilus (Arnold 1985). This is higher than has been previously reported (Kröger 2004) and indicates a high level of survivability even in juveniles. Kröger (2011) concluded that specimens of the Ordovician Anthoceras with conch diameters above 30 mm had a considerably lower risk of predation than smaller shells. Yomogida and Wani (2012) suggested a higher risk of fatality by predatory attacks in earlier ontogenetic stages of modern Nautilus. Although the shell of Lepidoceras lepidum is small, there is a higher frequency of sublethal damages in early ontogenetic stages, and the most serious damages occur in juveniles. The distinct sculpture makes it possible to identify L. lepidum even from shell fragments, which are, however, very rare. Thus, there is no evidence for massive crushing of shell of Lepidoceras lepidum in the studied section.
Quantification of healed damages is limited by the preservation of the shell wall (Fig. 16, Tables 1, 3). The number of specimens with a damage in the categories 3–5 with more than half of the shell wall preserved is 32% (n = 35), and the categories 0–2 with less than half of the shell wall preserved it is 15% (n = 38). The number of specimens with a shell damage repair in category 3 and 4 is 35% (n = 23) and 38% (n = 8), respectively. Four individuals with a well-preserved shell over the entire surface show no repairs. The total number of individuals with damages was certainly higher than preserved, but there is no indication that it was significantly higher. This is particularly valid for the large damages that can be recognized on the shell relics and often on internal moulds. Given the lack of quantitative data on healed damages in early Paleozoic cephalopods, it is difficult to compare predation rates. However, the proportion of individuals with damages roughly corresponds to that of Ordovician endocerids from the Baltic (Kröger 2011).
A more detailed study of the frequency of damages in different cephalopod groups is prevented by a lack of data, but it is a promising area of research. However, the study of the cephalopod collections from Lištice and Butovice (Na břekvici), i.e., localities with Lepidoceras lepidum, indicates that a higher number of damages are observed in demersal cephalopods, whereas healed damages are very rare in pelagic and nektonic cephalopods (e.g., Sphooceras; Turek and Manda 2012). No damage has been found on the small shells of the genus Kionoceras (i.e., corresponding in size to Lepidoceras), which are morphologically similar to Lepidoceras at sites where they occur together. However, Kionoceras appears to have been a better swimmer which is consistent with its significantly wider geographical distribution. The frequency of healed damages may serve as an indirect evidence of the lifestyle of Silurian cephalopods.
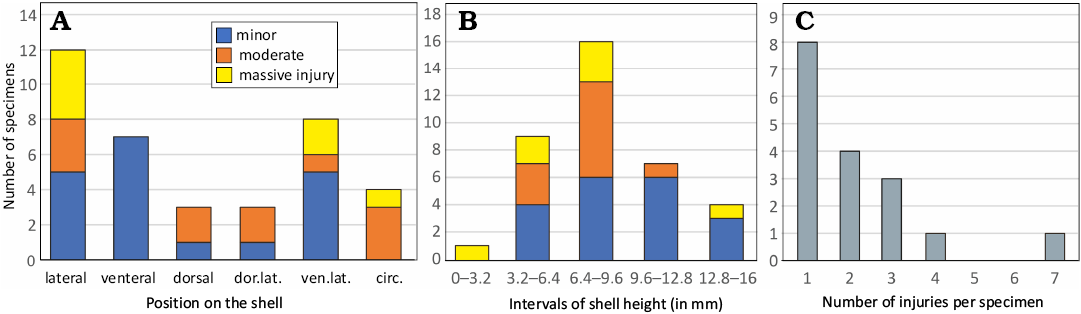
Fig. 16. Shell repairs in Lepidoceras lepidum related to the position on the shell (A), shell height (B), and number of specimens with one to seven repairs on the shell (C). Abbreviation: circ., circumference; dor.lat, dorsolateral; ven.lat, ventrolateral.
Shell pathologies.—Two specimens with a pathological shell interpreted as a result of parasitic infestation were recognised. One immature shell (Fig. 3D) shows a pathological septum (fourth from the aperture) with the suture slightly curved laterally towards the apex. The sutures of the following septa, which run diagonally to the axis of the shell, are straight. There is a circular depression on the internal mould of the phragmocone chamber following the anomalous septum, which may correspond to the pathological pit. A similar case of anomalous septa associated with pits has been described in an oncocerid coming from the same locality (Manda and Turek 2009b) and also in a discosorid from Upper Devonian of Russia (Mironenko 2018). It was considered to be the result of parasite infestation (De Baets et al. 2021). The anomalous course of septa has not yet been described in orthoceratids.
CGS SM 187 (for dimensions see Table 1) is a fully-grown specimen with pathologic development of apertural part of the body chamber showing irregular shell wall thickening with pits (Fig. 7). The shell wall thickness slightly increases around the shell circumference, where the shell is 9.8 mm in length and 8.4 mm in height. On the internal mould, the wrinkle layer and low longitudinal ridges suddenly disappear. This is followed by a sudden increase in the thickness of the shell wall around the entire circumference; the thickening of the shell is irregular with a ventrolateral maximum of almost 2 mm. Subsequently, the shell thickness decreases again adaperturally until it reaches almost the normal thickness. The wrinkle layer is again indicated. The shell axis direction shifts towards the dorsum for a length of about 2.8 mm just at the aperture. The line of a sudden increase in the shell thickness is reflected on the shell surface by an irregular sculpture persisting to the aperture. However, this is not accompanied by a change in shell expansion. The radial ridges and growth lines are irregular and wavy; thin radial lirae appear in some places, while the shell is smooth in others. On the thickened shell there is a large dorsal pit, followed dorsolaterally by small pits; the area of small pits is surrounded by gradually disappearing fine ridges forming an irregular network.
The described specimen CGS SM 187 (Fig. 7) shows one of the most striking pathologies ever described in an early Paleozoic cephalopod shell. The anomalous growth and secretion of the shell is likely a response to an ultimately lethal parasite infestation, probably located between the mantle and the shell (for discussion see De Baets et al. 2021). Pit-like pathologies on internal moulds are known from several Silurian cephalopods but have not yet been recorded in association with a change in sculpture.
Anomalous growth line development was found in one embryonic shell (Fig. 7B). It is manifested by a repeated irregular course of growth lines, their insertion and duplication. The causes of the anomalous sculpture development are uncertain. Similar embryonic shell anomalies have not yet been detected in orthoceratids, but are known in nautilids (Chirat 2001).
The frequency of pathologies in the studied collection shows that some kind of lethal or survivability-reducing pathology caused by parasites affected at most a few percent of the population. The pathologies found on the Lepidoceras lepidum shells correspond to those found in multiceratoids and nautilids (see De Baets et al. 2021). So far, the fossil record from the Silurian of the Prague Basin contains pathologies exclusively in demersal cephalopods.
Conclusions
Lepidoceras lepidum is a small endogastric orthoceratid with a prominent reticulate sculpture. From the late Wenlock to the late late Ludlow, there is a tendency for the curvature of the shell to increase. Number of longitudinal ridges did not change during life and there is a prominent intraspecific variation in their number. The distribution pattern and shell morphology of the only species of the genus, Lepidoceras lepidum (Barrande, 1866), suggest that it was a specialized demersal cephalopod since hatching.
The embryonic shell reached a height of about 2.5 mm and a length of 4–5 mm and started with non-accretion initial portion of the shell. Its shape varies between straight conical and curved. The hatching is usually manifested by more or less distinct growth walls. There is an increase in egg size and a simultaneous decrease in shell size and length of the body chamber, compared to its relatives. This implies a reduction in fertility and an increase in incubation time to a rate comparable to some K-strategy oncocerids. There is no correlation between body and egg size. Septal crowding and thickening of the apertural margin of the shell wall are adult modifications found. The modifications are not necessarily quite terminal either, but can generally be regarded as indicative of maturity or near-maturity. The adult modifications illustrate a variation in the size of the fully-grown shell which has not been found in orthoceratids before. Orthoceratomorph muscle scars consist of a pair of retractors appears mid-dorsally and gradually expand over half the shell circumference. Longer convex dorsal side and shorter straight ventral side of the body chamber influenced the muscle function, the efficiency of jet propulsion was probably higher than in straight-shelled orthoceratids. The colour pattern consists of irregular transverse bands; this is the first time such a colour pattern has been detected in orthoceratids. Slightly curved shells exhibit irregular colour bands while more curved shells show less irregular bands. This is the second reported case of adaptive convergence between orthoceratids and oncocerids and support for the protective function of colouration.
Shell repairs in L. lepidum were quantified with respect to the repair size and position on the shell. In juvenile stage, there is a higher frequency of repairs, especially massive ones. Juveniles were able to survive even extensive shell loss of around 25%. The relatively high frequency of shell repairs probably indirectly illustrates the demersal habit because shell repairs are generally very rare in Silurian pelagic cephalopods. The study of the frequency and extent of shell repair and preliminary examination of co-occurring species points at the possibility of a comparative study of predation marks. However, there is a lack of data on the frequency and extent of cephalopod shell repairs allowing comparisons of individual morphotypes across the early Paleozoic, which should be a part of future research. Pathological shells representing about four percent of available specimens. Anomalous growth of the embryonic shell is described for the first time in orthoceratids. The morphology and its intraspecific variation in L. lepidum is among the best known among orthoceratoids, although the internal structure of the embryonic shell has not been determined. Lepidoceras lepidum is a model example of the evolution of a demersal orthocerid known from only one peri-Gondwanan basin over a period of about 5 million years.
Acknowledgements
We are indebted Lenka Váchová (National Museum, Prague, Czech Republic) for optical photographing and Martina Aubrechtová (Czech Academy of Sciences, Prague, Czech Republic) kindly improved the English and made a number of suggestions to improve the manuscript. The authors thank Jessica Cundiff for help during our stay in the Museum of Comparative Zoology in Harvard. The comments of the journal referees, Alexander Pohle (Ruhr University Bochum, Germany) and an anonymous reviewer, were important for the final form of the manuscript. This research was supported by the Czech Grant Agency through the project GA20-23363S (Biostratigraphy and faunal dynamics of the Silurian (ŠM), the Strategic Research Plan of the Czech Geological Survey (DKRVO/ČGS 2023–2027; internal task No. 311410) and by the Ministry of Culture of the Czech Republic (IP DKRVO 2024-2028/2.IIIa) (VT).
Editor: Andrzej Kaim
References
Arnold, J.M. 1985. Shell growth, trauma, and repair as an indicator of life history for Nautilus. Veliger 27: 386–396.
Arnold, J.M., Landman, N.H., and Mutvei, H. 1987. Development of the embryonic shell of Nautilus. In: W.B. Saunders and N.H.E. Landman (eds.), Nautilus. The Biology and Paleobiology of a Living Fossil, 373–400. Springer, Dordrecht. Crossref
Aubrechtová, M., Turek, V., and Zicha, O. 2020. Early ontogenetic growth stages of Middle Ordovician orthoceratoid cephalopods from Bohemia. Acta Palaeontologica Polonica 65: 575–588. Crossref
Barrande, J. 1865–1877. Systême silurien du Centre de la Bohéme, Ière partie: Recherches Paléontologiques, vol. II, Classe de Mollusques, Ordre des Céphalopodes; 1865: Série 1, planches 1–107; 1866: Série 2, planches 108–244; 1867: Série 3, 712 pp.; 1868: Série 4, planches 245–350; 1870: Série 5, 266 pp., Série 6, planches 351–460; 1874: Série 7, 804 pp.; 1877: Série 8, 742 pp., Série 9, 743 pp., Supplement 1, 297 pp., Supplement 2, planches 461–544. Privately published, Praha.
Barskov, I.S. 2018. Morphological diversity of the external shell sculpture of Paleozoic cephalopods: How did it arise and what does it mean? Paleontological Journal 52: 1741–1745. Crossref
Beck, S.M., De Baets, K., Klug, C., and Korn, D. 2021. Analysis of septal spacing and septal crowding in Devonian and Carboniferous ammonoids. Swiss Journal of Palaeontology 140: 1–21. Crossref
Bek, J., Štorch, P., Tonarová, P., and Libertín, M. 2022. Early Silurian (mid-Sheinwoodian) palynomorphs from the Loděnice-Špičatý vrch, Prague Basin, Czech Republic. Bulletin of Geosciences 97: 385–396. Crossref
Blind, W. 1988. Comparative investigations on the shell morphology and structure of Nautilus pompilius, Orthoceras sp., Pseudorthoceras sp., and Kionoceras sp. In: J. Wiedmann and J. Kullmann (eds.), Cephalopods—Present and Past, 273–289. Schweizerbart’sche Verlagsbuchhandlung, Stuttgart.
Bond, P.N. and Saunders, W.B. 1989. Sublethal injury and shell repair in Upper Mississippian ammonoids. Paleobiology 15: 414–428. Crossref
Chamberlain, J.A. Jr. 1993. Locomotion in ancient seas: constraint and opportunity in cephalopod adaptive design. Geobios 15: 49–61. Crossref
Chen, J.-Y., Liu G.-W., and Chen T.-E. 1981. Silurian nautiloid faunas of central and southwestern China [in Chinese with English summary]. Memoirs of the Nanjing Institute of Geology and Palaeontology 13: 1–104.
Chirat, R. 2001. Anomalies in embryonic shell growth in post-Triassic Nautilida. Paleobiology 27: 485–499. Crossref
Chirat, R. and von Boletzky, S. 2003. Morphogenetic significance of the conchal furrow in nautiloids: evidence from early embryonic shell development of Jurassic Nautilida. Lethaia 36: 161–170. Crossref
Davis, R.A, Landman, N.H., Dommergues, J.L., Marchand, D., and Bucher, H. 1996. Mature modifications and sexual dimorphism in ammonoids. In: N.H. Landman, K. Tanabe, and R.A. Davis (eds.), Ammonoid Paleobiology, 463–539. Plenum Press, New York. Crossref
De Baets, K., Hoffmann, R., and Mironenko, A. 2021. Evolutionary history of cephalopod pathologies linked with parasitism. In: K. De Baets and J.W. Huntley (eds.), The Evolution and Fossil Record of Parasitism, Topics in Geobiology 50: 203–249. Springer, Dordrecht. Crossref
De Baets, K., Klug, C., Korn, D., and Landman, N.H. 2012. Early evolutionary trends in ammonoid embryonic development. Evolution 66: 1788–1806.
Doguzhaeva, L. 2002. Adolescent bactritoid, orthoceroid, ammonoid and coleoid shells from the Upper Carboniferous and Lower Permian of the South Urals. Abhandlungen der Geologichen Bundesanstalt 57: 9–55.
Doguzhaeva, L.A., Mutvei, H., and Mapes, R.H. 1999. Early ontogeny of the siphuncle and shell in the Early Carboniferous Rayonnoceras (Actinocerida) from Arkansas, USA. In: F. Olóriz and F.J. Rodríguez-Tovar (eds.), Advancing Research on Living and Fossil Cephalopods, 255–261. Springer, Boston, MA. Crossref
Dzik, J. 1984. Phylogeny of the Nautiloidea. Paleontologia Polonica 45: 1–155.
Erben, H.K. and Flajs, G. 1975. Über die Cicatrix der Nautiloideen. Mitteilungen des Geologisch-Paläontologischen Instituts der Universität Hamburg 44: 59–68.
Evans, D.H. 2007. A Middle Ordovician cephalopod fauna from Cuzco Province, southern Perú and its palaeobiogeographical significance. Geological Journal 42: 25–36. Crossref
Ferretti, A. and Kříž, J. 1995. Cephalopod limestone biofacies in the Silurian of the Prague basin, Bohemia. Palaios 10: 240–253. Crossref
Flower, R.H. 1939. Study of Pseudorthoceratidae. Palaeontographica Americana 11: 1–214.
Flower, R.H. 1962 Notes on the Michelinoceratida. New Mexico Institute of Mining and Technology, State Bureau of Mines and Mineral Resources, Memoir 10: 21–40.
Foerste, A.F. 1930. The color patterns of fossil cephalopods and brachiopods, with notes on gastropods and pelecypods. Contribution from the Museum of Paleontology University of Michigan 3: 109–150.
Foerste, A.F. 1932. Black River and other cephalopods from Minnesota, Wisconsin, Michigan, and Ontario, and other areas. Denison University Bulletin, Journal of the Scientific Laboratories 27: 47–136.
Foerste, A.F. 1936. Silurian cephalopods of the Port Daniel area on Gaspe Peninsula, Eastern Canada. Journal of the Scientific Laboratories of Denison University 31: 21–92.
Gnoli, M. and Serventi, P. 2008. A new cephalopod from the early Silurian of the Carnic Alps (Italian side). Rivisita Italiana di Paleontologia e Stratigrafia 114: 171–178. Crossref
Havlíček, V. 1981. Development of a linear sedimentary depression exemplified by the Prague Basin (Ordovician–Middle Devonian; Barrandian area – central Bohemia). Sborník geologických věd, Geologie 35: 7–48.
Havlíček, V. 1995. New data on the distribution of brachiopods in the Motol and lowest Kopanina Formations (Wenlock, lower Ludlow, Prague Basin, Bohemia). Věstník Ústředního ústavu geologického 70: 47–63.
Havlíček, V. 1998. Prague Basin. In: I. Chlupáč, V. Havlíček, J. Kříž, Z. Kukal, and P. Štorch, Palaeozoic of the Barrandian (Cambrian to Devonian), 39–41. Český geologický ústav, Praha.
Hewitt, R.A. 1988. Significance of early septal ontogeny in ammonoids and other ectocochliates. In: J. Wiedmann and J. Kullmann, J. (eds.), Cephalopods Present and Past, 207–214. E. Schweizerbartische Verlagsbuchhandlung, Stuttgart.
Histon, K. 2012. The Silurian nautiloid-bearing strata of the Cellon Section (Carnic Alps, Austria): color variation related to events. Palaeogeography, Palaeoclimatology, Palaeoecology 367–368: 232–256. Crossref
Hoffmann, R. and Keupp, H. 2015. Ammonoid paleopathology. In: C. Klug, D. Korn, K. De Baets, I. Kruta, and R.H. Mapes (eds.), Ammonoid Paleobiology, Volume I: From Anatomy to Ecology, 877–926. Springer, Dordrecht. Crossref
Holland, C.H. 1984. Form and function in Silurian cephalopods. Special Papers in Palaeontology 32: 151–164.
Holland, C.H. 2007. Cyrtoconic nautiloid cephalopods from the British Silurian. Proceedings of the Geologists’ Association 118: 365–337. Crossref
Horný, R. 1956. O rodu Dawsonoceras Hyatt, 1884 (Nautiloidea) ze středočeského siluru. Sborník Ústředního Ústavu geologického, Oddíl paleontologický 22: 425–452.
Hyatt, A. 1884. Genera of fossil cephalopods. Proceedings of the Boston Society of Natural History 22: 273–338.
Jaitly, R., Ehrnsten, E., Hedlund, J., Cant, M., Lehmann, P., and Hayward, A. 2022. The evolution of predator avoidance in cephalopods: A case of brain over brawn? Frontiers in Marine Science 9: 909192. Crossref
King, A.H. and Evans, D.H. 2019. High-level classification of the nautiloid cephalopods: a proposal for the revision of the Treatise Part K. Swiss Journal of Palaeontology 138: 65–85. Crossref
Klofak, S.M., Landman, N.H., and Mapes, R.H. 1999. Embryonic development of primitive ammonoids and the monophyly of the Ammonoidea. In: F. Oloriz and F.J. Rodriquez-Tovar (eds.), Advancing Research on Living and Fossil Cephalopods, 23–42). Kluwer Academic / Plenum Publishers, New York. Crossref
Klug, C. 2007. Sublethal injuries in Early Devonian cephalopod shells from Morocco. Acta Palaeontologica Polonica 52: 749–759.
Klug, C., Zatoń, M., Parent, H., Hostettler, B., and Tajika, A. 2015c. Mature modifications and sexual dimorphism. In: C. Klug, D. Korn, K. De Baets, I. Kruta, and R.H. Mapes (eds.), Ammonoid Paleobiology, Volume 1: From Anatomy to Ecology, 261–328. Springer, Dordrecht. Crossref
Kobluk, D.R. and Mapes, R.H. 1989. The fossil record, function and possible origins of shell color patterns in Paleozoic marine invertebrates. Palaios 4: 63–85. Crossref
Kolebaba, I. 1973. Embryonální stádia hlavonožců ze svrchních poloh Liteňského souvrství (silur, spodní wenlock). Časopis Národního Muzea, Oddíl přírodovědný 142: 28–40.
Kolebaba I. 1977. New information on longitudinally sculptured orthceroids. Časopis pro mineralogii a geologii 22: 125–138.
Kříž, J. 1992. Silurian field excursions: Prague Basin (Barrandian), Bohemia. National Museum of Wales, Geologica Series 13: 1–111.
Kříž, J. 1998a. Recurrent Silurian–lowest Devonian cephalopod limestones of Gondwanan Europe and Perunica. New York State Museum Bulletin 491: 183–198.
Kříž, J. 1998b. Silurian. In: I. Chlupáč, V. Havlíček, J. Kříž, Z. Kukal, and P. Štorch, Palaeozoic of the Barrandian (Cambrian to Devonian), 79–101. Český geologický ústav, Praha.
Kříž, J. 1999. Bivalvia dominated communities of Bohemian type from the Silurian and Lower Devonian carbonate facies. In: A.J. Boucot and J.D. Lawson (eds.), Final Report, Project Ecostratigraphy. Paleocommunities: A Case Study from the Silurian and Lower Devonian, 229–252. Cambridge University Press, Cambridge.
Kříž, J. 2008. A new bivalve community from the lower Ludlow of the Prague Basin (Perunica, Bohemia). Bulletin of Geosciences 83: 237–280. Crossref
Kříž, J., Dufka, P., Jaeger, H., and Schönlaub, H.P. 1993. The Wenlock/Ludlow boundary in the Prague Basin (Bohemia). Jahrbuch der Geologischen Bundesanstalt 136: 809–839.
Kröger, B. 2004. Large shell injuries in Middle Ordovician Orthocerida (Nautiloidea, Cephalopoda). GFF 126: 311–316. Crossref
Kröger, B. 2006. Early growth-stages and classification of orthoceridan Cephalopods of the Darriwillian (Middle Ordovician) of Baltoscandia. Lethaia 39: 129–139. Crossref
Kröger, B. 2011. Size matters—analysis of shell repair scars in endocerid cephalopods. Fossil Record 14: 109–118. Crossref
Kröger, B. and Aubrechtová, M. 2019. The cephalopods of the Kullsberg Limestone Formation, Upper Ordovician, central Sweden and the effects of reef diversification on cephalopod diversity. Journal of Systematic Palaeontology 17: 961–995. Crossref
Kröger, B. and Isakar, M. 2006. Revision of annulated orthoceridan cephalopods of the Batoscandic Ordovician. Fossil Record 9: 137–163. Crossref
Kröger, B. and Landing, E. 2007. Early Orthoceratoid cephalopods from the Argentine Precordillera (Lower–Middle Ordovician). Journal of Paleontology 81: 1266–1283. Crossref
Kröger, B. and Mapes, R. 2004. Lower Carboniferous (Chesterian) embryonic orthoceratid nautiloids. Journal of Paleontology 78: 560–573. Crossref
Kröger, B. and Mapes, R. 2007. Carboniferous actinoceratoid Nautiloidea (Cephalopoda): A new perspective. Journal of Paleontology 81: 714–724. Crossref
Kröger, B., Klug, C., and Mapes, R. 2005. Soft−tissue attachments in orthocerid and bactritid cephalopods from the Early and Middle Devonian of Germany and Morocco. Acta Palaeontologica Polonica 50: 329–342.
Kröger, B., Servais, T., and Zhang, Y. 2009. The origin and initial rise of pelagic cephalopods in the Ordovician. PLoS ONE 4 (9): e7262. Crossref
Kuhn, O. 1940. Paläozoologie in Tabellen. 50 pp. Fischer Verlag, Jena.
Manda, Š. 1996. Cyrtograptus lundgreni Biozone in the southwestern part of the Svatý Jan Volcanic Centre (Wenlock, Prague Basin). Věstník Českého geologického ústavu 71: 369– 374.
Manda, Š. 2008. Palaeoecology and palaeogeographic relations of the Silurian phragmoceratids (Nautiloidea, Cephalopoda) of the Prague Basin (Bohemia). Bulletin of Geosciences 83: 39–62. Crossref
Manda, Š. and Kříž, J. 2006. Environmental and biotic changes of the subtropical isolated carbonate platforms during Kozlowskii and Lau events (Prague Basin, Silurian, Ludlow). GFF 128: 161–168. Crossref
Manda, Š. and Turek, V. 2009a. A Silurian oncocerid with preserved colour pattern and muscle scars (Nautiloida). Bulletin of Geosciences 84: 755–766. Crossref
Manda, Š. and Turek, V. 2009b. Minute Silurian oncocerids with unusual colour pattern (Nautiloidea). Acta Palaeontologica Polonica 54: 503–512. Crossref
Manda, Š. and Turek, V. 2015. Colour patterns on Silurian orthocerid and pseudorthocerid conchs from Gotland—palaeoecological implications. Estonian Journal of Earth Sciences 64: 74–79. Crossref
Manda, Š. and Turek, V. 2019. Embryonic and early juvenile development in the Silurian basal nautilid Peismoceras Hyatt, 1894. Swiss Journal of Palaeontology 138: 123–139. Crossref
Manda, Š. and Turek, V. 2024. Early ontogeny and distribution of the orthocerid cephalopod Calorthoceras Chen, 1981—taxonomic and palaeoecological implications. Bulletin of Geosciences 99: 323–342. Crossref
Manda, Š., Štorch, P., Frýda, J., Slavík, L., and Tasáryová, Z. 2019. The mid-Homerian (Silurian) biotic crisis in off-shore settings of the Prague Synform, Czech Republic: integration of the graptolite fossil record with conodonts, shelly fauna and carbon isotope data. Palaeogeography, Palaeoclimatology, Palaeoecology 528: 14–34. Crossref
Marek, J. 1971. The genus Cyrtocycloceras Foerste, 1936 (Nautiloidea) from the Silurian of central Bohemia. Sborník geologických Věd, Paleontologie 14: 107–133.
Mergl, M. 2024. Lingulates of the Monograptus belophorus Biozone (Motol Formation, Sheinwoodian, Wenlock) of the Barrandian area, Czech Republic: insight into remarkable lingulate brachiopod diversity in the Silurian. Bulletin of Geosciences 99: 1–42. Crossref
Mironenko, A.A. 2018. First possible evidence of parasite infestation in Upper Devonian Discosorida (Nautiloidea). Swiss Journal of Palaeontology 137: 77–82. Crossref
Murchison, R.I. 1839. The Silurian System Founded on Geological Researches in the Counties of Salop, Hereford, Padnor, with Descriptions of the Coal Fields and Overlying Formations. Part 1. 768 pp. John Murray, London.
Mutvei, H. 1957. On the relations of the principal muscles to the shell in Nautilus and some fossil nautiloids. Arkiv for Mineralogi och Geologi 2: 219–254.
Mutvei, H. 2002. Nautiloid systematics based on siphuncular structure and position of muscle scars. Abhandlungen der Geologischen Bundesanstalt 57: 379–392.
Mutvei, H. and Doguzhaeva, L.A. 1997. Shell ultrastructure and onto-genetic growth in Nautilus pompilius L. (Mollusca: Cephalopoda). Palaeontographica, Abteilung A 246: 33–52. Crossref
Mutvei, H, Arnold, J.M., and Landman, N.H. 1993. Muscles and attachment of the body to the shell in embryos and adults of Nautilus balauensis (Cephalopoda). American Museum Novitates 3059: 1–15.
Niko, S., Sone, M., and Leman, M.S. 2020. Ordovician Orthocerida and Pseudorthocerida (Cephalopoda: Nautiloidea) from the Lower Setul Limestone of the Langkawi Islands, Malaysia. Journal of Systematic Palaeontology 18: 381–414. Crossref
Peterman, D.J., Barton, C.C., and Yacobucci, M.M. 2019. The hydrostatics of Paleozoic ectocochleate cephalopods (Nautiloidea and Endoceratoidea) with implications for modes of life and early colonization of the pelagic zone. Palaeontologica Electronica 22.2.27A: 1–29. Crossref
Pohle, A., Fuchs, D., Korn, D., and Klug, C. 2020. Spatial distribution of oncocerid cephalopods on a Middle Devonian bedding plane suggests semelparous life cycle. Scientific Reports 10: 2847. Crossref
Pohle, A., Kröger, B., Warnock, R.C.M., King, A.H., Evans, D.H., Aubrechtová, M., Cichowolski, M., Fang, X., and Klug, C. 2022. Early cephalopod evolution clarified through Bayesian phylogenetic inference. BMC Biology 20: art. 88. Crossref
Prantl, F. 1936. O novém nálezu polohy s Aulacopleura konincki (Barr.). Časopis Národního musea, Oddíl přírodovědný 60: 1–8.
Ristedt, H. 1968. Zur Revision der Orthoceratidae. Abhandlungen der mathematisch-naturwissenschaftlichen Klasse 4: 1–77.
Ristedt, H. 1971. Zum Bau der orthoceriden Cephalopoden. Palaeontographica Abteilung A 137: 155–195.
Rocha, F., Guerra, Á., and González, Á.F. 2001. A review of reproductive strategies in cephalopods. Biological Reviews 76: 291–304. Crossref
Schindewolf, O.H. 1933. Vergleichende Morphologie und Phylogenie der Anfangskammern tetrabranchiater Cephalopoden. Eine Studie über Herkunft, Stammenentwicklung, und System der niederen Ammoeen. Abhandlungen der Preussischen Geologischen Landesanstalt, Neue Folge 148: 1–115.
Serpagli, E. and Gnoli, M. 1977. Upper Silurian cephalopods from Southwestern Sardinia. Bollettino della Paleontologica Italiana 16: 153–196.
Sowerby, J.C. 1818. The Mineral Conchology of Great Britain, Volume 2. 251 pp. Printed by B. Meredith, London.
Štorch, P. 2023. Graptolite biostratigraphy and biodiversity dynamics in the Silurian System of the Prague Synform (Barrandian area, Czech Republic). Bulletin of Geosciences 98: 1–78. Crossref
Štorch, P., Manda, Š., and Loydell, D.K. 2014. The Early Ludfordian leintwardinensis graptolite Event and the Gorstian-Ludfordian boundary in Bohemia (Silurian, Czech Republic). Palaeontology 57: 1003–1043.
Stridsberg, S. 1985. Silurian oncocerid cephalopods from Gotland. Fossils and Strata 18: 1–65. Crossref
Sweet, W.C. 1959. Muscle attachment impressions in some Paleozoic nautiloid cephalopods. Journal of Paleontology 33: 293–304.
Teichert, C. 1964. Morphology of hard parts. In: R.C. Moore (ed.), Treatise on Invertebrate Paleontology, Part K, Mollusca 3, Cephalopoda, K13–K54. Geological Society of America and University of Kansas Press, Lawrence. Crossref
Teichert, C. 1967. Major features of cephalopod evolution. In: C. Teichert and E.L. Yochelson (eds.), Essays in Paleontology and Stratigraphy: R.C. Moore Commemorative Volume, 162–210. University of Kansas Press, Lawrence.
Troedsson, G.T. 1926. On the Middle and Upper Ordovician faunas of northern Greenland. I. Cephalopods. Meddelelser om Grønland 71: 1–157.
Turek, V. 1983. Hydrodynamic conditions and the benthic community of upper Wenlockian calcareous shale in western part of the Barrandian (Kosov quarry). Časopis pro mineralogii a geologii 28: 245–260.
Turek, V. 2008. Boionautilus gen. nov. from the Silurian of Europe and North Africa (Nautiloidea, Tarphycerida). Bulletin of Geosciences 83: 141–152. Crossref
Turek, V. 2009. Colour patterns in Early Devonian cephalopods from the Barrandian Area: Taphonomy and taxonomy. Acta Palaeontologica Polonica 54: 491–502. Crossref
Turek, V. and Manda, Š. 2012. “An endocochleate experiment” in the Silurian straight-shelled cephalopod Sphooceras. Bulletin of Geosciences 87: 767–813. Crossref
Turek, V. and Manda, Š. 2020. Discontinuous, asymmetric and irregular colour patterns in Silurian oncocerids (Nautiloidea) with cyrtoconic shells. Bulletin of Geosciences 95: 333–367. Crossref
Vodička, J. and Manda, Š. 2019. A taxonomical and statistical study of chitinozoan distribution across the lundgreni Event (Wenlock, Silurian) from the Prague Basin, Czech Republic: A specific pattern driven by ecological changes. Marine Micropaleontology 149: 44–63. Crossref
Yomogida, S. and Wani, R. 2012. Higher risk of fatality by predatory attacks in earlier ontogenetic stages of modern Nautilus pompilius. Lethaia 46: 317–330. Crossref
Zou, Xi-Ping 1988. Ordovician nautiloid faunas from Lunshan, Jurong, Jiangsu. Acta Palaeontologica Sinica 27: 309–330.
Acta Palaeontol. Pol. 70 (3): 517–541, 2025
https://doi.org/10.4202/app.01243.2025