

A new hoverfly genus from the Oligocene of France with unusual morphology
VALENTIN NIDERGAS, JIŘÍ HADRAVA, PAULINE COSTER, and ANDRÉ NEL
Nidergas, V., Hadrava, J., Coster, P., and Nel, A. 2025. A new hoverfly genus from the Oligocene of France with unusual morphology. Acta Palaeontologica Polonica 70 (4): 811–816.
Here, we describe Intricodon cryptodaemoniacus, a new fossil genus and species of hoverfly (Diptera: Syrphidae), representing the second described hoverfly from the middle Oligocene of Céreste (Luberon, Southern France). It has unique, highly specialized characters like hind legs strongly thickened with processes on femur and tibia and the shape of the wing veins R4+5 and M1. Based on the visible characters on the well preserved compression fossil specimen, the new genus resembles some taxa currently attributed to the Merodontini in the wing venation and/or leg morphology. The systematic affinities and potential palaeoecology of the new genus are discussed through the comparison to the more similar Recent genera.
Key words: Insecta, Diptera, Syrphidae, Merodontini, Oligocene, Cenozoic.
Valentin Nidergas [valentin.nidergas@gmail.com; ORCID: https://orcid.org/0000-0002-4314-5619 ], André Nel [anel@mnhn.fr; ORCID: https://orcid.org/0000-0002-4241-7651 ], Institut de Systématique, Évolution, Biodiversité (UMR 7205) Muséum national d’Histoire naturelle, CNRS, Sorbonne Université, EPHE-PSL, Université des Antilles, F-75005 Paris, France.
Jiří Hadrava [hadravaj@natur.cuni.cz; ORCID: https://orcid.org/0000-0003-3247-6399 ], Department of Zoology, Faculty of Science, Charles University, Viničná 7, CZ-128 43, Praha 2, Czech Republic; and Institute of Entomology, Biological Centre, Czech Academy of Science, Branišovská 31, CZ-370 05 České Budějovice, Czech Republic.
Pauline Coster [pauline.coster@parcduluberon.fr; ORCID: https://orcid.org/0000-0003-4015-252X ], Réserve naturelle nationale géologique du Luberon, Parc naturel régional du Luberon, 60 Place Jean Jaurès, 84400 Apt, France.
Received 23 March 2025, accepted 9 September 2025, published online 19 December 2025.
Copyright © 2025 V. Nidergas et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
With more than 6,200 extant species, Syrphidae are a highly diversified group of Diptera. They exhibit a wide range of ecological strategies and provide important ecosystem services (Ssymank et al. 2021). Recent phylogenetic inferences have estimated their origin in the Cretaceous period, followed by a rapid radiation during the Paleogene (Mengual et al. 2023; Wong et al. 2023; Wu et al. 2024).
Although more than 100 fossil species of hoverflies have been described so far (Evenhuis 1994), the number of described fossil hoverflies is low compared to many other fly families (e.g., Tipuloidea or Sciaroidea). Several new Syrphidae have been documented in recent studies, and numerous others still remain to be described (Nidergas et al. 2018; Hadrava et al. 2020). Describing and classifying new fossil taxa is important for understanding the morphological evolution of the study group throughout the ages (e.g., Daňková et al. 2025), and for improving the molecular phylogeny dating, using them as calibrating points.
In the present study, we describe a new genus and species from the Oligocene lacustrine deposits near the village of Céreste, Luberon, southern France.
Institutional abbreviations.—PNRL, Réserve naturelle nationale géologique du Lubéron, France.
Other abbreviations.—C, costa; CuA, cubitus anterior; dm-m, crossvein between M1 and M4; M, median vein; M1, first branch of M; M4, second branch of M; m-cu, crossvein between M4 and CuA; R, radial vein; R1, anterior branch of R; r1, cell between R1 and R2+3; R2+3, second branch of R; r2+3, cell between R2+3 and R4+5; R4+5, third branch of R; r4+5, cell between R4+5 and M1; r-m, crossvein between R4+5 and M1; Rs, radial sector; Sc, subcosta; sprs, spurious vein.
Nomenclatural acts.—This published work and the nomenclatural acts it contains have been registered in Zoobank LSID: urn:lsid:zoobank.org:pub:467F5171-4DCD-4F20-BB0E-93909B21D378.
Material and methods
The fossiliferous outcrop is located west of La Bastide du Bois, south of the small village of Céreste, in Luberon (France); for details on the site and the fossil assemblage, see Nel et al. (2024 and references therein). These fossiliferous sites are protected by the Réserve naturelle nationale géologique du Lubéron (Coster and Legal 2021).
The fossil was found in the year 2000 by AN. Drawings were made with Inkscape v0.92 using a drawing tube mounted on a Nikon D800. Photographs were made with a reflex Nikon D800 mounted on a Nikon SMZ25 stereomicroscope, manipulated with DxO PhotoLab v1.1.2 and Gimp v2.8.6 and then focus-stacked with the “D-map” algorithm on Zerene Stacker v1.04. We follow Ssymank et al. (2021) for the morphological terminology.
Systematic palaeontology
Order Diptera Linnaeus, 1758
Family Syrphidae Latreille, 1802
Subfamily Eristalinae Newman, 1834
Tribe ?Merodontini Edwards, 1915
Genus Intricodon nov.
Zoobank LSID: urn:lsid:zoobank.org:act:24010AA2-4B4F-4172-9EA6- 7511F12BAC32.
Type species: Intricodon cryptodaemoniacus gen. et sp. nov.; see below.
Etymology: Combination of Latin intricatus, complicated; in reference to the difficulty placing this fossil genus in an extant tribe due to its unusual morphology; and the suffix odon, which refers to the extant genus Merodon Meigen, 1803, due to the likely taxonomic affinity between Intricodon and the Merodontini tribe.
Diagnosis.—As for the type species, by monotypy.
Remarks.—The new fossil genus is assigned to the Syrphidae (Fig. 1) with no doubt due to the characteristic wing venation, with the presence of the vena spuria crossing the r-m vein between veins Rs-R4+5 and M, and the veins M1 and dm-m running parallel to the wing margin (Fig. 2). It does not seem to be related to the subfamily Microdontinae nor the tribe Volucellini because its vein M1 is not recessive (i.e., apically making an obtuse angle with the vein R4+5, directed toward the wing base instead of the wing apex when joining the vein R4+5), and its vein R2+3 is not basally curved as in Microdontinae (Reemer and Ståhls 2013). Intricodon gen. nov. is also not related to Syrphinae, because of its complex venation with strongly sinuate dm-m and distal part of M1 (false margin), strongly thickened legs, while Syrphinae have generally thin legs and a straighter false margin of the wing. Our new genus seems to be not related to subfamily Pipizinae because of the ending point of costa after wing apex and the vein R4+5 is apically curved in Intricodon gen. nov. while the latter vein is straight apically in this group (Mengual et al. 2015). The remaining possible attribution is the paraphyletic Eristalinae sensu lato.
Young et al. (2016) divided the Eristalinae into the Brachyopini, Callicerini, Cerioidini, Eristalini, Merodontini, Milesiini, Rhingiini, Sericomyiini, and Volucellini. Moran et al. (2022: 129) indicated that the “tribes Cerioidini, Volucellini and parts of Merodontini (including Merodon)” would be placed “outside of an otherwise monophyletic Eristalinae”. However, Wong et al. (2023) placed the Brachyopini, Callicerini, Cerioidini, Eristalini, Merodontini, Milesiini, Rhingiini, and Volucellini in Eristalinae, excluding the tribe Sericomyiini. Thus there seems that there is still no real consensus in the definition and limits of the Eristalinae.

Fig. 1. Syrphid hoverfly Intricodon cryptodaemoniacus gen. et sp. nov., holotype PNRL 2071, from Oligocene of Céreste, France. Photograph of habitus.
The Sericomyiini have the apices of R1 and R2+3 strongly approximate, unlike in the new fossil. The Volucellini, Cerioidini, Eristalini, and Milesiini have a shape of M1 and R4+5 very different from that of Intricodon gen. nov. Also the Cerioidini Monoceromyia Shannon, 1925, Polybiomyia Shannon, 1925, and Sphiximorpha Rondani, 1850, have the abdomen constricted. Ceriana Rafinesque, 1815, Monoceromyia Shannon, 1925, Primocerioides Shannon, 1927, and Sphiximorpha Rondani, 1850, have a frontal proeminence, absent in Intricodon gen. nov. (Van Steenis et al. 2016). The Brachyopini and Rhingiini have M1 making a right angle with R4+5, vs. distinctly acute in Intricodon gen. nov.
Intricodon gen. nov. shares some morphological characteristics with Merodontini that can be potential synapomorphies: R1 joining C beyond 0.6 of the distance from Sc to R2+3, M4 ending in posterior margin of wing (“dm-m stump vein present”), as proposed by Doczkal and Pape (2009) and Doczkal et al. (2016). But these characters are also present in some other groups. Also the distal end of M1 is more or less recurrent, forming an acute outer angle with R4+5 in nearly all Merodontini (Vockeroth 1964; Vockeroth and Thompson 1981; Hurkmans 1993; Doczkal and Pape 2009; Morales et al. 2009; Doczkal et al. 2016; Liu et al. 2022), while it is ending directly into R4+5 without making a strong curve in Intricodon gen. nov.
In Myolepta Newman, 1838 (Brachyopini), Notiocheilosia Thompson, 1972, Cyphipelta Bigot, 1859, and other genera currently placed in the Merodontini, such as Cepa Thompson & Vockeroth in Thompson, 2007, Nausigaster Williston, 1883 (a genus placed as sister group of the Merodontini by Doczkal and Pape, 2009) and Psilota Meigen, 1822, the distal end of M1 is not recurrent, but progressive (directed to the apex) and forming an acute angle with R4+5 as in Intricodon gen. nov. (Bezzi 1925; Hull 1949; Thompson 1972, 1999, 2007, 2008; Vockeroth and Thompson 1981; Barahona-Segovia and Barceló 2019; Young et al. 2020). Unlike in the other genera listed above, in the genera Cepa, Nausigaster, and Psilota the wing vein M1 has a pronounced curve midway somewhat similar to that in Intricodon cryptodaemoniacus gen. et sp. nov. And here, the medial anteroposterior length of the tergite III is distinctly smaller than tergites II and IV, which is also the case in Nausigaster, while Psilota has the tergites III and IV of nearly the same lengths. In Cepa and Psilota the vein R4+5 is nearly straight in nearly all its course as in Intricodon gen. nov., while in some Nausigaster species (but not all) the vein R4+5 has a smooth curve midway (Hull 1949; Thompson 1972). Additionally, in Cepa, Nausigaster, and Psilota the vein R4+5 has not a pronounced posterior curve just basal of its fusion with M1, like in Intricodon gen. nov. Nausigaster strongly differs from Intricodon gen. nov. in the shape of the abdomen with long tergite IV (Williston 1883: pl. 2: 15). Psilota also has a very different shape of abdomen with tergite III as long as tergite II (Young et al. 2020). Cepa also has a very different shape of abdomen, small oval, and compact (Thompson 1999, 2007).
Because of the similarities in venation with Nausigaster and the strong similarities in the hind leg morphology with Merodon, we propose to provisionally put Intricodon gen. nov. in the Merodontini. Nevertheless, Wong et al. (2023) did not include Nausigaster in their phylogenetic analysis. Wu et al. (2024: 8) indicated that “Due to the lack of data on Psilota Meigen, 1822, and Nausigaster Shannon, 1921, our study was unable to fully validate the monophyly of the Merodontini”. Rotheray et al. (2000: 133) indicated that “Nausigaster larvae were completely different [from those of the Merodontini] and were closely related to Criorhina Meigen, 1822, in the Xylotini”. Cepa was not included in the analyses of Wong et al. (2023) and Wu et al. (2024). In the analysis of Wong et al. (2023: fig. 2), Psilota does not fall at all with Merodon. Thus the positions of Nausigaster, Psilota, and Cepa need to be clarified to precise that of Intricodon gen. nov.
Some extant genera have their R4+5 pedicel greatly reduced and weakly marked, with the conjunction point between R4+5 and M1 almost reaching the tip of the costa, as it is the case in the New World genus Nausigaster.
Stratigraphic and geographic range.—Oligocene, Céreste, France.
Intricodon cryptodaemoniacus sp. nov.
Figs. 1–5.
Zoobank LSID: urn:lsid:zoobank.org:act:5453A719-68A2-4717-94A0- 9F5B161C1EB3.
Etymology: From Greek crypto, hidden and daemon, demon; in reference to the ornamentation of abdomen that looks like a demoniac angry face when watched upside-down.
Type material: Holotype PNRL 2071, a nearly complete male, part.
Type locality: Céreste, Alpes-de-Haute-Provence, France.
Type horizon: Campagne–Calavon Formation (European Mammal Reference Levels biozone MP23–24, 33.9–27.82 Ma) (Feist 1977; Cavelier et al. 1984; Apostolescu and Guernet 1992; Ménouret 2014), Rupelian, Oligocene.
Material.—Holotype only.
Diagnosis.—Small-sized species; head deformed but with face not pronounced (Fig. 1), compound eyes holoptic; basoflagellomeres oval-shaped. At least wing veins C, Sc, R, and M with microtrichia as long as the width of the veins; costa ending significantly after wing apex; subcostal vein basally straight, not running parallel to vein R, and making a pronounced angle at its apical 4/5; vein R1 slightly thicker than costa (ratio 4:3); crossvein r-m very long, about 0.8 the length of part of R4+5 basal to its junction with r-m; vein R2+3 nearly straight before its apical 4/5, and apically curved; vein R4+5 strongly curved basally, straight till distal 2/3 of its course after vein r-m, and finally curved downward and obliquely ending into M1; vein M1 making a strong U-shaped curve in its apical third (Fig. 2), and strongly concave apically; veins R4+5 and M1 joining together to form a pedicel half as long as r-m; vein dm-m strongly curved apically before its confluence point with M1. Hind femur strongly thickened with processes on femur and tibia (Fig. 3). Abdomen oval shaped (Fig. 4); tergites more densely setose laterally than in their median parts; medial length of tergite III distinctly shorter than the lengths of the tergites II and IV; abdominal tergite II with two sub-triangular pale spots with rounded angles, and tergite III with two oblique elongate sub-rectangular spots, 3× as long as wide.
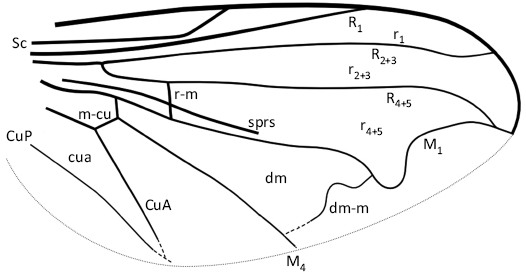
Fig. 2. Syrphid hoverfly Intricodon cryptodaemoniacus gen. et sp. nov., Reconstruction of wing. Abbreviations: CuA, cubitus anterior; cua, cubital cell; CuP, cubitus posterior; dm, cell between M1 and M4; dm-m, crossvein between M1 and M4; M, median vein; M1, first branch of M; M4, second branch of M; m-cu, crossvein between M4 and CuA; R, first branch of radius; R1, anterior branch of R; r1, cell between R1 and R2+3; R2+3, second branch of R; r2+3, cell between R2+3 and R4+5; R4+5, third branch of R; r4+5, cell between R4+5 and M1; r-m, crossvein between R4+5 and M1; Sc, subcosta; sprs, spurious vein. Not to scale.
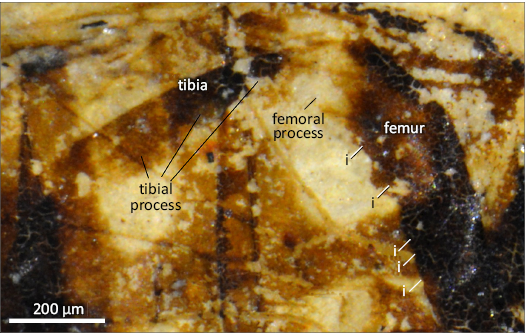
Fig. 3. Syrphid hoverfly Intricodon cryptodaemoniacus gen. et sp. nov., holotype PNRL 2071, from Oligocene of Céreste, France. Photograph of hind leg. Abbreviation: i, indentations of femur.
Description.—Body: 5.3 mm long. Head: 1.8 mm high, 1.1 mm long; face poorly preserved but apparently not produced; compound eyes poorly preserved, holoptic, diameter ca. 0.39 mm. Thorax ca. 0.84 mm long, 0.90 mm high; no visible setae on thorax. Wing: hyaline, ca. 4.1 mm long, 0.79 mm wide; basal part of pterostigma between Sc and R1 hyaline. Legs: Hind femur bicolor, strongly thickened, with dorsal margin making a smooth obtuse angle, about 153°, and a large, clear-coloured anteroventral apical process and at least five indentations along its anteroventral margin, gradually larger towards apex; hind tibia strongly thickened and flattened toward its apex (maximum width about 1.3× basal width), with a short, strong ventral process basally, and a strongly flattened “plate” on apical half, five times as long as wide, with a dense line of small punctuations, which could be interpreted as the bases of setae. Abdomen: Ovoid, 1.27 mm long, 1.01 mm wide; tergite III apparently slightly shorter than tergite II and IV; tergites II and III with two pale elongate spots, separate by a brown median zone, those of tergite II being subtriangular with anterior margin rounded, postero-distal angle acute, and postero-proximal angle straight, those of tergite III sub-quadrangular, about 3× longer than wide; spots of tergites II and III with about same inclination with posterior margin of tergites II, viz. 11–16°; tergite IV uniformly dark, without apparent markings. Genitalia: Hypandrium large, as long as tergite IV, almost reaching middle of tergite III in dorsal view.
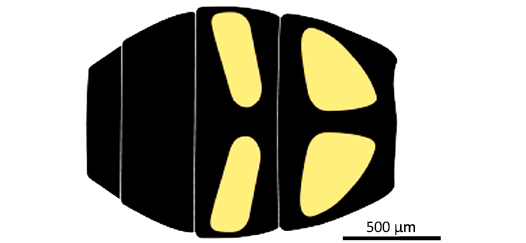
Fig. 4. Syrphid hoverfly Intricodon cryptodaemoniacus gen. et sp. nov. Reconstruction of abdomen.

Fig. 5. Syrphid hoverfly Intricodon cryptodaemoniacus gen. et sp. nov.. Reconstruction of habitus (artwork by Carim Nahaboo).
Remarks.—Nidergas et al. (2018) already described the pipizine syrphid genus and species Oligopipiza quadriguttata from the same layers. Syrphids are not infrequent in these layers and at least two other species need to be described.
The highly specialized hind leg of Intricodon cryptodaemoniacus gen. et sp. nov. strongly resembles those of some extant species of Merodon and Eumerus Fabricius, 1798, in the presence of an apical anteroventral process and indentations along the apicoventral margin of the metafemur and of a strong ventral tooth on the metatibia (Speight and Langlois 2020; Speight et al. 2021). Nevertheless, species of Merodon and Eumerus differs from the new fossil in the abdomen with tergite 3 as long as or longer than tergite 2 (Vujić et al. 2018; Speight et al. 2021).
No Eristalinae sensu lato (and even no extant Syrphidae) have a vein dm-m strongly curved close to M1 similar to that of Intricodon cryptodaemoniacus gen. et sp. nov., even if in some species of Myolepta and Nausigaster, this vein is curved near M4 and less than in Intricodon cryptodaemoniacus gen. et sp. nov. (Fluke and Weems 1956; Thompson, 1972). Also, no extant genus has such unusual venation similar to Intricodon cryptodaemoniacus gen. et sp. nov., with R4+5 fused with M1 and curved downward apically.
Stratigraphic and geographic range.—Oligocene, Céreste, France.
Discussion
The chaetotaxy on the wing veins Intricodon cryptodaemoniacus gen. et sp. nov. is very unusual. The selective advantage that would have led to this character remains unknown, as well as for the specialized structures on the hind femur and tibia of Intricodon cryptodaemoniacus gen. et sp. nov. There is a sexual dimorphism in some extant species of Merodon in the shape and development of the hind femoral and tibial processes, less developed in females than in males (Vujić et al. 2022). Thus, these structures could be related to the mating behavior in these flies.
Larvae of Merodon and Eumerus are phytophagous in soils or saprophagous (Orengo-Green et al. 2024; Gisbert 2024). Larvae of Nausigaster have been recorded from decaying cacti and bromeliads. It seems that, to date, nothing is known on the biology of the genus Cepa (Barahona-Segovia and Barceló 2019). Larvae of few Psilota species have been found in sap runs under tree bark (Kassebeer et al. 1998), otherwise, the biology of many species in this genus is unknown (Young et al. 2020). It is possible but not demonstrated that the larvae of Intricodon cryptodaemoniacus gen. et sp. nov. were phytophagous or saprophagous too.
Conclusions
Intricodon cryptodaemoniacus gen. et sp. nov. is a very enigmatic taxon, with a wing venation strongly different from those of the other Syrphidae. It confirms the richness and diversity of the syrphids in the Oligocene insect assemblage of Céreste. It also shows that, despite its relatively young age compared to the antiquity of this family, the Oligocene Syrphidae comprised some very original forms, and not only representatives of extant genera.
Acknowledgements
We sincerely thank Ximo Mengual (Museum Koenig Bonn, Germany) and an anonymous referee for their very useful comments on the first version of the paper. We also sincerely thank Emmanuel Delfosse and Christophe Daugeron (both Muséum national d’Histoire naturelle, Paris, France) for their courtesy and help in the consultation in collections of extant Syrphidae. We also thank Ximo Mengual for his help in finding extant related genera. The work of JH has been supported by Charles University Research Centre program No. UNCE/24/SCI/006.
Editor: Krzysztof Hryniewicz
References
Apostolescu, V. and Guernet, C. 1992. Les ostracodes oligocènes de la région Forcalquier-Manosque (Bassin continental d’Apt, Haute-Provence). Revue de Micropaléontologie 35: 91–115.
Barahona-Segovia, R.M. and Barceló, M. 2019. A new flower fly species of Cepa Thompson & Vockeroth (Diptera: Syrphidae) from the Valdivian evergreen forest hotspot. Zootaxa 4612: 431–439. Crossref
Bezzi, M. 1925. Monografia del genere Platynochaetus Wied. (Dipt.; Syrph.). Memorie della Societa Entomologica Italiana 3: 215–227.
Cavelier, C., Alabouvette, B., Amberger, G., Cautru, J.P., Charolais, J., Chateauneuf, J.J., Crochet, J.Y., Campredon, R., Debeglia, N., Durand, J.P., Feist-Castel, M., Freytet, P., Gannat, E., Gaudant, J., Giannerini, G., Godinot, M., Hartenberger, J.L., Hugueney, M., Kerckhove, C., Lefavrais-Raymond, A., Legendre, S., Lespinasse, P., Magne, J., Nury, D., Pairis, J.L., Plaziat, J.C., Remy, J.A., Schlund, J.M., Sige, B., Sittler, C., Sudre, J., Trauth, N., Truc, G., Valleron, M.M., Vianey-Liaud, M., and Villate, J. 1984. Paléogène. In: S. Debrand-Passard, S. Courbouleix, and M.J. Liehardt (eds.), Synthèse géologique du Sud-Est de la France, 389–468. BRGM, Orléans.
Coster, P. and Legal, S. 2021. An Early Oligocene fossil Lagerstätten from the lacustrine deposits of the Luberon UNESCO Global Geopark. Geoconservation Research 4 (2): 604–612.
Daňková, K., Hadrava, J., Straka, J., Mikát, M., Exnerová, A., Hulva, P., Nidergas, V., Pecharová, M., Nel, A. and Prokop, J. 2025. Highly accurate Batesian mimicry of wasps dates back to the Early Oligocene and was driven by non-passerine birds. Current Biology 35 (8): 1901–1909.e1. Crossref
Doczkal, D. and Pape, T. 2009. Lyneborgimyia magnifica gen. et sp. n. (Diptera: Syrphidae) from Tanzania, with a phylogenetic analysis of the Eumerini using new morphological characters. Systematic Entomology 34: 559–573. Crossref
Doczkal, D., Radenkovićn S., Lyneborg, L., and Pape, T. 2016. Taxonomic revision of the Afrotropical genus Megatrigon Johnson, 1898 (Diptera: Syrphidae). European Journal of Taxonomy 238: 1–36. Crossref
Evenhuis, N.L. 1994. Catalogue of the fossil flies of the World (Insecta: Diptera). 600 pp. Backhuys Publishers, Leiden.
Feist, M. 1977. Étude floristique et biostratigraphique des Charophytes dans les séries du Paléogène de Provence. Géologie Méditerranéenne 4: 109–138. Crossref
Fluke, C.L. and Weems, H.V. 1956. The Myoleptini of the Americas (Diptera. Syrphidae). American Museum Novitates 1758: 1–23.
Gisbert, A.A. 2024. Morphological Studies, Life Cycle and Applied Research on Hoverflies: an Approach from the Tribes Merodontini and Eristalini (Diptera: Syrphidae, Eristalinae). 349 pp. Ph.D. Thesis, Universidad de Alicante, Departamento de Ciencias Ambientales y Recursos Naturales, Alicante.
Hadrava, J., Nidergas, V., Dankova, K., Pecharova, M., Nel, A., and Prokop, J. 2020. Blera miocenica: a new species of Early Miocene hoverfly (Diptera: Syrphidae) from the Czech Republic and its palaeoenvironmental significance. Insect Systematics & Evolution 51: 811–819. Crossref
Hull, F.M. 1949. The morphology and inter-relationship of the genera of syrphid flies, recent and fossil. Transactions of the Zoological Society of London 26: 257–408. Crossref
Hurkmans, W. 1993. A monograph of Merodon (Diptera: Syrphidae). Part 1. Tijdschrift voor Entomologie 136: 147–234.
Kassebeer, C.F., Maibach, A., and Rotheray, G.E. 1998. The third (= final) stage larva of Psilota anthracina Meigen and Psilota decessa (Hutton) (Diptera, Syrphidae). Entomologist’s Monthly Magazine 134: 39–43.
Liu, X., Zhao, L., and Huo, K. 2022. A new species in the genus Azpeytia Walker (Diptera: Syrphidae) from China. Entomotaxonomia 44: 144–148.
Mengual, X., Ståhls, G., and Rojo, S. 2015. Phylogenetic relationships and taxonomic ranking of pipizine flower flies (Diptera: Syrphidae) with implications for the evolution of aphidophagy. Cladistics 31: 491–508. Crossref
Mengual, X., Mayer, C., Burt, T.O., Moran, K.M., Dietz, L., Nottebrock, G., Pauli, T., Young, A.D., Brasseur, M.V., Kukowka, S., Kelso, S., Etzbauer, C., Bot, S., Hauser, M., Jordaens, K., Miranda, G.F.G. Ståhls, G., van Steenis, W., Peters, R.S., and Skevington, J.H. 2023. Systematics and evolution of predatory flower flies (Diptera: Syrphidae) based on exon-capture sequencing. Systematic Entomology 48 (2): 250–277. Crossref
Ménouret, B. 2014. Gisements paléontologiques à mammifères ou empreintes de pas de mammifères du Parc naturel régional du Luberon. Courrier scientifique du Parc naturel régional du Luberon et de la Réserve de biosphère Luberon-Lure 1: 56–74.
Morales, M.N., Massardo, D., Moreira, G.R.P., and Thompson, F.C. 2009. A new species of Alipumilio Shannon (Diptera, Syrphidae) found in association with the exudate resin of Schinus terebinthifolius Raddi (Anacardiaceae). Zootaxa 2112: 53–64. Crossref
Moran, K.M., Skevington, J.H., Kelso, S., Mengual, X., Jordaens, K., Young, A.D., Ståhls, G., Mutin, V., Bot, S., Van Zuijen, M., Ichige, K., Van Steenis, J., Hauser, M., and Van Steenis, W. 2022. A multigene phylogeny of the eristaline flower flies (Diptera: Syrphidae), with emphasis on the subtribe Criorhinina. Zoological Journal of the Linnean Society 194: 120–135. Crossref
Nel, A., Wei, Meicai, Niu, Gengyun, Coster, P., Boderau, M., Josse, H., Kundura, J.-P., Kundura, M.-H., Brisac, P., Boudet, L., and Jouault, C. 2024. The first sawfly from the Oligocene of Céreste (Southern France) (Hymenoptera: Tenthredinidae). European Journal of Taxonomy 917: 1–18. Crossref
Nidergas, V., Hadrava, J., Garrouste, R., Prokop, J., Schubnel, T., and Nel, A. 2018. The first pipizine hoverfly genus and species from the Oligocene of France (Diptera, Syrphidae). Acta Palaeontologica Polonica 63: 539–548. Crossref
Orengo-Green, J.J., Ricarte, A., Hauser, M., Dominique Langlois, D., and Marcos-García, M.A. 2024. On the immature stages of some Merodontini hoverflies (Diptera: Syrphidae) from Europe and Africa. Arthropod Structure & Development 78: 101328. Crossref
Reemer, M. and Ståhls, G. 2013. Generic revision and species classifications of the Microdontinae (Diptera, Syrphidae). ZooKeys 288: 1–213. Crossref
Rotheray, G.E., Marcos-Garcia, M.A., Hancock, E.G., and Gilbert, F.S. 2000. The systematic position of Alipumilio and Nausigaster based on early stages (Diptera, Syrphidae). Studia Dipterologica 7: 133–144.
Speight, M.C.D. and Langlois, D. 2020. Keys to the males of Merodon species known from France, 2020 (Diptera: Syrphidae). Syrph the Net, the database of European Syrphidae 111: 1–60.
Speight, M.C.D., Fisler, L., Pétremand, G., and Hauser, M. 2021. A key to the males of the Eumerus species known from Switzerland & surrounding parts of central Europe (Diptera: Syrphidae). Syrph the Net, the database of European Syrphidae 112: 1–36.
Ssymank, A., Jordaens, K., De Meyer, M., Reemer, M., and Rotheray, G.E. 2021. Syrphidae (flower flies or hoverflies). In: A.H. Kirk-Spriggs and B.J. Sinclair (eds.), Manual of Afrotropical Diptera. Volume 3. Brachycera–Cyclorrhapha, Excluding Calyptratae, 1439–1491. South African National Biodiversity Institute, Pretoria.
Thompson, F.C. 1972. A contribution to a generic revision of the Neotropical Milesinae (Diptera: Syrphidae). Arquivos de Zoologia 23: 73–215.
Thompson, F.C. 1991. The flower fly genus Ornidia (Diptera: Syrphidae). Proceedings of the Entomological Society of Washington 93: 248–261.
Thompson, F.C. 1999. A key to the genera of the flower flies (Diptera: Syrphidae) of the Neotropical region including descriptions of new genera and species and a glossary of taxonomic terms. Contributions on Entomology, International 3: 321–378.
Thompson, F.C. 2007. A new Costa Rican flower fly (Diptera: Syrphidae) and a replacement name for a Neotropical flower fly genus. Studia Dipterologica 14: 167–170.
Thompson, F.C. 2008. A conspectus of New Zealand flower flies (Diptera: Syrphidae) with the description of a new genus and species. Zootaxa 1716: 1–20. Crossref
Van Steenis, J., Ricarte, A., Vujić, A., Birtele, D., and Speight, M.C.D. 2016. Revision of the West-Palaearctic species of the tribe Cerioidini (Diptera, Syrphidae). Zootaxa 4196: 151–209. Crossref
Vockeroth, J.R. 1964. The genus Alipumilio Shannon (Diptera: Syrphidae). The Canadian Entomologist 96: 922–924. Crossref
Vockeroth, J.R. and Thompson, F.C. 1981. Chapter 52. Syrphidae. In: J.F. McAlpine, B.V. Peterson, G.E. Shewell, H.J. Teskey, J.R. Vockeroth, and D.M. Wood (coordinators), Manual of Nearctic Diptera. Volume 2, 713–743. Research Branch, Agriculture Canada Monograph, Ottawa.
Vujić, A., Radenkovic, S., and Likov, L. 2018. Revision of the Palaearctic species of the Merodon desuturinus group (Diptera, Syrphidae). ZooKeys 771: 105–138. Crossref
Vujić, A., Radenković, S., Likov, L., Gorše, I., Djan, M., Ristić, Z.M., and Barkalov, A.V. 2022. Three new species of the Merodon ruficornis group (Diptera: Syrphidae) discovered at the edge of its range. Zootaxa 5182: 301–347. Crossref
Vujić, A., Radenković, S., Zorić, L., Likov, L., Tot, T., Veselić, S., and Djan, M. 2021. Revision of the Merodon bombiformis group (Diptera: Syrphidae)—rare and endemic African hoverflies European Journal of Taxonomy 755: 88–135. Crossref
Williston, S.W. 1883. On the North American Asilidae (Dasypogoninae, Laphrinae), with a new genus of Syrphidae. Transactions of the American Entomological Society 11: 1–35. Crossref
Wong, D., Norman, H., Creedy, T.J., Jordaens, K., Moran, K.M., Young, A., Mengual, X., Skevington, J.H., and Vogler, A.P. 2023. The phylogeny and evolutionary ecology of hoverflies (Diptera: Syrphidae) inferred from mitochondrial genomes. Molecular Phylogenetics and Evolution 184: 107759. Crossref
Wu, G., Li, H., Mengual, X., Wang, Y., and Zhao, R. 2024. Phylogenetic analysis of Syrphidae (Insecta, Diptera) based on low-coverage whole genomes. Insect Systematics and Diversity 114 (2): 1–13. Crossref
Young, A.D., Lemmon, A.R., Skevington, J.H., Mengual, X., Ståhls, G., Reemer, M., Jordaens, K., Kelso, S., Lemmon, E.M., Hauser, M., De Meyer, M., Misof, B., and Wiegmann, B.M. 2016. Anchored enrichment dataset for true flies (order Diptera) reveals insights into the phylogeny of flower flies (family Syrphidae). BMC Evolutionary Biology 16: art. 143. Crossref
Young, A.D., Skevington, J.H., and Van Steenis, W. 2020. Revision of the Psilota Meigen, 1822 flower flies (Diptera: Syrphidae) of Australia. Zootaxa 4737: 1–126. Crossref
Acta Palaeontol. Pol. 70 (4): 811–816, 2025
https://doi.org/10.4202/app.01255.2025