

New Miocene limoniid craneflies from Dominican amber and their evolutionary importance
KATARZYNA KOPEĆ, IWONA KANIA-KŁOSOK, ANDREW ROSS, AGNIESZKA SOSZYŃSKA, and WIESŁAW KRZEMIŃSKI
Kopeć, K., Kania-Kłosok, I., Ross, A., Soszyńska, A., and Krzemiński, W. 2026. New Miocene limoniid craneflies from Dominican amber and their evolutionary importance. Acta Palaeontologica Polonica 71 (1): 119–132.
This paper describes three new species belonging to the genus Styringomyia, based on specimens preserved in Early Miocene (Burdigalian) Dominican amber: Styringomyia caridadi sp. nov., S. caribeana sp. nov., and S. grimaldii sp. nov. Previously, only five extinct species of Styringomyia were known, including two from Dominican amber. These new discoveries increase the total number of species known from fossils to eight. The genus Styringomyia is morphologically intriguing, characterized by the highly complex structure of the male and female terminalia, particularly the morphology of the gonostyles. The hypopygial features of the five Dominican amber species resemble those of certain extant Styringomyia species found in Australia. These findings contribute to our broader understanding of cranefly diversity and significantly enhance our knowledge of Miocene Styringomyia and this stage of evolution of the genus. Notably, despite the abundance of Styringomyia fossils in Dominican amber, the genus is not known to inhabit the island of Hispaniola today.
Key words: Diptera, Limoniidae, Chioneinae, fossil insects, taxonomy, evolution, Miocene, Dominican Republic, Hispaniola.
Katarzyna Kopeć [kopeck1981@gmail.com; ORCID: https://orcid.org/0000-0001-6449-3412] and Wiesław Krzemiński [wieslawk4@gmail.com; ORCID: https://orcid.org/0000-0001-5685-891X], Institute of Systematics and Evolution of Animals, Polish Academy of Sciences, Sławkowska 17, 31–016 Kraków, Poland.
Iwona Kania-Kłosok [ikania@ur.edu.pl; ORCID: https://orcid.org/0000-0002-2325-4308] (corresponding author), Faculty of Biology, Nature Protection and Sustainable Development, University of Rzeszów, Zelwerowicza 4, 35–601 Rzeszów, Poland.
Andrew Ross [A.Ross@nms.ac.uk; ORCID: https://orcid.org/0000-0003-2751-9091] Department of Natural Sciences, National Museums Scotland, Chambers St., Edinburgh EH1 1JF, UK.
Agnieszka Soszyńska [agnieszka.soszynska@biol.uni.lodz.pl; ORCID: https://orcid.org/0000-0002-2661-6685], University of Lodz, Faculty of Biology and Environmental Protection, Department of Invertebrate Zoology and Hydrobiology, Łódź, Poland.
Received 6 November 2025, accepted 8 January 2026, published online 26 March 2026.
Copyright © 2026 K. Kopeć et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The cranefly Styringomyia Loew, 1845, belongs to the family Limoniidae in the infraorder Tipulomorpha of the order Diptera. The Tipulomorpha includes the extant families Limoniidae, Pedicidae, Tipulidae, Cylindrotomidae, and Trichoceridae, as well as the extinct families Archilimoniidae and Psychotipidae. The latter two families were included in Tipulomorpha by Krzemiński and Krzemińska (2003), and all six families were assigned to the suborder Polyneura. Morphologically, Styringomyia is particularly distinctive differing significantly from most other Limoniidae in wing venation, the structure of the male hypopygium, and the female copulatory organs. The genus comprises 176 described extant species distributed across the Neotropical, Ethiopian, Australian, and Palearctic regions, but it has not been recorded in the Nearctic region. The first fossil representatives of this genus were discovered by Hermann Loew preserved in East African copal and Baltic amber (Loew 1845, 1850). The first extant species, Styringomyia didyma Grimshaw, 1901, was described half a century later from Hawaii. Detailed information on the history of discoveries and research on this fascinating genus was provided by Edwards (1914) and Ribeiro (2003). A total of six fossil species have been described so far, although one was subsequently synonymized: Styringomyia venusta Loew, 1845, from East African copal, S. gracilis Loew, 1850, from Baltic amber, S. concinna (Cockerell, 1917) (=S. extensa Cockerell & Haines, 1921, see Krzemiński et al. 2019) from the Bembridge Marls of the Isle of Wight, S. dominicana Podenas & Poinar, 1999, from Dominican amber and S. optiva Podenas & Poinar, 2001, also from Dominican amber (Table 1). These, along with the three new species described here, demonstrate that Dominican amber has yielded the most diverse known fauna of extinct Styringomyia species (Arillo and Ortuño 2005).
Table 1. List of fossil species of Styringomyia, including their location, preservation material, and age.
|
Species |
Locality |
Type of material |
Age |
|
Styringomyia venusta Loew, 1845 |
East Africa |
African copal inclusion |
Quaternary |
|
Styringomyia caridadi sp. nov. |
Dominican Republic |
Dominican amber inclusion |
Miocene: Burdigalian |
|
Styringomyia caribeana sp. nov. |
Dominican Republic |
Dominican amber inclusion |
Miocene: Burdigalian |
|
Styringomyia dominicana Podenas & Poinar, 1999 |
Dominican Republic |
Dominican amber inclusion |
Miocene: Burdigalian |
|
Styringomyia grimaldii sp. nov. |
Dominican Republic |
Dominican amber inclusion |
Miocene: Burdigalian |
|
Styringomyia optiva Podenas & Poinar, 2001 |
Dominican Republic |
Dominican amber inclusion |
Miocene: Burdigalian |
|
Styringomyia concinna (Cockerell, 1917) (=Styringomyia extensa Cockerell & Haines, 1921) |
England: Isle of Wight |
compression fossil |
Eocene: Priabonian |
|
Styringomyia gracilis Loew, 1850 |
Baltic region |
Baltic amber inclusion |
Eocene: Priabonian |
Dominican amber is mined in the Dominican Republic on the island of Hispaniola and is considered to be of Early Miocene (Burdigalian) age (Iturralde-Vinent and MacPhee 1996; Penney 2010; Grimaldi and Ross 2017). This stage ranges 20.45–15.98 Mya (International Chronostratigraphic Chart v.2024/12, https://stratigraphy.org/chart). The resin that fossilized into Dominican amber was produced by trees belonging to an extinct species of the genus Hymenaea (Seyfullah et al. 2018). A total of eight Styringomyia specimens preserved in Dominican amber are now known (Table 2). The new discoveries presented here enhance our understanding of the diversity and evolution history of these flies during the Miocene as well as shed light on the possible course of evolution of this group. Investigation of the morphological similarities between fossil representatives of the genus and extant species currently found in Australia may help clarify both the evolution pathways of this group and the development of its current geographical distribution.
Table 2. List of specimens of species known from Dominican amber.
|
Number |
Sex |
Type |
Collection |
Species |
|
NMS G.2019.6.3 |
male |
holotype |
NMS |
Styringomyia caridadi sp. nov. |
|
AMNH 11833 |
male |
paratype |
AMNH, coll. J. Brodzinsky |
Styringomyia caridadi sp. nov. |
|
MP/5259 |
male |
holotype |
ISEA PAS, coll. K. Kopeć |
Styringomyia caribeana sp. nov. |
|
REG # 10937 |
male |
paratype |
SI, coll. J. Brodzinsky/M. Lopez-Penha |
Styringomyia caribeana sp. nov. |
|
D-7-202 |
male |
holotype |
G.O. Poinar coll. |
Styringomyia dominicana Podenas & Poinar, 1999 |
|
D-7-202A |
male |
paratype |
G.O. Poinar coll. |
Styringomyia dominicana Podenas & Poinar, 1999 |
|
AMNH DR-8-73 |
male |
holotype |
AMNH |
Styringomyia grimaldii sp. nov. |
|
D-7-39H |
male |
holotype |
G.O. Poinar coll. |
Styringomyia optiva Podenas & Poinar, 2001 |
Institutional abbreviations.—AMNH, American Museum of Natural History, New York City, USA; ISEA PAS MP, Natural History Museum of the Institute of Systematics and Evolution of Animals, PAS, Kraków, Poland; NMS, National Museums Scotland, Edinburgh, Scotland, UK; SI, Smithsonian Institution, Washington DC, USA.
Other abbreviations.—A1, A2, anal veins; C, costal vein; Cu, cubital vein; d, discal cell; h, humeral vein; M1+2–M4, medial veins; Mb, medial-basal vein; m-cu, medial-cubital crossvein; R1–R5, radial veins; r-m, radial-medial crossvein; Rs, radial sector; Sc, subcostal vein; sc-r, subcostal radial crossvein; I–IV, palpomeres 1–4.
Nomenclatural acts.— The electronic edition of this article conforms to the requirements of the amended International Code of Zoological Nomenclature, and hence the new names contained herein are available under that Code from the electronic edition of this article. This published work and the nomenclatural acts it contains have been registered in ZooBank. The LSID for this publication is: LSID urn:lsid:zoobank.org:pub:8BBAE62B-D78A-430F-8A7B-B66A264454DF.
Material and methods
The five specimens studied herein (the representatives of extinct species) are all preserved as inclusions in Dominican amber. Two specimens are housed in the collections of AMNH; one in SI; one in NMS; and one in ISEA PAS.
The specimens were examined with a Nikon SMZ 1500 stereomicroscope equipped with a Nikon DS–Fi1 camera and measured with NIS–Elements D 3.0 software, as well as a Nikon SMZ25 stereomicroscope equipped with a Nikon DS-Ri2 digital camera. Measurements were taken only from undamaged structures. Drawings were created by tracing the specimen and photographs, prepared by IKK. Descriptive terminology follows McAlpine et al. (1981), Ribeiro (2003) and Podenas (2011).
Systematic palaeontology
Order Diptera Linnaeus, 1758
Infraorder Tipulomorpha Rohdendorf, 1961
Family Limoniidae Speiser, 1909
Subfamily Chioneinae Rondani, 1841
Genus Styringomyia Loew, 1845
Type species: Styringomyia venusta Loew, 1845, monotypic, Dominican amber, Miocene.
Styringomyia caridadi sp. nov.
Figs. 1–3.
ZooBank LSID: urn:lsid:zoobank.org:act:EF125B23-6307-4E88-AADD-8C6315DD9B5C.
Etymology: Dedicated to Jorge Caridad, Director of the Amber World Museum, in recognition of his donation of the holotype to National Museums Scotland.
Type material: Holotype: NMS G.2019.6.3, male, well preserved male imago lacking part of legs. Paratype: AMNH 11833, male, coll. Jacob Brodzinsky, an inclusion in Dominican amber, well preserved male imago lacking part of legs.
Type locality: Dominican amber, Dominican Republic.
Type horizon: Lower Miocene (Burdigalian).
Material.—Type material only.
Diagnosis.—Pedicel massive, bulbous, 1.5 × as long as wide; last palpomere longer than penultimate one; wing without petiole; r-m elongate, as long as basal section of R4+5; d-cell elongate, 5.5 × as long as wide; m-cu in 0.16 × of d-cell length from fork of Mb; hypopygium slender, narrow, 2 × as long as wide, basal part of gonocoxite broad, approximately 2 × as long as wide, distal part of gonocoxite narrow, elongate, approximately 1.5 × as long as its basal part; gonocoxite with two very elongate terminal bristle (b) at apex; four apical extensions of gonostylus present: extension I, tiny, not very elongate; extension II, narrow, angled toward the internal part of hypopygium, ending with a long, strongly sclerotized spine; extension III, narrowed and slightly bent at the apex toward the inner part of the hypopygium, strongly sinusoidal; extension IV, middle part widened, significantly broader than its distal part, slightly sinusoidal, apical part forming an almost right angle with the basal part with a narrowed, sharp apex; tergite IX elongate, approximately 2 × as long as wide, broad, and truncate at apex; sternite X triangular, tapering towards the tip.
Description.—Body (Fig. 1A1, 1B1) 6.8 mm long (holotype), 6.9 mm long (paratype), pale brown, wings without a color pattern, pterostigma absent.
Head (Fig. 1A1, A2, A5): antenna (Figs. 1A2, A5, 2A2) 1.7 mm long (1/0.2; 2/0.1; 3/0.1; 4–16/0.1) in both holotype and paratype; longer than head but shorter than head and thorax combined; longer than palpus; scape narrow, elongate, cylindrical, 1.5 × as long as wide; pedicel massive, spherical, much shorter than scapus, 1.5 × as long as wide, flagellomeres elongate, ovoid, approximately 2 × as long as wide, became more slender to the apex of antenna; last flagellomere as long as penultimate one, with two not very elongate setae at apex, flagellomeres with a few elongate setae, six of them very elongate, longer than segment bearing them; palpus (Figs. 1A2, A5; 2A3) 0.4 mm long (1–4/0.1) (holotype), 0.6 mm long (1/0.2; 2–4/0.1) (paratype), palpomeres elongate, narrow, first palpomere longer than the rest, palpomeres 2–3 of equal length, approximately 2 × as long as wide, palpomeres with few not very elongate setae, not longer than segments bearing them.
Thorax (Fig. 1A1, B1): Wing (Figs. 1A1, A6, B1, 2A1) 4.6 mm long, 1.0 mm wide (holotype), 4.7 mm long, 1.0 mm wide (paratype), 4.5 × as long as wide; Rs almost straight, slightly arched basally, elongate, shorter than R4+5, 3 × as long as R2+3, R1 ending just before fork of Rb; R4+5 2 × as long as Rs; M3 approximately 1.2 × the length of d-cell; d-cell 1.1 mm long (holotype and paratype); tip of Cu beyond fork of M3+4 on M3 and M4, in approximately 0.3 × of M4; A1 and A2 slightly waved, tip of A1 just beyond level of R2+3, tip of A1 in approximately 0.5 × of Mb.

Fig. 1. Limoniid cranefly Styringomyia caridadi sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). A. Holotype NMS G.2019.6.3, male; body in ventral view (A1), palpi in ventral view (A2), hypopygium in dorsal (A3) and ventral (A4) views; antenna (A5), wing (A6). B. Paratype AMNH 11833, male; body in ventral view.
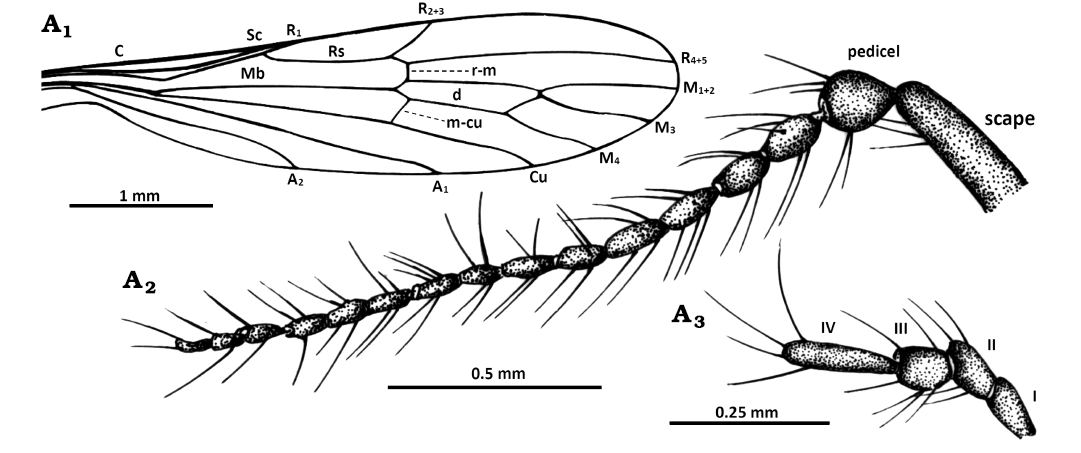
Fig. 2. Limoniid cranefly Styringomyia caridadi sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). Holotype NMS G.2019.6.3, male; wing (A1), antenna (A2), and palpus (A3). Abbreviations: A1, A2, anal veins; C, costal vein; Cu, cubital vein; d, discal cell; M1+2–M4, medial veins; Mb, medial-basal vein; m-cu, medial-cubital crossvein; R1–R5, radial veins; r-m, radial-medial crossvein; Rs, radial sector; Sc, subcostal vein; I–IV, palpomeres 1–4.
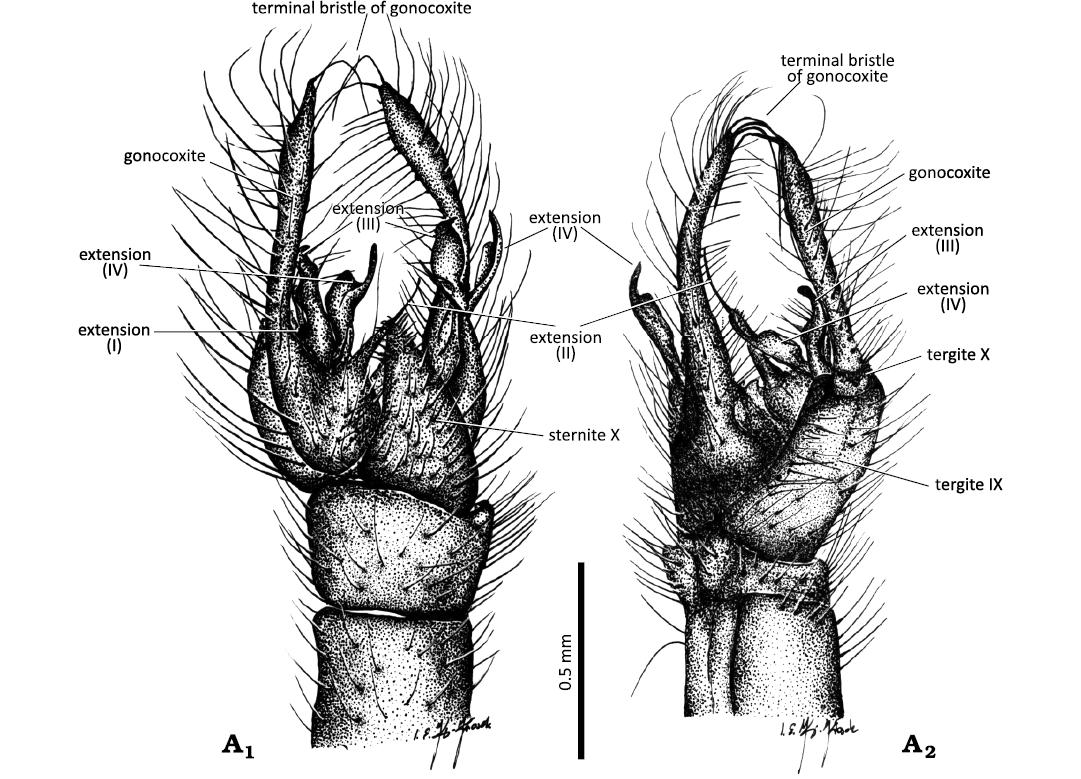
Fig. 3. Limoniid cranefly Styringomyia caridadi sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). Holotype NMS G.2019.6.3, male; hypopygium in latero-dorsal (A1) and latero-ventral (A2) views.
Abdomen (Fig. 1A1, A3, A4, B1): Hypopygium pale brown, with a brighter distal part of gonocoxite (Fig. 1A3, A4) 0.9 mm long (holotype), 0.7 mm long (paratype), elongate and slender, constituting approximately 0.2 × the body length; gonocoxite 0.8 mm long (holotype), 0.6 mm long (paratype), basal part approximately 2 × as long as wide; four apical extensions of gonostylus, aedeagus tubular, relatively narrow, interbases slightly elongate, narrow, angled toward the internal part of hypopygium, sternite X almost triangular in shape, gradually narrowing from base to tip, tergite IX broad, truncated at the apex.
Remarks.—Styringomyia caridadi sp. nov. is most similar to S. caribeana sp. nov.; however, in S. caridadi, the petiole is absent, whereas in S. caribeana it is elongate, as long as 0.15 × the length of M3. Additionally, the pedicel of S. caridadi is massive and bulbous, while in S. caribeana it is elongate and cylindrical. In S. caridadi, the d-cell is 5.5 × as long as wide, and the m-cu is situated at 0.16 × the length of d-cell from the fork of Mb; in contrast, in S. caribeana, the d-cell is 7 × as long as wide, with the m-cu located at 0.25 × the d-cell length from the fork of Mb. Sternite X in S. caridadi is broad and truncate at the apex, whereas in S. caribeana it is rounded appically. S. caridadi also lacks of a petiole, similar to S. grimaldii sp. nov., but differs in that the r-m vein in S. caridadi is well developed and as long as the basal section of R4+5, while in S. grimaldii is nearly reduced. However, the most distinctive differences between these two species are visible in the morphology of the hypopygium. In S. caridadi, the hypopygium is relatively narrow and slender, while in S. grimaldii it is massive and broad. The gonocoxite of S. caridadi is elongate, with a very narrow distal portion bearing two long terminal bristles (b) at the apex. It also features four apical extensions of the gonostylus, with extension III narrowed, slightly bent apically toward the inner side of the hypopygium, and strongly sinusoidal in shape. The tergite IX of S. caridadi is elongate, approximately 2 × as long as wide, and truncate at apex; sternite X is triangular, tapering toward the tip. In S. grimaldii, the gonocoxite is less elongate, lacks the terminal bristle (b) at the apex, and extension III is flattened with an outward-pointing tip forming a right angle to the longitudinal axis of the gonostylus. Tergite IX is also elongate (2 × as long as wide), but slightly rounded at the apex; the sternite is broad at the base, tapering strongly toward the tip to form a narrow, tonque-shaped structure.
In the two species described from Dominican amber by Podenas and Poinar (1999, 2001), S. dominicana and S. optiva, only one elongate terminal bristle (b) is present at the apex of the gonocoxite, whereas two are present in S. caridadi and S. grimaldii. Moreover, the hypopygium of S. optiva is quite distinct; it is rather short and massive than slender and is characterized by a flattened intermediate arm (extension III) with an outward-pointing tip forming a right angle with the longitudinal axis of the gonostylus. The inner arm (extension IV) with a basal part that is broader than the apical portion, slightly sinusoidal in shape, with the apical part forming an almost right angle with the basal part and terminating in an acute tip (Podenas and Poinar 2001). In S. caridadi, the hypopygium is slender; the tip of extension III does not point outwards, although it is bent, but is direct inward, toward the inner part of the hypopygium. It is narrowed at the apex and strongly sinusoidal. Extension IV has a middle part wider than the apex, is slightly sinusoidal, and its apical part forms an almost right angle with the basal part, ending in a sharp, strongly narrowed apex. However, this narrowed and sharp apex is distinctly shorter than that of S. optiva.
Geographic and stratigraphic range— Dominican amber, Dominican Republic, Lower Miocene (Burdigalian).
Styringomyia caribeana sp. nov.
Figs. 4–6.
ZooBank LSID: urn:lsid:zoobank.org: act:D2692737-E9EF-49BE-9D95-BE323DE2A3EA.
Etymology: Derived from Caribbean.
Type material: Holotype: ISEA PAS MP/5259, male, coll. KK, well preserved male imago lacking part of legs. Paratype REG. #10937, male, inclusion in Dominican amber, coll. Jacob Brodzinsky/ Marianela (Mañeña) Lopez-Penha, deposited in the Smithsonian Institution, well preserved male imago lacking part of legs.
Type locality: Dominican amber, Dominican Republic.
Type horizon: Lower Miocene (Burdigalian).
Material.—Type material only.
Diagnosis.—Pedicel elongate, cylindrical, 1.5 × as long as wide; last palpomere longer than penultimate one; wing with petiole; petiole approximately 0.15 × the length of M3; vein r-m elongate, as long as basal section of R4+5; d-cell elongate, 7 × as long as wide; crossvein m-cu located at 0.25 × the length of d-cell from fork of Mb; hypopygium slender and narrow, 2 × as long as wide; basal part of gonocoxite wide, approximately 2 × as long as wide, distal part narrow and elongate, approximately 1.5 × as long as the basal part; gonocoxite with two very elongate terminal bristle (b) at apex; four apical extensions of gonostylus present; extension I small, not elongate; extension II narrow, directed toward the internal part of hypopygium, ending in a long, strongly sclerotized spine; extension III narrowed and slightly bent at the apex toward the inner part of the hypopygium, strongly sinusoidal; extension IV with a widened middle part, much wider than its distal part, slightly sinusoidal, with the apical part forming an angle with the basal part and in a narrowed, sharp apex; tergite IX elongate, approximately 2 × as long as wide, rounded at the apex, with strongly sclerotized small spine-shaped structure at the tip; sternite X triangular, with strongly tapered tip.
Description.—Body (Fig. 4A1, B1) 8.2 mm long (holotype), 7.4 mm long (paratype), pale brown, wings without color pattern, pterostigma absent.
Head (Fig. 4A1, B1): Antenna (Fig. 4A1, A2, B1, 5A2) 1.7 mm long (holotype and paratype) (1/0.2; 2–16/0.1); longer than head and palpus; scape massive, elongate, widened distally, 2.5 × as long as wide, longer than pedicel; pedicel spherical 1.5 × as long as wide; flagellomeres elongate, ovoid, approximately 3 × as long as wide, becoming more slender towards the apex of antenna; last flagellomere as long as penultimate one, with three moderately elongate setae at apex, each flagellomeres bears four elongate setae, slightly longer than the segment itself; palpus (Figs. 4A4, 5A3) not very elongate 0.4 mm long (holotype) (1–4/0.1), 0.8 mm long (paratype) (1–4/0.2), palpomeres elongate, almost cylindrical, palpomeres 1–3 of comparable length, with the last palpomere longer than the penultimate one; all palpomeres bears several elongate setae, not exceeding the length of the segments bearing them.
Thorax (Fig. 4A1, B1): Wing (Figs. 4A1, A3, B1, B2, 5A1) 5.2 mm long, 1.0 mm wide (holotype), 5.0 mm long, 0.7 mm wide (paratype), approximately 5–7 × as long as wide; vein Rs almost straight, slightly arched basally, elongate, shorter than R4+5 but 3 × longer than R2+3, vein R1 ends beyond fork of Rs; vein M3 shorter than d-cell; d-cell 1.11 mm long (holotype), approximately 7 ×as long as wide; tip of M4 reaches well beyond petiole and passes the halfway point of R4+5; tip of A2 before fork of Rb.
Abdomen (Figs. 3A1, A2; 4A1, A5, A6, B1): Hypopygium pale brown (Figs. 4A1, A5, A6, B1, 6A1, A2) 1.0 mm long, with gonocoxite 0.9 mm long (both holotype and paratype), slender, elongate, comprising approximately 0.1 × of the total body length; gonocoxite elongate and small; gonostylus with four extensions.

Fig. 4. Limoniid cranefly Styringomyia caribeana sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). A. Holotype ISEA PAS MP/5259, male; body in lateral view (A1), antenna (A2), wing (A3), palpus (A4), hypopygium in latero-dorsal (A5) and latero-ventral (A6) views. B. Paratype AMNH 10937, male; body in latero-ventral view (B1), wing (B2).
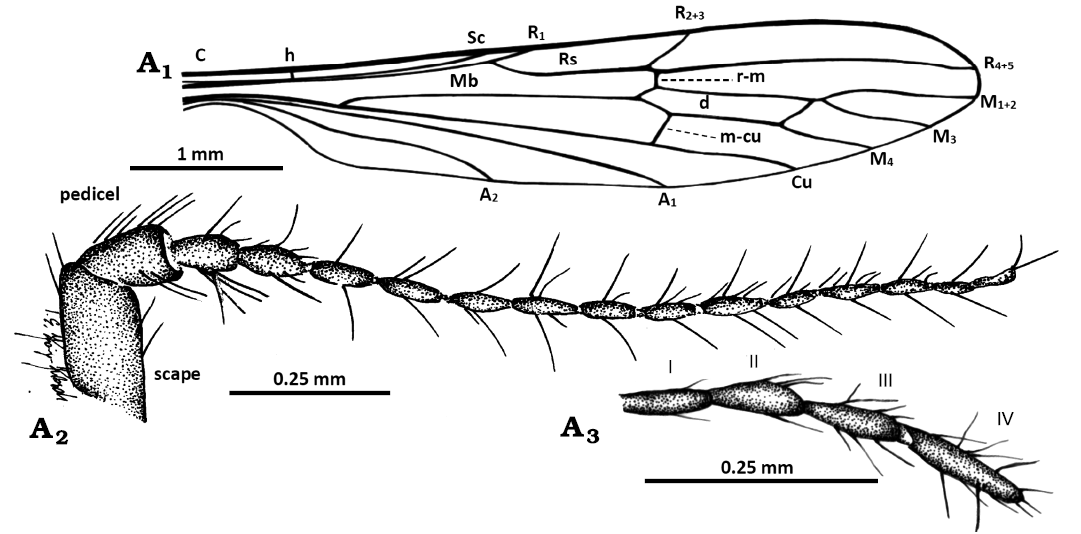
Fig. 5. Limoniid cranefly Styringomyia caribeana sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). Holotype ISEA PAS MP/5259, male; wing (A1), antenna (A2), and palpus (A3). Abbreviations: A1, A2, anal veins; C, costal vein; Cu, cubital vein; d, discal cell; h, humeral vein; M1+2–M4, medial veins; Mb, medial-basal vein; m-cu, medial-cubital crossvein; R1–R5, radial veins; r-m, radial-medial crossvein; Rs, radial sector; Sc, subcostal vein; I–IV, palpomeres 1–4.
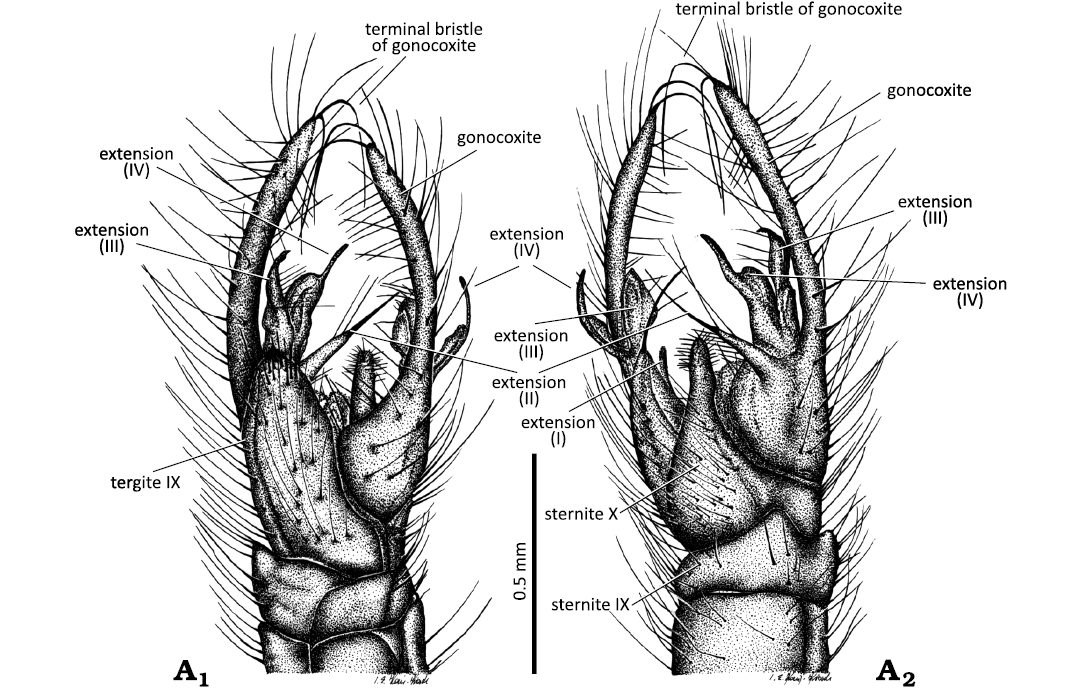
Fig. 6. Limoniid cranefly Styringomyia caribeana sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). Holotype ISEA PAS MP/5259, male; hypopygium in latero-ventral (A1) and latero-dorsal (A2) views.
Remarks.—Styringomyia caribeana sp. nov. differs from the species described by Podenas and Poinar (1999, 2001), as both S. dominicana and S. optiva possess only one elongate terminal bristle (b) at the apex of the gonocoxite, wheras two such bristles are present in S. caribeana. The hypopygium of S. optiva is relatively short and massive, and is characterized by a flattened intermediate arm (extension III) with an outward-pointing tip forming a right angle with the longitudinal axis of the gonostylus. The inner arm (extension IV) has a basal part broader than the apical one, is slightly sinusoidal, and its apical part forms an angle with the basal part, ending in anacute tip (Podenas and Poinar 2001). In S. caribeana, the hypopygium is slender. The tip of extension III is bent inward, toward the inner part of the hypopygium, and is narrowed at the apex, strongly sinusoidal. Extension IV is characterized by a middle part wider than the apex, slightly sinusoidal, with the apical part forming an angle with the basal part, and ending in a sharp, strongly narrowed apex, which is distinctly shorter than in S. optiva. In S. grimaldii sp. nov., four apical extensions of the gonostylus are present, but the terminal bristle (b) at the apex of the gonocoxite is absent. In this species, extension I of the gonostylus is narrow and not very elongate, extension II is direct inward, toward the hypopygium, and ends with a short, triangular apical part bearig a thick, short spine at the tip, extension III is flattened with an outward-pointing tip forming a right angle with the longitudinal axis of the gonostylus, extension IV is short, narrowed at the middle, and ends with a very narrow, short, and strongly sclerotized tip. Styringomyia caribeana is most similar to S. caridadi sp. nov. (see the comparison with S. caridadi above).
Geographic and stratigraphic range— Dominican amber, Dominican Republic, Lower Miocene (Burdigalian).
Styringomyia grimaldii sp. nov.
Figs. 7, 8.
ZooBank LSID: urn:lsid:zoobank.org:act:6454CCE7-568B-4960-938A-AB06FF4958AE.
Etymology: Dedicated to the eminent scientist and entomologist David A. Grimaldi (AMNH).
Holotype: AMNH DR-8-73 (male), not well preserved male imago lacking head and thorax, but with preserved parts of palpi.
Type locality: Dominican amber, Dominican Republic.
Type horizon: Lower Miocene (Burdigalian).
Material.—Holotype only.
Diagnosis.—Last palpomere as long as penultimate one; wing without petiole, r-m short, almost completely reduced; d-cell elongate, 3.5 × as long as wide; m-cu at 0.2 × length from fork of Mb; hypopygium wide, as long as wide; basal part of gonocoxite massive, very short and wide, approximately 1.5 × as long as wide, distal part of gonocoxite elongate and narrow 1.5 × as long as its basal part; four apical extensions of gonostylus present, without terminal bristle (b) at the gonocoxite apex; extension I narrow, not very elongate; extension II angled inward, with a short, triangular apical part bearing a thick and short spine at apex, extension III flattened, with an outward-pointing tip forming a right angle with longitudinal axis of gonostylus; extension IV short, narrowed in the middle, with a very narrow, short, strongly sclerotized tip; tergite IX elongate, 2 × as long as wide, slightly rounded at the apex; sternite widened at the base, strongly tapering distally, forming a narrow tongue-shaped structure.
Description.—Body (Fig. 8A1) pale brown, wings without color pattern, pterostigma absent.
Head: Palpomeres almost the same length, not very elongate (Fig. 7A2).
Thorax: Wing (Figs. 7A1, 8A1, A3) 5.0 mm long, 1.0 mm wide, approximately 4.5 × as long as wide; Rs almost straight, slightly arched basally, elongate, shorter than R4+5, 4 × as long as R2+3, R1 ending at 0.3 × the length of Rs; R5 2 × as long as Rs; M3 approximately 1.3 × the length of d-cell; d-cell 1.11 mm long; tip of Cu just beyond the fork of M3+4 on M3 and M4.
Abdomen (Fig. 7A3 8A1, A3): Hypopygium pale brown, 0.7 mm long, with a brighter distal part of gonocoxite (Figs. 7A3, 8A1, A2) 0.5 mm long, massive, constitute approximately 0.2 × the body length; basal part of gonocoxite massive, 1.5 × as long as wide; aedeagus tubular, thick, massive, wider than gonostylus; interbases slightly elongate, thick, angled toward the external part of hypopygium.
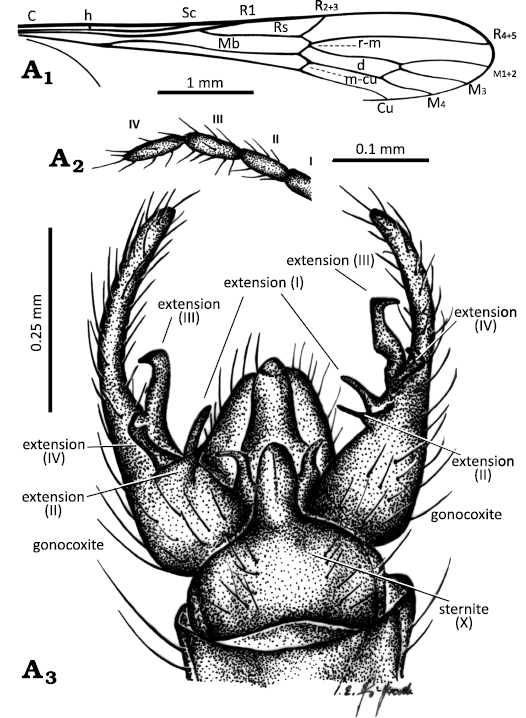
Fig. 7. Limoniid cranefly Styringomyia grimaldii sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). Holotype AMNH 11833, male; wing (A1), palpus (A2), hypopygium in dorsal view (A3). Abbreviations: C, costal vein; Cu, cubital vein; d, discal cell; h, humeral vein; M1+2–M4, medial veins; Mb, medial-basal vein; m-cu, medial-cubital crossvein; R1–R5, radial veins; r-m, radial-medial crossvein; Rs, radial sector; Sc, subcostal vein; I–IV, palpomeres 1–4.

Fig. 8. Limoniid cranefly Styringomyia grimaldii sp. nov. from Dominican amber, Dominican Republic, Lower Miocene (Burdigalian). Holotype AMNH 11833, male; body in latero-ventral view (A1), hypopygium in dorsal view (A2), wing (A3).
Remarks.—Styringomyia grimaldii sp. nov. is characterized by the absence of a petiole in the wing venation, similar to S. caridadi sp. nov., in which the petiole is also absent. In other species of Styringomyia from Dominican amber, the petiole is present. In S. grimaldii, vein r-m is almost completely reduced, whereas in S. caridadi this vein is well developed, approximately as long as the basal section of R4+5. However, the most important differences are visible in the morphology of the hypopygium. The hypopygium of S. grimaldii is massive, with a wide and relatively short basal part of gonocoxites and only moderately elongate distal parts In contrast, in other species of Styringomyia from Dominican amber, the hypopygium is slender and elongate, with narrow and elongate distal parts of the gonocoxites. The basal part of the gonocoxite of S. grimaldii is broadened, approximately 2 × as long as wide; the distal part is not strongly elongate, only slightly longer than the basal part.
Geographic and stratigraphic range— Dominican amber, Dominican Republic, Lower Miocene (Burdigalian).
Results
Key to Miocene species of Styringomyia
1. Gonocoxite with apical bristle …………………..……… 2.
– Gonocoxite without apical bristle …....S. grimaldii sp. nov.
2. Gonocoxite with one apical bristle …………………….. 3.
– Gonocoxite with two apical bristles…......……………… 4.
3. Hypopygium slender, elongate; extension III flattened, with a slightly bent tip; inner arm of extension IV with a basal part broader than the apical part, straight; apical part elongate, with a sharp tip……………..…………… .......................................S. dominicana Podenas & Poinar, 1999
– Hypopygium rather broad, not very elongate;
extension III flattened, with an outward-pointing tip forming a right
angle with the longitudinal axis of the gonostylus;
extension IV with a basal part broader than the apical
part, slightly sinusoidal; apical part forming an almost right angle
with the basal part, with an acute tip
...........................................................................
S. optiva Podenas
& Poinar, 2001
4. Pedicel massive, bulbous; wing without petiole; d-cell 5.5 × as long as wide; basal section of R5 elongate, as long as r-m; basal section of M3 (from the fork of M3+4 to the petiole) straight; tergite IX elongate, approximately 2 × as long as wide, truncate at apex; sternite X triangular, tapering towards the tip ………………………...… S. caridadi sp. nov.
– Pedicel elongate, cylindrical; wing with petiole; d-cell 7 × as long as wide; basal section of R5 very short, 0.5 × the length of r-m; basal section of M3 (from the fork of M3+4 to the petiole) oblique; tergite IX elongate, approximately 2 × as long as wide, rounded at the apex, with a strongly sclerotized, small spine-like structure at the tip; sternite X triangular with a strongly tapered tip ................................ S. caribeana sp. nov.
Discussion
The genus Styringomyia occupies a rather unique position within the family Limoniidae. Based on larval characteristics, it possesses a reduced head capsule compared to a more massive head capsule found in the lower Eriopterinae. Oosterbroek and Theowald (1991) placed Styringomyia at the base of the so-called higher clade Eriopterinae [= Chioneinae] (Theischinger et al. 2018). Among modern representatives, the majority of species are found in the Oriental region (72 species) and Afrotropical region (55 species), with additional presence in the Australian and Oceanian regions (41 species). The genus is less diverse in the Neotropical and Palearctic regions each with only eight species. Although Styringomyia is sparsely represented in the fossil record, with only eight known species (including those described herein), the oldest dating back to the late Eocene, the body morphology of the Miocene representatives preserved in Dominican amber shows greater similarity to extant Australian species. Theischinger et al. (2018) identified three closely related species groups occurring in Australia: S. bancrofti group (five species), S. terraereginae group (two species), and S. bipunctata group (three species). The remaining five Australian species were not assigned to any of these groups. In the S. bipunctata group (S. bipunctata Theischinger & Billingham, 2018; S. coronata Theischinger & Billingham, 2018, and S. williamsi Theischinger & Billingham, 2018), male gonocoxites are evenly curved, almost conical, with the basal half noticeably expanded and a rather slender apical spine. The gonostyli consist of a single, slightly curved element that is subbasally expanded, bearing setae and short spines (Theischinger et al. 2018). Males of the S. terraereginae group (S. terraereginae Theischinger & Billingham, 2018, and S. aterrima Theischinger & Billingham, 2018) are characterized by long, slender gonocoxites bearing a longer spine at about midlength, a distinctly shorter apical spine, and gonostyli composed of three elements, including a whip-like one. The S. bancrofti group (S. bancrofti Edwards, 1914; S. baroalba Theischinger & Billingham, 2018; S. collessi Theischinger & Billingham, 2018, and S. remex Theischinger & Billingham, 2018) is characterized by conical gonocoxites with a broad basal lobe and a thick apical spine. Their gonostyli bear several comb-shaped elements (Theischinger et al. 2018). A narrow, slender hypopygium with very elongated gonocoxites is a feature observed in Miocene Styringomyia species. The gonostylus structures are positioned closer to the base of the gonocoxite than to its apex, similar to the condition in modern Australian species. Another distinctive feature of most extinct species is the presence of one or two very elongated, narrow structures at the apex of the gonocoxite, resembling the long, slender spines seen in some modern Australian taxa.
Craneflies of the genus Styringomyia currently inhabit tropical and subtropical climate zones. However, research on the modern fauna has shown that this genus is not represented on the island of Hispaniola (including the Dominican Republic) today (Oosterbroek 2025). It is quite surprising that the eight species found in the Neotropical region today (Alexander 1945a, b, 1946; Ribeiro 2003) are morphologically very different from those known from the Miocene, preserved in Dominican amber. Although representatives of the genus are rare in the fossil record, morphological comparison indicates a closer relationship between the extinct Miocene species from Dominican amber to the modern fauna of Australia, rather than the modern fauna of South America. Extant Styringomyia species from the Neotropical region, described and revised by Ribeiro (2003), are characterized by relatively broad and short gonocoxites, approximately the same size as the gonostyli. In contrast, the Miocene species from Dominican amber possess very elongate and narrow gonocoxites. Even in S. grimaldii sp. nov., which has a short and massive hypopygium, this structure is significantly more elongate than in species known from the modern Neotropical fauna. Of the 15 species currently known from Australia, as many as five (S. aterrima, S. baroalba, S. bipunctata, S. coronate, and S. williamsi), have very elongate and narrow gonocoxites, similar to those observed in species preserved in Dominican amber. Other Australian species, such as S. collessi and S. remex, also show some morphological similarities in the structures of the hypopygium. While their gonocoxites are as elongate, their shape resembles that found in the Miocene species. The two elongate bristles found at the apex of the gonocoxites in S. caridadi sp. nov. and S. caribeana sp. nov. are likely homologous with the slender apical spine observed in the S. bipunctata group.
The evolution of this group of insects may have evolved a gradual reduction in the number of elongate bristles to a single, not very elongate spine at the apex of the gonocoxite, or even a complete loss of these structures. Over time, the shape of the gonocoxite likely evolved from a more elongate form, as seen in fossil representatives of the genus, to a shorter form, with the elements of the gonostylus shifted toward the apex of the gonocoxite. However, more fossil specimens and further research are needed to verify this hypothesis.
But, why has Styringomyia disappeared from the Caribbean? This question still remains unanswered, perhaps further research on the genus will clarify this issue.
Most species of this genus are currently found in the warmer regions of the world. Similarly, in the distant past, these insects most likely favored warm climatic conditions. This hypothesis is supported by the presence of their fossils resins deposits from present-day Central America and subfossil sites of Africa, as well as in Baltic amber (Fig. 9), dated to the Priabonian (Grimaldi and Ross 2017; Ross et al. 2024, in press).
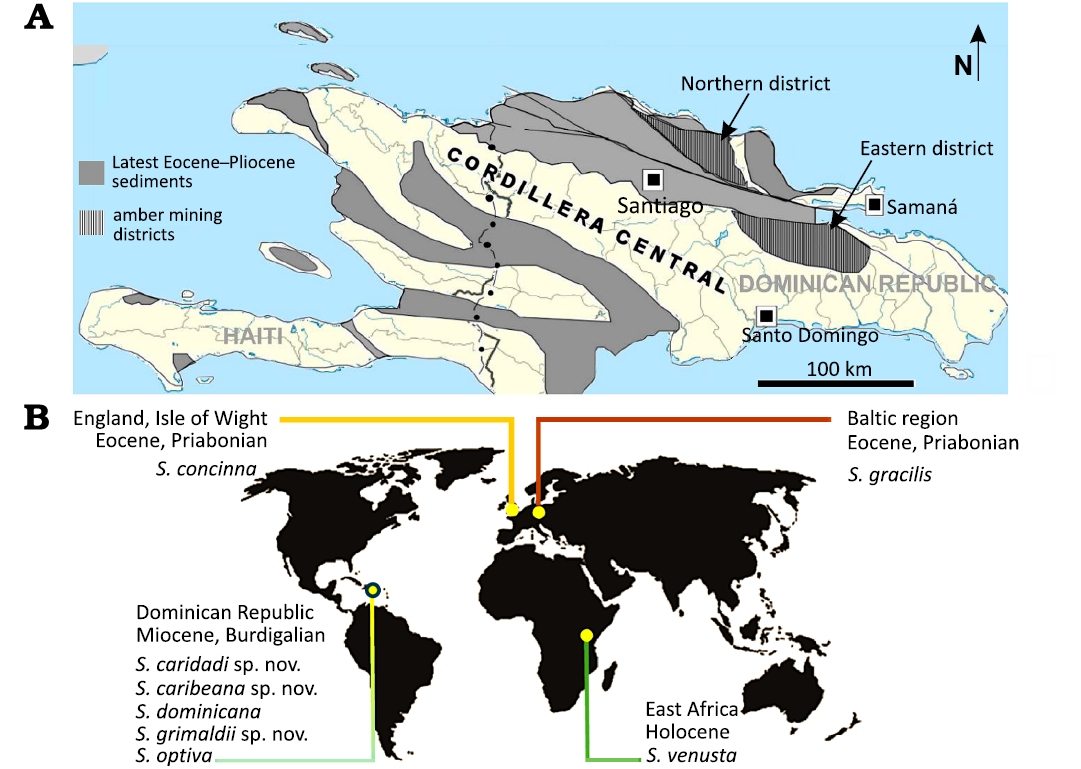
Fig. 9. A. Hispaniola map
indicating actual amber mining districts in Dominican Republic
(modified after Iturralde-Vinent and MacPhee 2019)
with
national and subnational borders according to
https://commons.wikimedia.org/wiki/File:Hispaniola_location_map.svg).
B. Localities where extinct species of Styringomyia have been found.
Conclusions
Morphological analysis of fossil members of Styringomyia from Miocene Dominican amber indicates their closer relationship to the modern Australian fauna than to the modern Neotropical fauna. Key features, such as the strongly elongated and narrow gonocoxites and the presence of slender apical structures, show significant similarities to the morphology of some modern Australian species, particularly the S. bipunctata group.
The fossil record suggests that the original hypopygium morphology in Styringomyia was characterized by more elongated gonocoxites and a more basal position of the gonostyle elements, whereas modern forms tend to shorten these structures and move them toward the apex of the gonocoxite. This may indicate directional evolutionary changes within the genus.
The absence of modern Styringomyia representatives in Dominican amber, despite their documented presence in the fossil record, suggests a regional extinction of the genus. The geographic distribution of both fossil and modern species indicates a preference for warm climates. Further research and the discovery of new fossil specimens are necessary to fully understand the evolutionary and biogeographic history of the genus Styringomyia.
Authors’ contributions
KK, conceived and designed the study, obtaining material, lead and performed the data analysis, interpretations and analysis of the material, writing and corrections of the manuscript; IKK, conceived and designed the study, lead and performed the data analysis, interpretations and analysis of the material, environmental analysis, writing and corrections of the manuscript, graphics; AR, obtaining material, corrections of the manuscript; AS, writing and corrections of the manuscript; WK, conceived and designed the study, obtaining material, analysis, writing and corrections of the manuscript. All authors reviewed the manuscript.
Disclosure statement: No potential conflict of interest was reported by the author(s).
Data availability: All data generated or analyzed during this study are included in this published article.
Acknowledgements
We would like to thank David A. Grimaldi (AMNH) for providing access to the material. This research was carried out as part of a project founded by the National Science Centre, Poland, Grant No. 2020/37/B/NZ8/03042. Funding: Open Access funding enabled and organized by Institute of Systematics and Evolution of Animals, Polish Academy of Sciences, Kraków, Poland.
Editor: Andrzej Kaim
References
Alexander, C.P. 1945a. New or little-known Tipulidae (Diptera). LXXIII. Neotropical species. Annals and Magazine of Natural History 12: 234–264. Crossref
Alexander, C.P. 1945b. New or little-known Tipulidae (Diptera). LXXIV. Neotropical species. Annals and Magazine of Natural History 12: 390–419. Crossref
Alexander, C.P. 1946. Undescribed species of Tipulidae from the State of Sao Paulo, Brasil (Diptera). Livro Homenagem R.F. d’Almeida, Almeida Museum 1: 1–10.
Arillo, A. and Ortuño, V.M. 2005 Catalogue of fossil insect species described from Dominican amber (Miocene). Stuttgarter Beiträge zur Naturkunde B 352: 1–68.
Cockerell, T.D.A. 1917. New Tertiary insects. Proceedings of the United States National Museum 52: 373–84. Crossref
Cockerell, T.D.A. and Haines, F.H. 1921. Fossil Tipulidae from the Oligocene of the Isle of Wight, continued. Entomologist 54: 109–112.
Edwards, F.W. 1914. A revision of the tipulid genus Styringomyia Lw. Transactions of the Entomological Society of London 1914: 206–227. Crossref
Grimaldi, D.A. and Ross, A.J. 2017. Extraordinary Lagerstätten in Amber, with particular reference to the Cretaceous of Burma. In: N.C. Fraser and H.-D. Sues (eds.), Terrestrial Conservation Lagerstätten: Windows into the Evolution of Life on Land, 287–342. Dunedin Academic Press Ltd, Edinburgh Crossref
Grimshaw, P.H. 1901. Diptera. Fauna Hawaiiensis 3: 1–77.
Iturralde-Vinent, M.A. and MacPhee, R.D.E. 1996. Age and paleogeographical origin of Dominican amber. Science 273: 1850–1852. Crossref
Iturralde-Vinent, M.A.and MacPhee, R.D.E. 2019. Remarks on the age of Dominican amber. Palaeoentomology 2: 236–240. Crossref
Krzemiński, W. and Krzemińska, E. 2003. Triassic Diptera: review, revisions and descriptions. Acta Zoologica Cracoviensia 46 (Supplement: Fossil Insects): 153–184.
Krzemiński, W., Blagoderov, V., Azar, D., Lukashevich, E., Szadziewski, R., Wedmann, S., Nel, A., Collomb, F.M., Waller, A., and Nicholson, D.B. 2019. True flies (Insecta: Diptera) from the Late Eocene Insect Limestone (Bembridge Marls) of the Isle of Wight, England, UK. Earth & Environmental Science Transactions of the Royal Society of Edinburgh 110: 495–554. Crossref
Linnaeus, C. 1758. Systema nature per regna tria naturae, secundum classes, ordines, genera, species, cum caracteribus, differentiis, synonymi, locis. Tomus I. Editio decima, reformata. L. Salvii, Holmiae [= Stockholm]. Crossref
Loew, H. 1845. Dipterologische Beitrage. In: F.G. Kiessling (ed.), Zu der offentlichen Prufung der Schuler des Koniglichen Friedrich-Wilhelms-Gymnasiums zu Posen, 1–50. W. Decker, Poznań.
Loew, H. 1850. Über den Bernstein Und die Bernsteinfauna. In: H. Loew (ed.), Program der Keiserischen Realschule Meseritz, 1–44. E.S. Mittler, Berlin. Crossref
McAlpine, J.F.B., Peterson, B., Shewell, G., Teskey, H., Vockeroth, J., and Wood, D. 1981. Manual of Nearctic Diptera. Vol. 1. Agriculture Canada Research Branch Monograph 27: 1–674.
Oosterbroek, P. 2025. CCW: Catalogue of the Craneflies of the World (Diptera, Tipuloidea: Pediciidae, Limoniidae, Cylindrotomidae, Tipulidae) https://ccw.naturalis.nl/stats.php, http://nlbif.eti.uva.nl/ccw/index.php (accessed 27 November 2025).
Oosterbroek, P. and Theowald, B. 1991. Phylogeny of the Tipuloidea based on characters of larvae and pupae (Diptera, Nematocera) with an index to the literature except Tipulidae. Tijdschrift voor Entomologie 134: 211–267.
Penney, D. 2010. Dominican amber. In: D. Penney (ed.), Biodiversity in Fossils in Amber from Major World Deposits, 22–41. Sri Scientific Press, Manchester.
Podenas, S. 2011. Styringomyia Loew, 1845 (Diptera, Limoniidae) of Ghana. Transactions of the American Entomological Society 137: 283–295. Crossref
Podenas, S. and Poinar, G.O. 1999. New crane flies (Diptera: Limoniidae) from Dominican amber. Proceedings of the Entomological Society of Washington 101: 595–610.
Podenas, S. and Poinar, G.O. 2001. New crane flies (Diptera: Tipulidae, Limoniidae) from Dominican and Mexican amber. Proceedings of the Entomological Society of Washington 103: 863–878.
Ribeiro, G.C. 2003. Systematics of the Neotropical species of Styringomyia Loew (Diptera: Tipulomorpha: Limoniidae). Zootaxa 253: 1–35. Crossref
Rohdendorf, B.B. 1961. The most ancient infraorders of Diptera from the Triassic of central Asia. Journal of Palaeontology 3: 90–100. [In Russian].
Rondani, C. 1841. Progetto di una classificazione in famiglie degli insetti ditteri Europei. Memoria terza per servir alla ditterologia italiana. 28 pp. Donati, Parma.
Ross, A.J., Bojarski, B., and Szwedo, J. 2024 (in press). A critical review of the age of Baltic amber from the Samland Peninsula, Russia. Earth and Environmental Science Transactions of the Royal Society of Edinburgh.
Ross, A.J., Zhou, J., Hoffeins, C., and Crighton, B. 2024. The first fossil chylizine fly (Diptera: Psilidae) with a discussion on the ages of related fossil Acalyptratae. Insect Systematics and Diversity 8: 4. Crossref
Seyfullah, L.J., Beimforde, C., Corso, J.D., Perrichot, V., Rikkinen, J., and Schmidt, A.R. 2018. Production and preservation of resins– past and present. Biological Reviews 93: 1684–1714. Crossref
Speiser, P. 1909. Orthoptera. Nematocera. In: Y. Sjöstedt (ed.), Wissenschaftliche Ergebnisse der Schwedischen zoologischen Expedition nach dem Kilimandjaro, dem Meru und den umgebenden Massaisteppen Deutsch-Ostafrikas 1905–1906, 31–65. Almoqvist & Wiksells, Uppsala.
Theischinger, G., Billingham, Z.D., and Martin, J. 2018. The genus Styringomyia Loew in Australia (Diptera: Tipuloidea: Limoniidae). Linzer biologische Beiträge 50: 1587–1633.
Acta Palaeontol. Pol. 71 (1): 119–132, 2026
http://doi.org/10.4202/app.01305.2025